Advanced glycation endproducts (AGE) are adducts formed by the non-enzymatic glycation or oxidation of macromolecules (1). AGE forms during aging and its formation is accelerated under pathophysiologic states such as diabetes, Alzheimer’s disease, renal failure and immune/inflammatory disorders. Receptor for Advanced Glycation Endoproducts (RAGE), named for its ability to bind AGE, is a multi-ligand receptor belonging the immunoglobulin (Ig) superfamily. Besides AGE, RAGE binds amyloid beta -peptide, S100/calgranulin family proteins, high mobility group B1 (HMGB1, also know as amphoterin) and leukocyte integrins (1, 2). The human RAGE gene encodes a 404 amino acid residues (aa) type I transmembrane glycoprotein with a 22 aa signal peptide, a 320 aa extracellular domain containing an Ig-like V-type domain and two Ig-like Ce-type domains, a 21 aa transmembrane domain and a 41 aa cytoplasmic domain (3). The V-type domain and the cytoplasmic domain are important for ligand binding and for intracellular signaling, respectively. Two alternative splice variants, lacking the V-type domain or the cytoplasmic tail, are known (1, 4). RAGE is highly expressed in the embryonic central nervous system (5). In adult tissues, RAGE is expressed at low levels in multiple tissues including endothelial and smooth muscle cells, mononuclear phagocytes, pericytes, microglia, neurons, cardiac myocytes and hepatocytes (6). The expression of RAGE is upregulated upon ligand interaction. Depending on the cellular context and interacting ligand, RAGE activation can trigger differential signaling pathways that affect divergent pathways of gene expression (1, 7). RAGE activation modulates varied essential cellular responses (including inflammation, immunity, proliferation, cellular adhesion and migration) that contribute to cellular dysfunction associated with chronic diseases such as diabetes, cancer, amyloidoses and immune or inflammatory disorders (1).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 176907
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human RAGE
Gln24-Ala344
Accession # Q15109
Gln24-Ala344
Accession # Q15109
Specificity
Detects human RAGE in direct ELISAs and Western blots. In Western blots, this antibody shows no cross-reactivity with recombinant rat (rr) RAGE or rmRAGE.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human RAGE Antibody
Detection of Human RAGE by Western Blot.
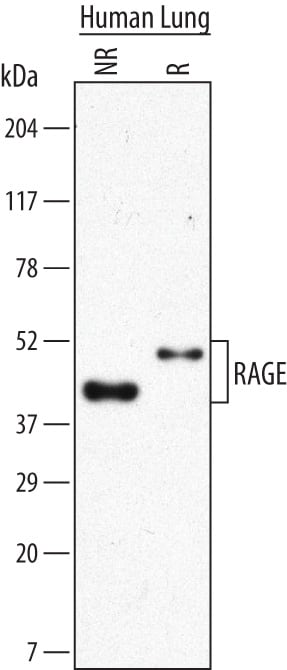
Western blot shows lysates of human lung tissue. PVDF Membrane was probed with 2 µg/mL of Mouse Anti-Human RAGE Monoclonal Antibody (Catalog # MAB1145) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF007). Specific bands were detected for RAGE at approximately 43 kDa under non-reducing (NR) conditions and 50 kDa under reducing (R) conditions (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.RAGE in Human Liver Cancer Tissue.
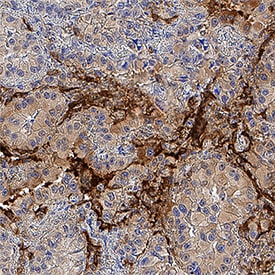
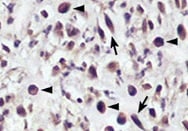
RAGE was detected in immersion fixed paraffin-embedded sections of human liver cancer tissue using Mouse Anti-Human RAGE Monoclonal Antibody (Catalog # MAB1145) at 0.5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC001). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cell nuclei. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of RAGE by Western Blot
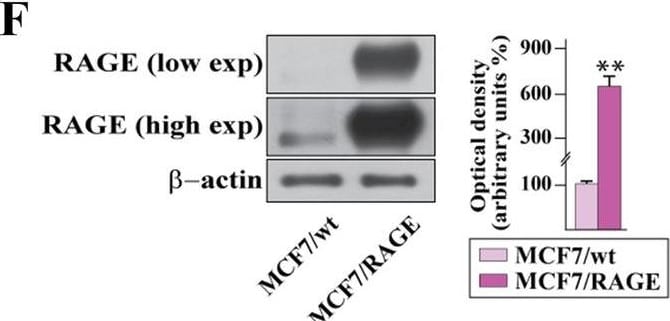
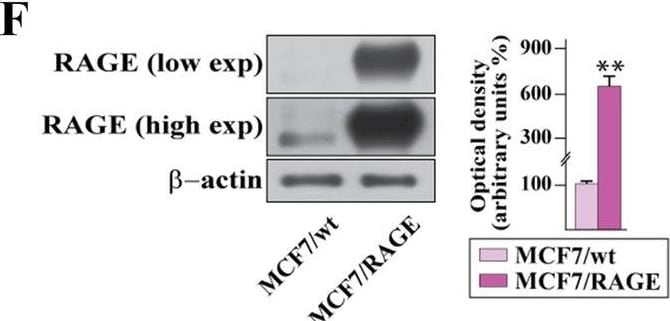
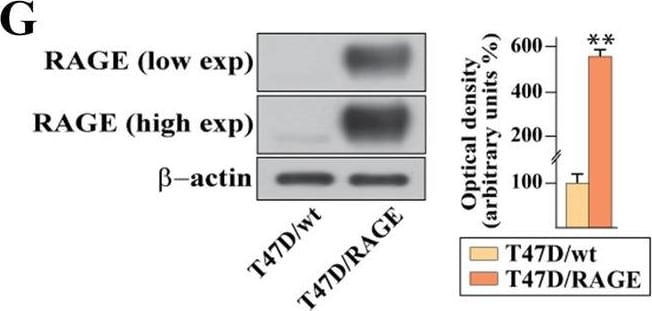
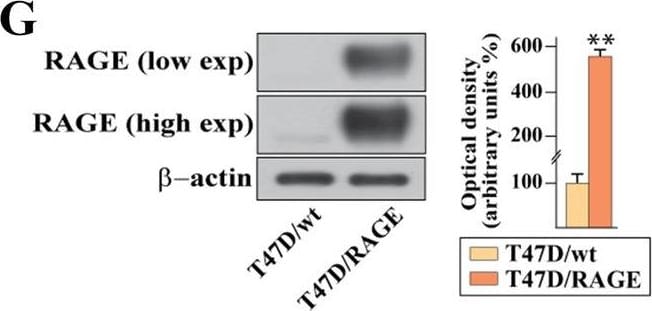
Validation of RAGE-overexpressing BC cells. A Kaplan–Meier plot showing the association of RAGE mRNA expression with the progression free interval (PFI) of the TCGA ER-positive BC patients. Samples were divided into RAGE high and low groups using the optimum cut-off. B-C Morphological appearance of wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells in phase-contrast microscopy; scale bar: 250 μm. Enlarged details are shown in the separate boxes. Flow cytometric histograms in RAGE-overexpressing compared with wild type MCF7 (D) and T47D (E) cells. FITC, fluorescein isothiocyanate. Side panels show the percentage of RAGE-positive cells. F-G Immunoblots of RAGE in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells. Side panels show densitometric analysis of the blots normalized to beta -actin, which was used as a loading control. Values represent the mean ± SD of three independent experiments performed in triplicate. H-I Evaluation of RAGE protein expression (green signal) by immunofluorescence experiment in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells; nuclei were stained by DAPI (blue signal). The images shown represent 10 random fields from three independent experiments. Scale bar: 25 μm. Side panels represent corrected total cell fluorescence (CTCF), which was calculated on at least 10 pictures from each sample. (*) indicates p < 0.05; (**) indicates p < 0.01; (**) indicates p < 0.001 Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37434266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of RAGE by Western Blot
Validation of RAGE-overexpressing BC cells. A Kaplan–Meier plot showing the association of RAGE mRNA expression with the progression free interval (PFI) of the TCGA ER-positive BC patients. Samples were divided into RAGE high and low groups using the optimum cut-off. B-C Morphological appearance of wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells in phase-contrast microscopy; scale bar: 250 μm. Enlarged details are shown in the separate boxes. Flow cytometric histograms in RAGE-overexpressing compared with wild type MCF7 (D) and T47D (E) cells. FITC, fluorescein isothiocyanate. Side panels show the percentage of RAGE-positive cells. F-G Immunoblots of RAGE in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells. Side panels show densitometric analysis of the blots normalized to beta -actin, which was used as a loading control. Values represent the mean ± SD of three independent experiments performed in triplicate. H-I Evaluation of RAGE protein expression (green signal) by immunofluorescence experiment in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells; nuclei were stained by DAPI (blue signal). The images shown represent 10 random fields from three independent experiments. Scale bar: 25 μm. Side panels represent corrected total cell fluorescence (CTCF), which was calculated on at least 10 pictures from each sample. (*) indicates p < 0.05; (**) indicates p < 0.01; (**) indicates p < 0.001 Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37434266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of RAGE by Western Blot
Validation of RAGE-overexpressing BC cells. A Kaplan–Meier plot showing the association of RAGE mRNA expression with the progression free interval (PFI) of the TCGA ER-positive BC patients. Samples were divided into RAGE high and low groups using the optimum cut-off. B-C Morphological appearance of wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells in phase-contrast microscopy; scale bar: 250 μm. Enlarged details are shown in the separate boxes. Flow cytometric histograms in RAGE-overexpressing compared with wild type MCF7 (D) and T47D (E) cells. FITC, fluorescein isothiocyanate. Side panels show the percentage of RAGE-positive cells. F-G Immunoblots of RAGE in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells. Side panels show densitometric analysis of the blots normalized to beta -actin, which was used as a loading control. Values represent the mean ± SD of three independent experiments performed in triplicate. H-I Evaluation of RAGE protein expression (green signal) by immunofluorescence experiment in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells; nuclei were stained by DAPI (blue signal). The images shown represent 10 random fields from three independent experiments. Scale bar: 25 μm. Side panels represent corrected total cell fluorescence (CTCF), which was calculated on at least 10 pictures from each sample. (*) indicates p < 0.05; (**) indicates p < 0.01; (**) indicates p < 0.001 Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37434266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of RAGE by Western Blot
Validation of RAGE-overexpressing BC cells. A Kaplan–Meier plot showing the association of RAGE mRNA expression with the progression free interval (PFI) of the TCGA ER-positive BC patients. Samples were divided into RAGE high and low groups using the optimum cut-off. B-C Morphological appearance of wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells in phase-contrast microscopy; scale bar: 250 μm. Enlarged details are shown in the separate boxes. Flow cytometric histograms in RAGE-overexpressing compared with wild type MCF7 (D) and T47D (E) cells. FITC, fluorescein isothiocyanate. Side panels show the percentage of RAGE-positive cells. F-G Immunoblots of RAGE in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells. Side panels show densitometric analysis of the blots normalized to beta -actin, which was used as a loading control. Values represent the mean ± SD of three independent experiments performed in triplicate. H-I Evaluation of RAGE protein expression (green signal) by immunofluorescence experiment in wild type (MCF7/wt and T47D/wt) and RAGE-overexpressing (MCF7/RAGE and T47D/RAGE) cells; nuclei were stained by DAPI (blue signal). The images shown represent 10 random fields from three independent experiments. Scale bar: 25 μm. Side panels represent corrected total cell fluorescence (CTCF), which was calculated on at least 10 pictures from each sample. (*) indicates p < 0.05; (**) indicates p < 0.01; (**) indicates p < 0.001 Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37434266), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human RAGE Antibody
Application
Recommended Usage
Immunohistochemistry
0.5-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human liver cancer tissue
Sample: Immersion fixed paraffin-embedded sections of human liver cancer tissue
Western Blot
2 µg/mL
Sample: Human lung tissue
Sample: Human lung tissue
Reviewed Applications
Read 1 review rated 5 using MAB1145 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: RAGE/AGER
References
- Schmidt, A. et al. (2001) J. Clin. Invest. 108:949.
- Chavakis, T. et al. (2003) J. Exp. Med. 198:507.
- Neeper, M. et al. (1992) J. Biol. Chem. 267:14998.
- Yonekura, H. et al. (2003) Biochem. J. 370:1097.
- Hori, O. et al. (1995) J. Biol. Chem. 270:25752.
- Brett, J. et al. (1993) Am. J. Pathol. 143:1699.
- Valencia, J.V. et al. (2004) Diabetes 53:743.
Long Name
Receptor for Advanced Glycation End Products
Alternate Names
AGER, SCARJ1
Gene Symbol
AGER
UniProt
Additional RAGE/AGER Products
Product Documents for Human RAGE Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human RAGE Antibody
For research use only
Citations for Human RAGE Antibody
Customer Reviews for Human RAGE Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human RAGE Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: wound tissueSpecies: HumanVerified Customer | Posted 10/23/2021
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars