Thrombospondin-4 (TSP-4) is a 140 kDa calcium-binding protein that interacts with other extracellular matrix molecules and modulates the activity of various cell types. TSP‑1 and -2 constitute subgroup A and form disulfide-linked homotrimers, whereas TSP-3, -4, and -5/COMP constitute subgroup B and form pentamers (1, 2). The human TSP-4 cDNA encodes a 961 amino acid (aa) precursor that includes a 26 aa signal sequence followed by an N-terminal heparin-binding domain, a coiled-coil motif, four EGF-like repeats, seven TSP type-3 repeats (one with an RGD motif), and a TSP C-terminal domain (3). Human TSP-4 shares 93% aa sequence identity with mouse and rat TSP-4. Within the TSP type-3 repeats and the TSP C-terminal domain, human TSP-4 shares 79% aa sequence identity with TSP-3 and COMP, and 58% aa sequence identity with TSP-1 and -2. The coiled-coil motif mediates pentamer formation with COMP, either homotypically or heterotypically (3-6). TSP-4 binds a variety of matrix proteins including collagens I, II, III, V, laminin-1, fibronectin, and matrilin-2 (4). Interactions of TSP-4 with non-collagenous proteins are independent of divalent cations, while interactions with collagenous proteins are enhanced in the presence of zinc (4). TSP-4 is expressed in heart, skeletal muscle, vascular smooth muscle, and vascular endothelial cells (7-9). It accumulates at neuromuscular junctions and synapse-rich regions and is upregulated in muscle by experimental denervation (8). TSP-4 mediates the adhesion of motor and sensory neurons and promotes neurite outgrowth (8). A polymorphism of TSP-4 (A387P) is associated with early coronary artery disease (10-12). Unlike wild type TSP-4, the A387P variant does not support HUVEC attachment and spreading (9). Integrin alpha M/ beta 2 enables activated neutrophil adhesion to both the variant A387P and wild type TSP-4, although the A387P variant induces a greater release of proinflammatory molecules (13).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 276523
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Thrombospondin-4
Ala22-Asn961 (Pro276Ala, Ala420Val)
Accession # P35443
Ala22-Asn961 (Pro276Ala, Ala420Val)
Accession # P35443
Specificity
Detects human Thrombospondin-4 in direct ELISAs and Western blots. Does not cross-react with recombinant human Thrombospondin-1, -2, or -3.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human Thrombospondin‑4 Antibody
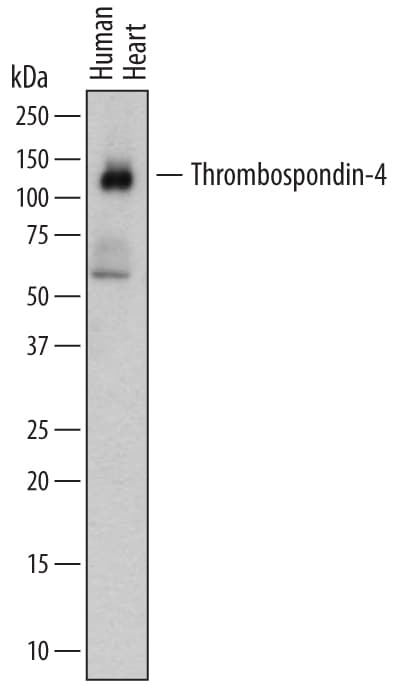
Detection of Human Thrombospondin‑4 by Western Blot.
Western blot shows lysates of human heart tissue. PVDF membrane was probed with 1 µg/mL of Mouse Anti-Human Thrombospondin-4 Monoclonal Antibody (Catalog # MAB2390) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF007). A specific band was detected for Thrombospondin-4 at approximately 130kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Thrombospondin-4 by Immunohistochemistry
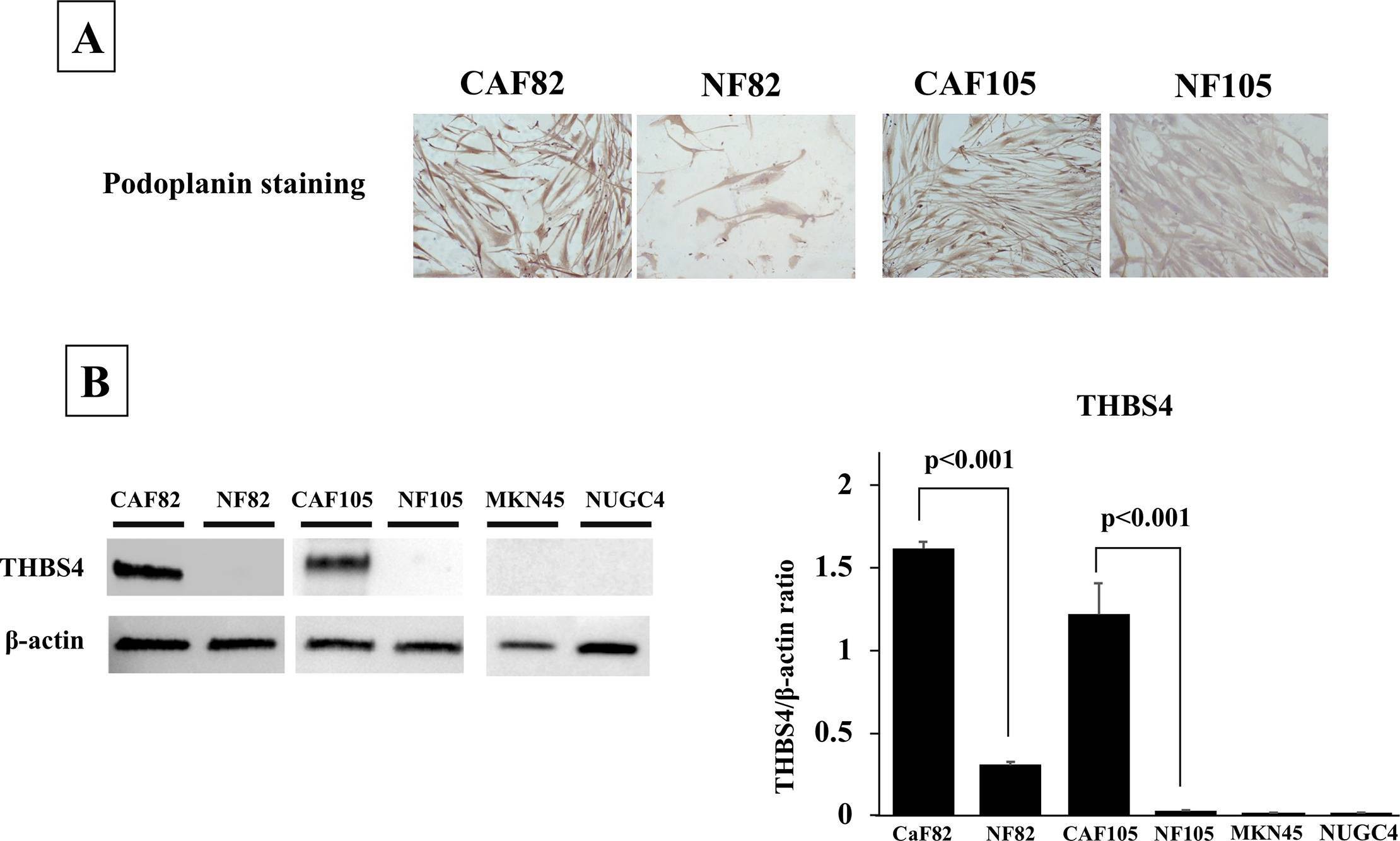
The picture of CAFs and NFs stained with Podoplanin and the western blot analysis of THBS4.(A), Representative picture of Podoplanin staining. Podoplanin was mainly stained at the cytoplasm fibroblasts. The expression level of Podoplanin was higher in CAFs, in compared with that in NFs. (B), THBS4 expression. CAFs expressed THBS4, but NFs and cancer cells did not. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31703077), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Thrombospondin‑4 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Human heart tissue
Sample: Human heart tissue
Reviewed Applications
Read 5 reviews rated 4 using MAB2390 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Thrombospondin-4
References
- Adams, J.C. and J. Lawler (2004) Int. J. Biochem. Cell Biol. 36:961.
- Stenina, O.I. et al. (2004) Int. J. Biochem. Cell Biol. 36:1013.
- Lawler, J. et al. (1995) J. Biol. Chem. 270:2809.
- Narouz-Ott, L. et al. (2000) J. Biol. Chem. 275:37110.
- Hauser, N. et al. (1995) FEBS Lett. 368:307.
- Sodersten, F. et al. (2006) Connect. Tissue Res. 47:85.
- Lawler, J. et al. (1993) J. Cell Biochem. 120:1059.
- Arber, S. and P. Caroni (1995) J. Cell Biol. 131:1083.
- Stenina, O.I. et al. (2003) Circulation 108:1514.
- Topol, E.J. et al. (2001) Circulation 104:2641.
- Wessel, J. et al. (2004) Am. Heart J. 147:905.
- Stenina, O.I. et al. (2005) FASEB J. 19:1893.
- Pluskota, E. et al. (2005) Blood 106:3970.
Alternate Names
THBS4, Thrombospondin4, TSP-4
Gene Symbol
THBS4
UniProt
Additional Thrombospondin-4 Products
Product Documents for Human Thrombospondin‑4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Thrombospondin‑4 Antibody
For research use only
Related Research Areas
Citations for Human Thrombospondin‑4 Antibody
Customer Reviews for Human Thrombospondin‑4 Antibody (5)
4 out of 5
5 Customer Ratings
Have you used Human Thrombospondin‑4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
5 reviews
Showing All
Filter By:
-
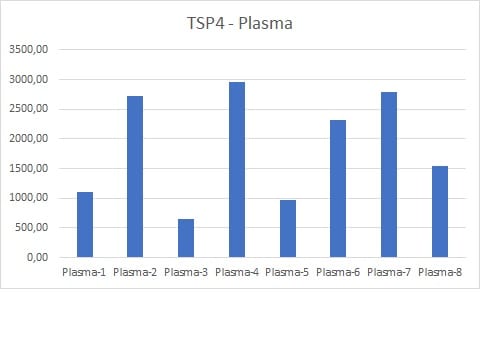
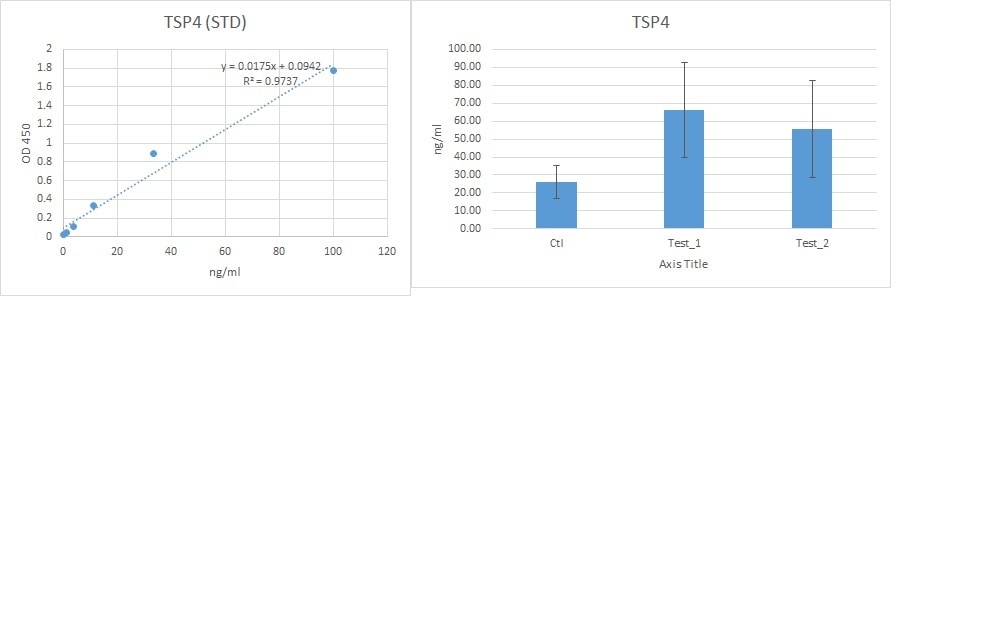
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 07/01/2024
-
Application: MicroarraysSample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 01/03/2020
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 10/04/2019We used this antibody for a sandwich ELISA in combination with pAb (AF2390)) and protein (2390-TH). This combination works very well for detecting the TSP4 in human serum and plasma
-
Application: MicroarraySample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 02/21/2019
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 23942617Species: HumanVerified Customer | Posted 02/19/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways