Programmed Death-1 (PD-1) is a type I transmembrane protein belonging to the CD28/CTLA-4 family of immunoreceptors that mediate signals for regulating immune responses (1). Members of the CD28/CTLA-4 family have been shown to either promote T cell activation (CD28 and ICOS) or downregulate T cell activation (CTLA-4 and PD-1) (2). PD-1 is expressed on activated T cells, B cells, myeloid cells, and on a subset of thymocytes. In vitro, ligation of PD-1 inhibits TCR-mediated T cell proliferation and production of IL-1, IL-4, IL-10, and IFN-gamma. In addition, PD-1 ligation also inhibits BCR mediated signaling. PD-1 deficient mice have a defect in peripheral tolerance and spontaneously develop autoimmune diseases (2, 3). Two B7 family proteins, PD-L1 (also called B7-H1) and PD-L2 (also known as B7-DC), have been identified as PD-1 ligands. Unlike other B7 family proteins, both PD‑L1 and PD‑L2 are expressed in a wide variety of normal tissues including heart, placenta, and activated spleens (4). The wide expression of PD-L1 and PD-L2 and the inhibitor effects on PD-1 ligation indicate that PD-1 might be involved in the regulation of peripheral tolerance and may help prevent autoimmune diseases (2). The human PD-1 gene encodes a 288 amino acid (aa) protein with a putative 20 aa signal peptide, a 148 aa extracellular region with one immunoglobulin-like V‑type domain, a 24 aa transmembrane domain, and a 95 aa cytoplasmic region. The cytoplasmic tail contains two tyrosine residues that form the Immunoreceptor Tyrosine-based Inhibitory Motif (ITIM) and Immunoreceptor Tyrosine-based Switch Motif (ITSM) that are important in mediating PD-1 signaling. Mouse and human PD-1 share approximately 60% aa sequence identity (4).
Human PD‑1 Antibody
R&D Systems | Catalog # MAB10861

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Met1-Gln167
Accession # Q15116
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human PD‑1 Antibody
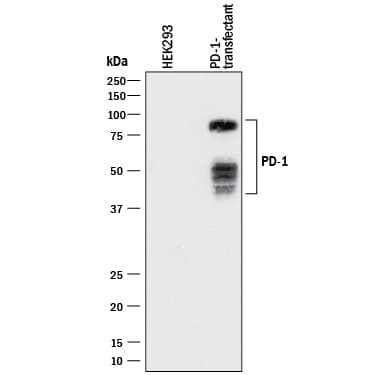
Detection of Human PD‑1 by Western Blot.
Western blot shows lysates of HEK293 human embryonic kidney cell line either mock transfected or transfected with human PD-1. PVDF membrane was probed with 1 µg/mL of Mouse Anti-Human PD-1 Monoclonal Antibody (Catalog # MAB10861) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). Specific bands were detected for PD-1 at approximately 40-80 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of PD‑1 in Human PBMCs by Flow Cytometry.
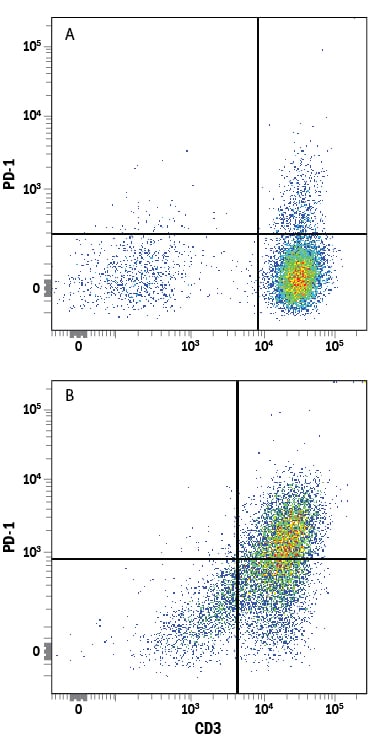
Human peripheral blood mononuclear cells (PBMCs) either (A) untreated or (B) treated with 5 μg/mL PHA for 2 days were stained with Mouse Anti-Human PD-1 Monoclonal Antibody (Catalog # MAB10861) followed by Allophycocyanin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0101B) and Mouse Anti-Human CD3e PE-conjugated Monoclonal Antibody (Catalog # FAB100P). Quadrant markers were set based on control antibody staining (Catalog # MAB002).B7-H1 Binding to PD-1-transfected HEK293 Human Cell Line is Blocked by Human PD-1 Antibody.
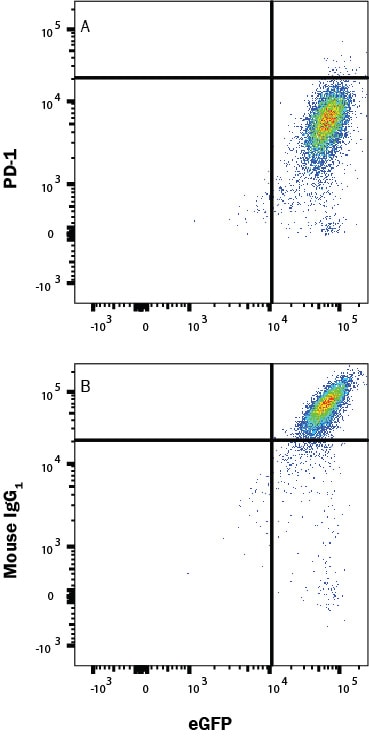
In a functional flow cytometry test, biotinylated recombinant human B7-H1 (10 ng/mL, BT156) binds to HEK293 human embryonic kidney cell line transfected with human PD-1 and eGFP. (A) Binding is completely blocked by 2.5 µg/mL of Mouse Anti-Human PD-1 Monoclonal Antibody (Catalog # MAB10861) but not by (B) 2.5 µg/mL of Mouse IgG1 Isotype Control (MAB002). Protein binding was detected by staining cells with Streptavidin-APC (F0050).Detection of PD-1 by Western Blot
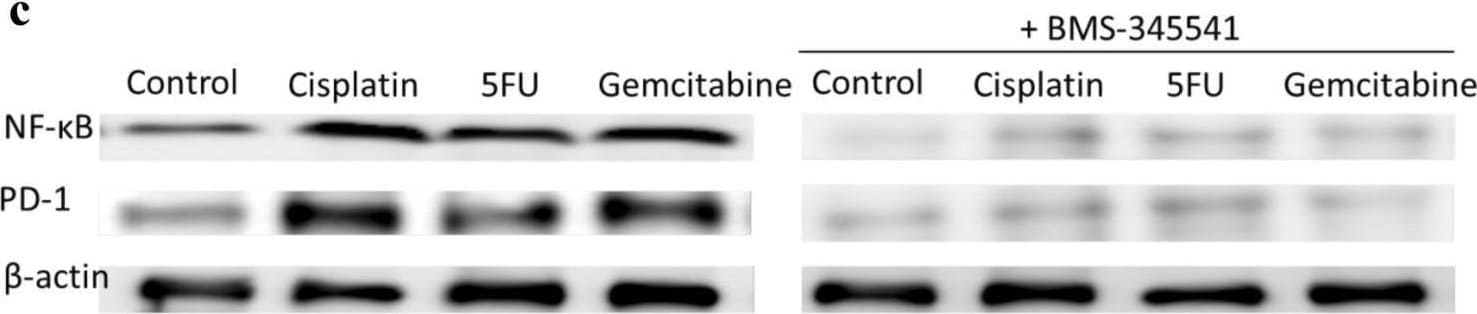
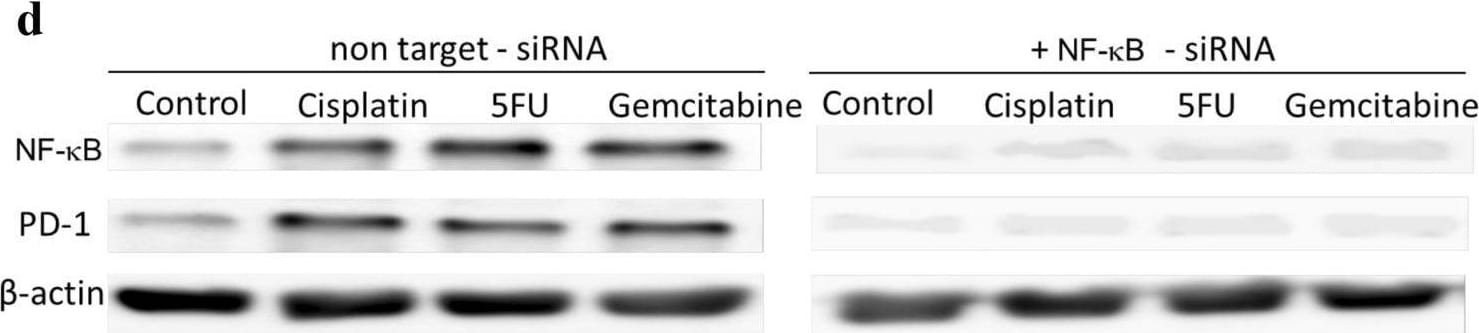
Chemotherapeutics induce PD-L1 expression in NPC cells and PD-1 expression in NK cells via upregulation of NF-kappa B. NPC cells a or NK cells c were incubated with the NF-kappa B inhibitor BMS-345541 for 1 h before incubation with chemotherapeutics. NPC cells were transfected with NF-kappa B siRNA b or NK cells with NF-kappa B siRNA d for 16 h and then incubated with chemotherapeutics. Expression of NF-kappa B, PD-L1 and PD-1 was analyzed by immunoblot Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32737537), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of PD-1 by Western Blot
Chemotherapeutics induce PD-L1 expression in NPC cells and PD-1 expression in NK cells via upregulation of NF-kappa B. NPC cells a or NK cells c were incubated with the NF-kappa B inhibitor BMS-345541 for 1 h before incubation with chemotherapeutics. NPC cells were transfected with NF-kappa B siRNA b or NK cells with NF-kappa B siRNA d for 16 h and then incubated with chemotherapeutics. Expression of NF-kappa B, PD-L1 and PD-1 was analyzed by immunoblot Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32737537), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human PD‑1 Antibody
Blockade of Receptor-ligand Interaction
CyTOF-ready
Flow Cytometry
Sample: Human peripheral blood mononuclear cells (PBMCs) treated with PHA
Western Blot
Sample: HEK293 human embryonic kidney cell line transfected with human PD-1
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: PD-1
References
- Ishida, Y. et al. (1992) EMBO J. 11:3887.
- Nishimura, H. and T. Honjo (2001) Trends in Immunol. 22:265.
- Latchman, Y. et al. (2001) Nature Immun. 2:261.
- Carreno, B.M. and M. Collins (2002) Annu. Rev. Immunol. 20:29.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional PD-1 Products
Product Documents for Human PD‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human PD‑1 Antibody
For research use only
Citations for Human PD‑1 Antibody
Customer Reviews for Human PD‑1 Antibody
There are currently no reviews for this product. Be the first to review Human PD‑1 Antibody and earn rewards!
Have you used Human PD‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Human PD‑1 Antibody
-
Q: What is the light chain of Human PD-1 Antibody, Catalog #s MAB10861 and FAB10861, Clone # 913429?
A: Catalog #s MAB10861 and FAB10861 have a kappa light chain.