Semaphorin 3E (Sema3E; previously SemaH) is one of six Class 3 (secreted) semaphorins which in the human share 40-50% amino acid (aa) identity. Class 3 semaphorins are potent chemorepellents that function in axon guidance and/or vascular tip cell guidance during development (1). Sema3E is highly expressed by a subset of motor neurons in developing somites, where it acts as a repulsive cue for PlexinD1-expressing endothelial cells of adjacent intersomitic vessels (2, 3). Crystal structures of semaphorins reveal that the 500 aa N-terminal Sema domain forms a seven-blade b-propeller similar to that found in integrin molecules; 14 conserved cysteine residues and one or more N-glycosylation sites are thought critical for forming the secondary structure (4). C-terminal to the Sema domain, Sema3E has a consensus sequence for furin cleavage which, when used, creates a 61kDa form that does not dimerize and is highly expressed in tumor cell lines with metastatic potential (5, 6). Further C-terminal are a cysteine-knot plexin/semaphorin/integrin (PSI) domain, an Ig-like domain, a cysteine for dimerization and a basic domain containing another furin site. Dimerization and cleavage at the C-terminal site are required for repulsing activity of class 3 semaphorins (7). Human Sema3E shares 90%, 85% and 57% aa identity with mouse, cow and dog Sema3E, respectively. Like other semaphorins, Sema3E signaling is transduced by a transmembrane Plexin dimer, which also has a Sema domain and is coupled to kinase pathways. Unlike other Class 3 semaphorins, Sema3E binds directly to its plexin and does not require interaction with a neuropilin for activity (7). Genetic disruption of either Sema3E or PlexinD1 creates mouse mutants with excessive and disorganized vascular growth and branching, indicating the importance of this ligand-receptor pair for vascular guidance (3, 8).
Human/Mouse Semaphorin 3E Antibody
R&D Systems | Catalog # AF3239

Key Product Details
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Rat, Primate - Chlorocebus aethiops (African Green Monkey), Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Semaphorin 3E
Thr25-Ser775 (Arg557Ala and Arg560Ala)
Accession # O15041
Thr25-Ser775 (Arg557Ala and Arg560Ala)
Accession # O15041
Specificity
Detects human Semaphorin 3E in direct ELISAs and Western blots. In direct ELISAs, less than 10% cross-reactivity with recombinant human (rh) Semaphorin 3A, rhSemaphorin 3C, rhSemaphorin 3F, and rhSemaphorin 3G is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse Semaphorin 3E Antibody
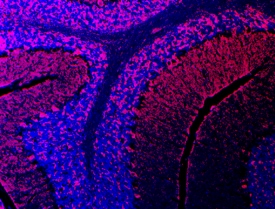
Semaphorin 3E in Mouse Brain.
Semaphorin 3E was detected in immersion fixed frozen sections of adult mouse brain using Goat Anti-Human Semaphorin 3E Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3239) at 10 µg/mL overnight at 4 °C. Tissue was stained using the Northern-Lights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cerebellum. View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Semaphorin 3E in Mouse Lung.
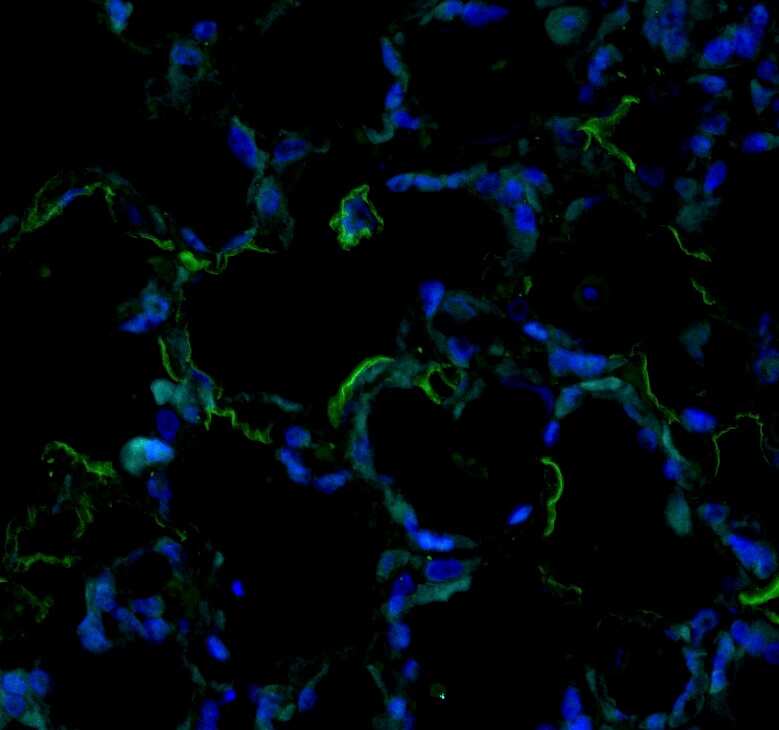
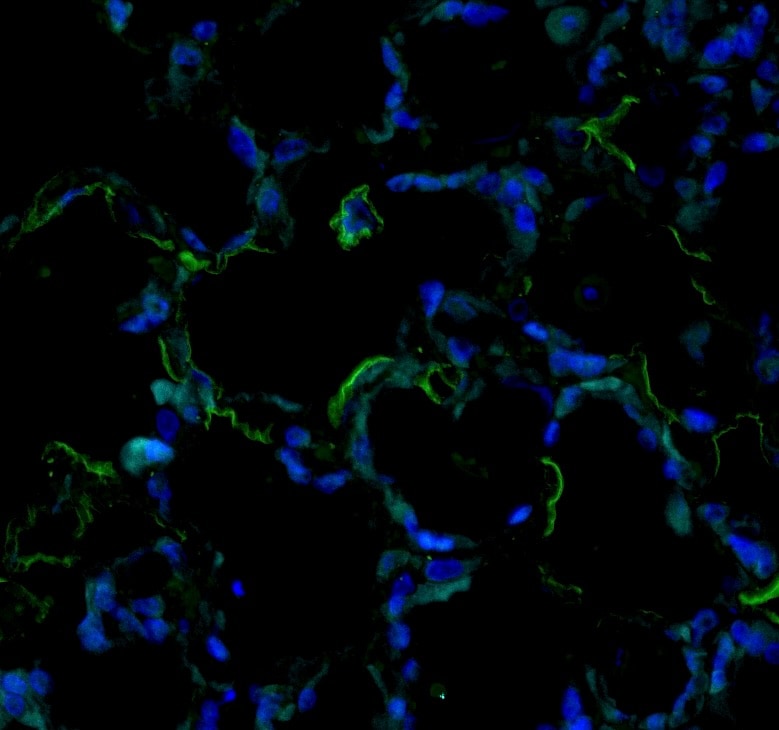
Immunohistochemistry (paraffin-embedded) on Native Adult rat lung tissue. Sema33 stained green, DAPI (blue). Image from a verified customer review.Applications for Human/Mouse Semaphorin 3E Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed MDA-MB-453 human breast cancer cell line
Sample: Immersion fixed MDA-MB-453 human breast cancer cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of adult mouse brain
Sample: Immersion fixed frozen sections of adult mouse brain
Western Blot
0.1 µg/mL
Sample: Recombinant Human Semaphorin 3E (Catalog # 3239-S3)
Sample: Recombinant Human Semaphorin 3E (Catalog # 3239-S3)
Reviewed Applications
Read 1 review rated 4 using AF3239 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Semaphorin 3E
References
- Eichmann, A. et al. (2005) Genes Dev. 19:1013.
- Cohen, S. et al. (2005) Eur. J. Neurosci. 21:1767.
- Gu, C. et al. (2005) Science 307:265.
- Gherardi, E. et al. (2004) Curr. Opin. Struct. Biol. 14:669.
- Christensen, C. et al. (1998) Cancer Res. 58:1238.
- Christensen, C. et al. (2005) Cancer Res. 65:6167.
- Adams, R. H. et al. (1997) EMBO J. 16:6077.
- Gitler, A. D. et al. (2004) Developmental Cell 7:107.
Alternate Names
Sema3E, SEMAH
Gene Symbol
SEMA3E
UniProt
Additional Semaphorin 3E Products
Product Documents for Human/Mouse Semaphorin 3E Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Semaphorin 3E Antibody
For research use only
Related Research Areas
Citations for Human/Mouse Semaphorin 3E Antibody
Customer Reviews for Human/Mouse Semaphorin 3E Antibody (1)
4 out of 5
1 Customer Rating
Have you used Human/Mouse Semaphorin 3E Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunohistochemistry (paraffin-embedded) on Native Adult rat lung tissue. Sema33 stained green, DAPI (blue).Sample Tested: Lung tissueSpecies: RatVerified Customer | Posted 07/19/2025Bio-Techne ResponseThis review reflects a new species or application tested on a primary antibody.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...