Osteoadherin (OSAD), also known as Osteomodulin, is an extracellular matrix keratan sulfate proteoglycan that belongs to the class II subfamily of small leucine-rich proteoglycans (SLRP). LRR motifs consist of approximately 20‑30 amino acids (aa) with conserved leucine spacing, folded into a structure with one beta -sheet and one alpha -helix (1, 2). The human OSAD cDNA encodes a 421 aa precursor that contains a 20 aa signal sequence and eleven tandem leucine rich repeats (3). Human OSAD shares 80‑84% aa sequence identity with bovine, mouse, and rat OSAD. Human OSAD shares 32‑35% aa sequence identity with human class II SLRPs Fibromodulin, Keratocan, Lumican, and PRELP. Bovine, mouse, and rat OSAD are expressed as 60‑85 kDa molecules, although the amino acid sequence for each predicts a size of 46‑47 kDa. The primary difference is due to the presence of extensive N-linked glycosylation that can also vary between tissues of the same species (4, 5). Human OSAD is expressed as an even larger 110 kDa molecule in teeth (6). OSAD contains eight sulfated tyrosine residues (4, 7) and is distinguished from other class II SLRPs by the presence of an approximately 70 aa C-terminal acidic domain (3). OSAD is expressed by fetal and adult osteoblasts but is not detectable in cartilage or tendon (3, 4, 8). In dental tissue, OSAD is expressed by odontoblasts and ameloblasts (5, 9‑11) and is involved in the mineralization of bone and teeth (5, 11, 12). OSAD promotes the adhesion of osteoblasts and odontoblasts to the surrounding matrix, an interaction that is mediated by Integrin alpha V beta 3 (4, 6).

Key Product Details
Species Reactivity
Human
Applications
Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 806001
Loading...
Product Specifications
Immunogen
HEK293 human embryonic kidney cell line transfected with human Osteoadherin/OSAD
Gln21-Gln421
Accession # Q99983
Gln21-Gln421
Accession # Q99983
Specificity
Detects human Osteoadherin/OSAD in ELISA.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human Osteoadherin/OSAD Antibody
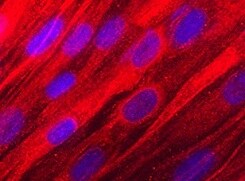
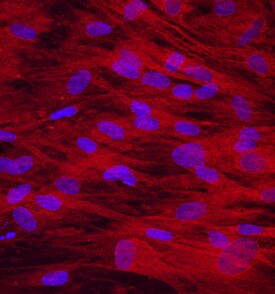
Osteoadherin/OSAD in Human Mesenchymal Stem Cells.
Osteoadherin/OSAD was detected in immersion fixed human mesenchymal stem cells differentiated into osteocytes using Mouse Anti-Human Osteoadherin/OSAD Monoclonal Antibody (Catalog # MAB2884) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Stem Cells on Coverslips.Applications for Human Osteoadherin/OSAD Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed human mesenchymal stem cells differentiated into osteocytes
Sample: Immersion fixed human mesenchymal stem cells differentiated into osteocytes
Reviewed Applications
Read 1 review rated 5 using MAB2884 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Osteoadherin/OSAD
References
- Matsushima, N. et al. (2000) Proteins 38:210.
- Kobe, B. and A.V. Kajava (2001) Curr. Opin. Struct. Biol. 11:725.
- Sommarin, Y. et al. (1998) J. Biol. Chem. 273:16723.
- Wendel, M. et al. (1998) J. Cell Biol. 141:839.
- Hultenby, P.U. et al. (2003) Eur. J. Oral Sci. 111:128.
- Lucchini, M. et al. (2004) J. Dent. Res. 83:552.
- Onnerfjord, P. et al. (2004) J. Biol. Chem. 279:26
- Shen, Z. et al. (1999) Matrix Biol. 18:533.
- Buchaille, R. et al. (2000) Bone 27:265.
- Buchaille, R. et al. (2000) Matrix Biol. 19:421.
- Couble, M.L. et al. (2004) Histochem. Cell Biol. 121:47.
- Ramstad, V.E. et al. (2003) Calcif. Tissue Int. 72:57.
Alternate Names
OMD, OSAD, SLRR2C
Entrez Gene IDs
4958 (Human)
Gene Symbol
OMD
UniProt
Additional Osteoadherin/OSAD Products
Product Documents for Human Osteoadherin/OSAD Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Osteoadherin/OSAD Antibody
For research use only
Related Research Areas
Customer Reviews for Human Osteoadherin/OSAD Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human Osteoadherin/OSAD Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Mesenchymal stem cellsSpecies: HumanVerified Customer | Posted 01/06/2022
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...