GDF-3 (previously called Vgr-2) is a TGF-beta superfamily member belonging to the growth/differentiation factor family (1, 2). GDF-3 is expressed in undifferentiated embryonic stem (ES) cells, adipose tissue and the brain (2-4). In ES cells, it maintains pluripotency and influences early cell fate decisions (5, 6). For example, frog embryos injected with GDF-3 develop a secondary dorsal axis and deletion of mouse GDF-3 can produce defects in the anterior visceral endoderm of the pre‑gastrulation embryo (5, 6). In adipocytes, GDF-3 is induced by a high fat diet and promotes adipogenesis (3). GDF-3 has been reported to oppose BMP’s functions and to have a nodal-like activity in early development (1). The 366 amino acid (aa) mouse GDF-3 contains a 22 aa signal sequence, a 230 aa propeptide and a 114 aa mature protein that contains one potential N-glycosylation site. Most of GDF-3 is present as the prepro form, while the mature GDF-3 is presumably the secreted, active form (1). The mature protein contains the cysteine-knot structure that is conserved throughout family members. Since it lacks the fourth cysteine, which is responsible for the formation of inter-molecular disulfide bond, GDF-3 may exist as a non-covalent homodimer. Mature mouse GDF-3 shares 90%, 83%, and 83% aa identity with rat, human and canine GDF-3, respectively. Among family members, mature GDF-3 is most similar to mouse BMP-6 (45% aa identity) and Xenopus VG‑1 (52% aa identity).
Human/Mouse GDF‑3 Antibody
R&D Systems | Catalog # AF958


Key Product Details
Species Reactivity
Validated:
Human, Mouse
Cited:
Mouse, Frog - Xenopus (African Clawed Frog)
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse GDF-3
Ala253-Gly366
Accession # Q07104
Ala253-Gly366
Accession # Q07104
Specificity
Detects human and mouse GDF-3 in direct ELISAs and Western blots. In Western blots, less than 5% cross-reactivity with recombinant mouse (rm) GDF‑5, rmGDF‑6, rmGDF‑7, rmGDF‑8, and rmGDF‑9 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse GDF‑3 Antibody
Detection of Human GDF‑3 by Western Blot.
Western blot shows lysates of BG01V human embryonic stem cells. PVDF membrane was probed with 1.5 µg/mL of Goat Anti-Human/Mouse GDF-3 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF958) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for GDF-3 at approximately 42 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.GDF‑3 in BG01V Human Stem Cells.
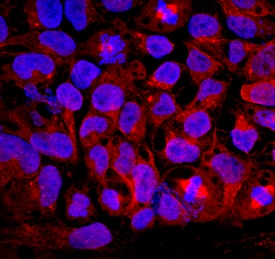
GDF-3 was detected in immersion fixed BG01V human embryonic stem cells using Goat Anti-Human/Mouse GDF-3 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF958) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.GDF‑3 in Mouse Thymus.
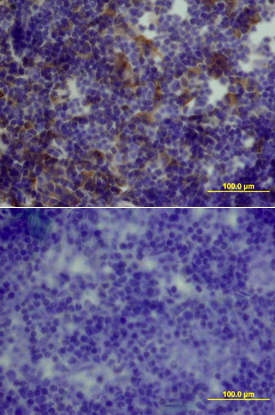
GDF-3 was detected in perfusion fixed frozen sections of mouse thymus using Mouse GDF-3 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF958) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Lower panel shows a lack of labeling if primary antibodies are omitted and tissue is stained only with secondary antibody followed by incubation with detection reagents. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.GDF‑3 in Mouse Thymus.
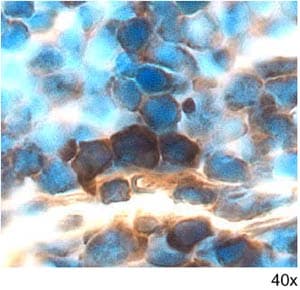
GDF-3 was detected in perfusion fixed frozen sections of mouse thymus using 5 µg/mL Mouse GDF-3 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF958) overnight at 4 °C. Tissue was stained with the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of GDF-3 by Immunohistochemistry
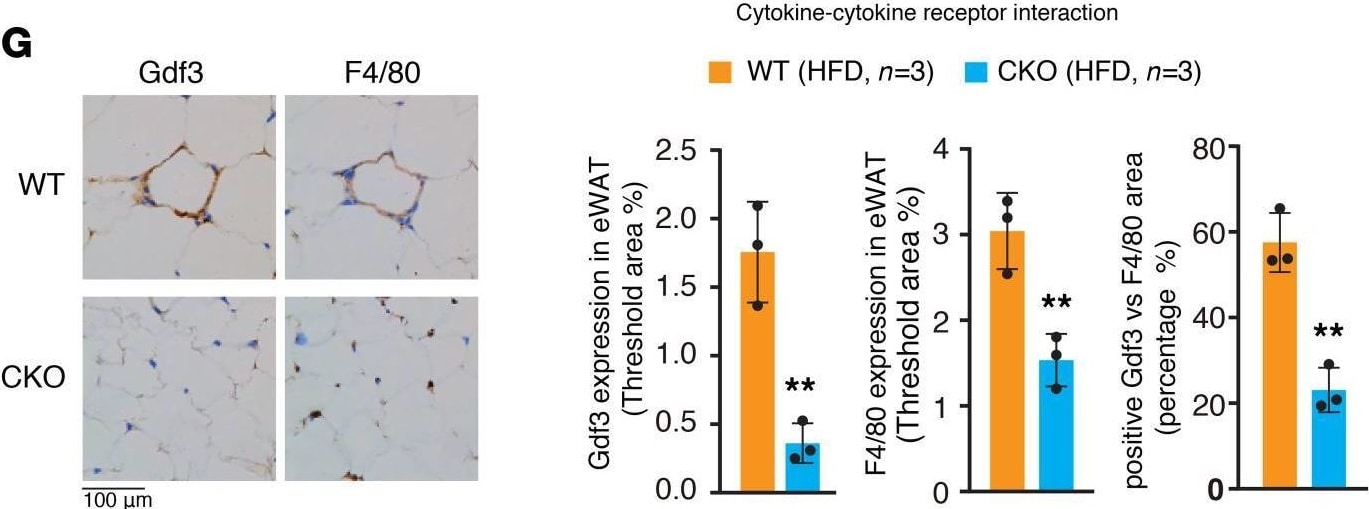
Brd4-CKO mice had reduced expression of Gdf3 in ATMs.(A) PCA of RNA-Seq data derived from CD11b+ ATMs of WT and Brd4-CKO mice fed a HFD for 20 weeks. (B) Volcano plot of mRNA-Seq analysis in ATMs as indicated in A. Brown dots represent genes increased in ATMs of Brd4-CKO mice vs. WT mice (fold change ≥ 2, adjusted P value ≤ 0.001, calculated by raw count value). Blue dots represent genes decreased in ATMs of Brd4-CKO mice vs. WT mice (fold change ≥ 2, adjusted P value ≤ 0.001, calculated by raw count value). Gray dots represent genes without significantly altered expression. Clusters of significantly altered genes (fold change ≥ 2, adjusted P value ≤ 0.001, calculated by raw count value) were identified using gene ontology terms (C) and KEGG pathways (D). (E) Heat map of the relative expression levels (scaled Z-score) of cytokine-cytokine receptor interaction-related genes clustered in (D). (F) mRNA levels of Gdf3 in CD11b+ ATMs isolated from WT or Brd4-CKO mice fed a HFD for 20 weeks. (G) Left panel: Gdf3 or F4/80 IHC staining of eWAT of WT or Brd4-CKO mice fed a HFD for 20 weeks. Right panel: statistical analysis of Gdf3-positive or F4/80-positive area percentage, the ratio of Gdf3-positive cells in F4/80-positive cells in eWAT of WT and Brd4-CKO mice fed a HFD. Data are mean and SD and are determined by an unpaired 2-tailed Student’s t test. n = 3 mice. **P < 0.01. Brd4-CKO, myeloid lineage-specific Brd4 knockout; HFD, high-fat diet–induced; eWAT, epididymal WAT; ATMs, adipose tissue macrophages; PCA, principal component analysis. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33830083), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse GDF‑3 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed BG01V human embryonic stem cells
Sample: Immersion fixed BG01V human embryonic stem cells
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse thymus
Sample: Perfusion fixed frozen sections of mouse thymus
Western Blot
1.5 µg/mL
Sample:
Sample:
BG01V human embryonic stem cells
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: GDF-3
References
- Levine, A.J. and A.H. Brivanlou (2006) Cell Cycle 5:1069.
- McPherron, A.C. and S-J. Lee (1993) J. Biol. Chem. 268:3444.
- Wang, W. et al. (2004) Biochem. Biophys. Res. Comm. 321:1024.
- Hexige, S. et al. (2005) Neurosci. Lett. 389:83.
- Levine, A.J. and A.H. Brivanlou (2005) Development 133:209.
- Chen, C. et al. (2006) Development 133:319.
Long Name
Growth Differentiation Factor 3
Alternate Names
GDF3, Vgr-2
Gene Symbol
GDF3
UniProt
Additional GDF-3 Products
Product Documents for Human/Mouse GDF‑3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse GDF‑3 Antibody
For research use only
Citations for Human/Mouse GDF‑3 Antibody
Customer Reviews for Human/Mouse GDF‑3 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse GDF‑3 Antibody and earn rewards!
Have you used Human/Mouse GDF‑3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...