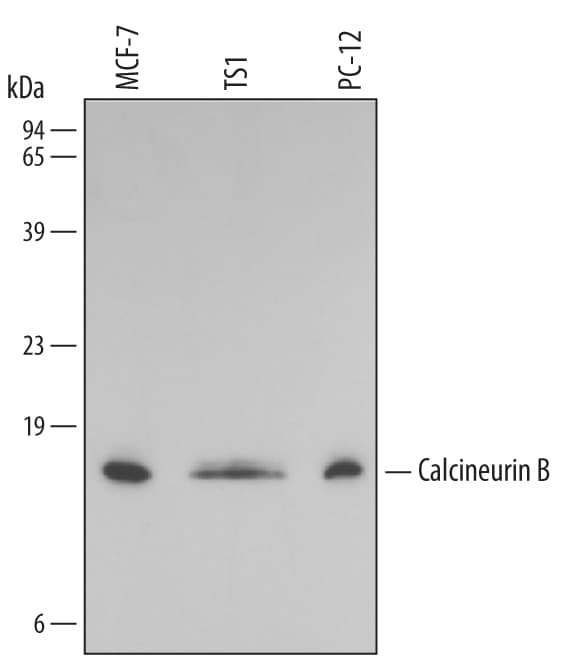
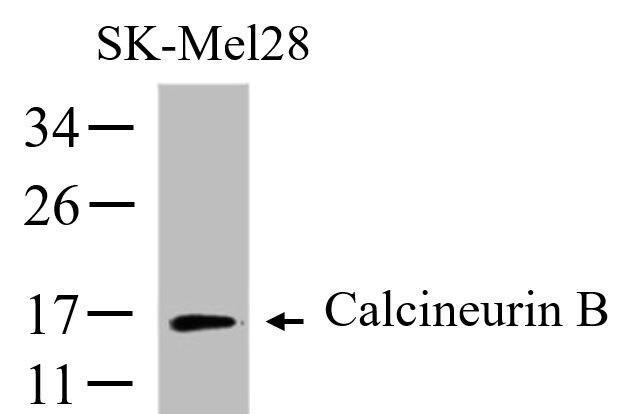
Background
Calcineurin, also called Protein Phosphatase 2B,
PP2B, PPP2B, Protein Phosphatase 3, and PPP3, is
an enzyme that dephosphorylates serine and
threonine residues in proteins. It is a heterodimer of
a 59,000 dalton catalytic A subunit and a
1,900 dalton regulatory B subunit that is activated
by the binding of calcium ions and calmodulin (1).
Calcineurin is expressed in many tissues, but its
levels are highest in the brain, where it may play a
role in learning and memory (2). It has many
substrates, including NFAT, a transcription factor
that is activated by dephosphorylation (3).
Complexes of the immunosuppressants cyclosporin
and FK506 with immunophilin proteins such as
cyclophilin and FKBP12 are potent and specific
inhibitors of Calcineurin activity (4). Alterations in
Calcineurin activity are suspected to play a role in
cardiac hypertrophy (5) and graft versus host
disease in organ transplantation (6).
References:
1. Stemmer, P.M and C.B. Klee (1994) Biochemistry 33:6859.
2. Zeng H. et al. (2001) Cell 107:617.
3. Okamura, H. et al. (2000) Molecular Cell 6:539.
4. Liu, J. et al. (1992) Biochemistry 31:3896.
5. Molkentin, J.D. (2000) Circulation Research 87:731.
6. Sanquer, S.A. et al. (2004) Transplantation 77:854.