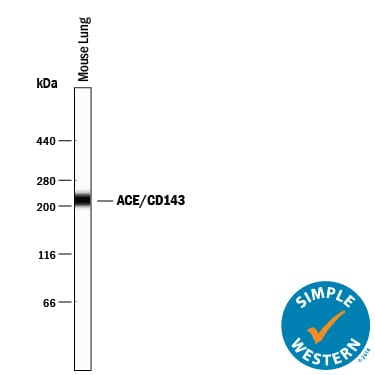
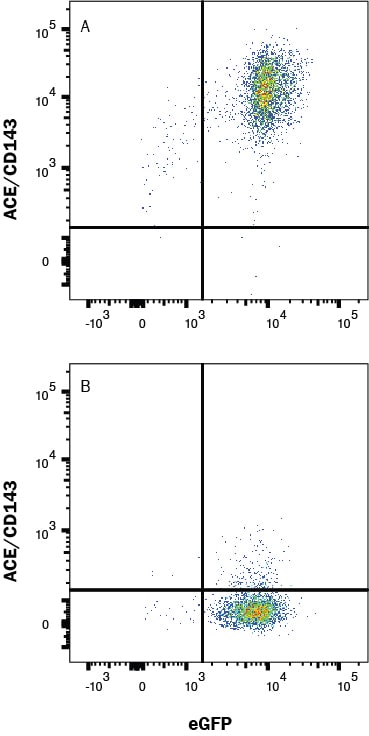
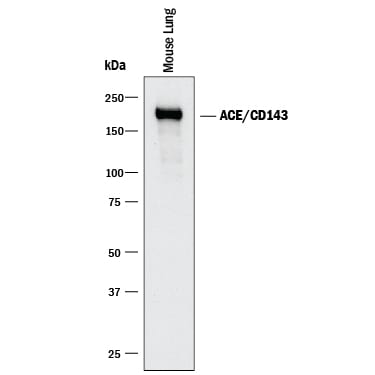
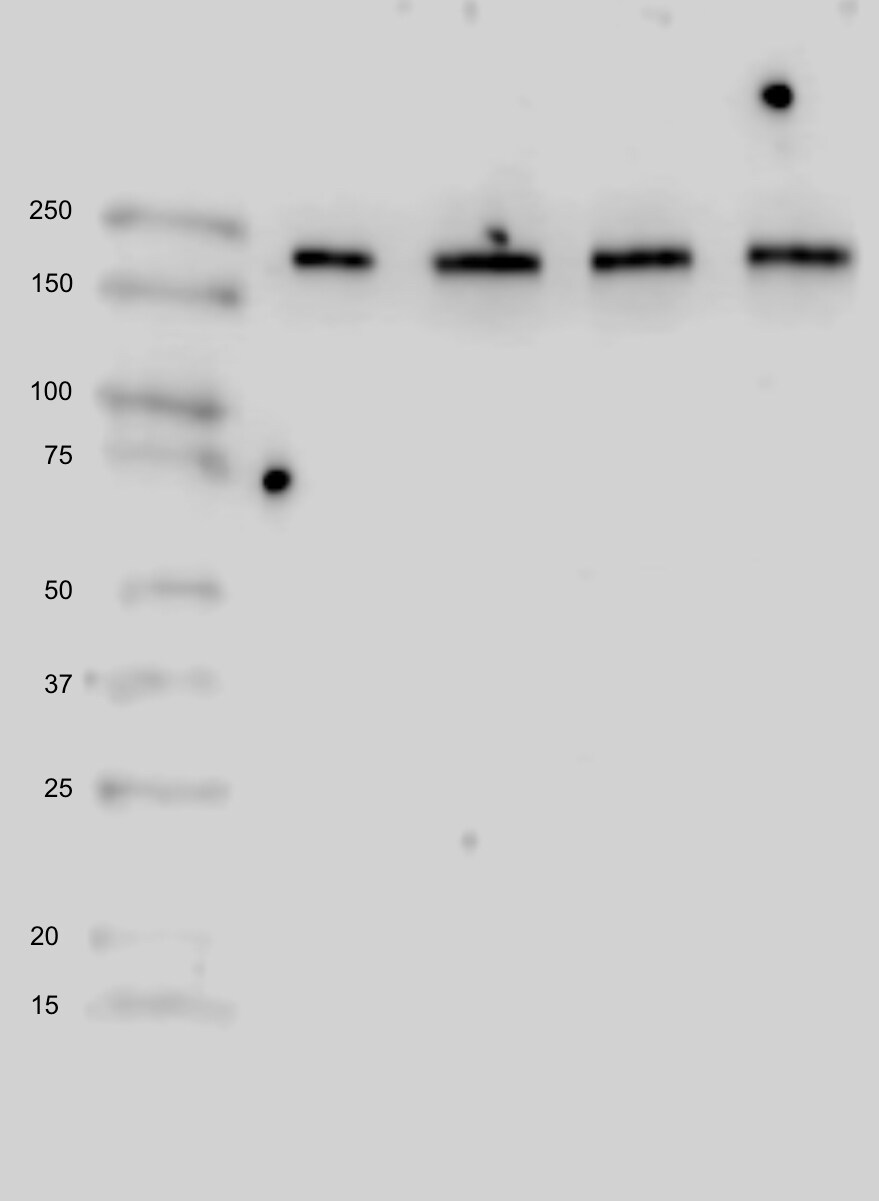
ACE (also known as peptidyl-dipetidase A) is a zinc metallopeptidase important for blood pressure control and water and salt metabolism (1). It cleaves the C-terminal dipeptide from angiotensin I to produce the potent vasopressor octapeptide angiotensin II and inactivates bradykinin by the sequential removal of two C-terminal dipeptides. In addition to the two physiological substrates, ACE cleaves C-terminal dipeptides from various oligopeptides with a free C-terminus. Because of its location and specificity, ACE plays additional roles in immunity, reproduction and neuropeptide regulation. For example, ACE degrades Alzheimer amyloid beta -peptide (A beta ), retards A beta aggregation, deposition, fibril formation, and inhibits cytotoxicity (2).
ACE is a type I membrane protein and exists in two isoforms (1). Somatic ACE, found in endothelial, epithelial and neuronal cells, comprises two highly similar catalytic domains called N- and C-domains. Germinal ACE, found exclusively in the testes, comprises a single catalytic domain identical to the C-domain of somatic ACE except for an N-terminal 67 residue germinal ACE-specific sequence. Physiological functions of the two tissue-specific isozymes are not interchangeable (3). For example, sperm-specific expression of the germinal ACE, not the somatic ACE, in ACE knockout male mice restored fertility.
Soluble ACE is present in many biological fluids, such as serum, seminal fluid, amniotic fluid and cerebrospinal fluid (1). The soluble ACE is derived from the membrane forms by actions of secretases or sheddases. The identities of the secretases have not been revealed, although they belong to the family of zinc metallopeptidases (4, 5).