CXCL13, also known as B-lymphocyte chemoattractant (BLC), is a CXC chemokine that is constitutively expressed in secondary lymphoid organs. Mouse BCA-1 cDNA encodes a precursor protein of 109 amino acid residues with a putative leader sequence of 21 residues. Mature mouse BCA-1 shares 64% amino acid sequence similarity with the human protein and 23‑34% amino acid sequence identity with other known CXC chemokines. Recombinant or chemically synthesized BCA-1 is a potent chemoattractant for B lymphocytes but not T lymphocytes, monocytes or neutrophils. BLR1, a G protein-coupled receptor originally isolated from Burkitt’s lymphoma cells, has now been shown to be the specific receptor for BCA-1. Among cells of the hematopoietic lineages, the expression of BLR1, now designated CXCR5, is restricted to B lymphocytes and a subpopulation of T helper memory cells. Mice lacking BLR1 have been shown to lack inguinal lymph nodes. These mice were also found to have impaired development of Peyer’s patches and defective formation of primary follicles and germinal centers in the spleen as a result of the inability of B lymphocytes to migrate into B cell areas.

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Neutralization
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, Bioassay, In vivo assay, IHC-F, In-vivo
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse CXCL13/BLC/BCA‑1
Ile22-Ala109
Accession # Q3U1E8
Ile22-Ala109
Accession # Q3U1E8
Specificity
Detects mouse CXCL13/BLC/BCA‑1 in direct ELISAs and Western blots. In direct ELISAs, less than 1% cross-reactivity with recombinant human (rh) CXCL13 and rhGCP-2 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse CXCL13/BLC/BCA‑1 Antibody
Chemotaxis Induced by CXCL13/BLC/BCA‑1 and Neutralization by Mouse CXCL13/BLC/BCA‑1 Antibody.
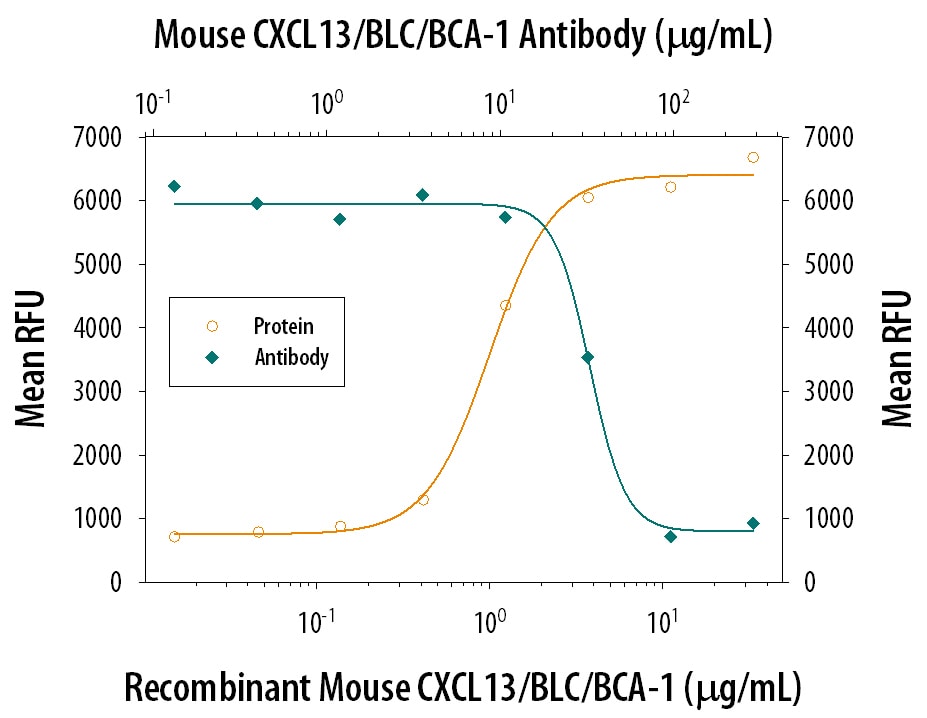
Recombinant Mouse CXCL13/BLC/BCA-1 (Catalog # 470-BC) chemoattracts the BaF3 mouse pro-B cell line transfected with human CXCR5 in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin staining. Chemotaxis elicited by Recombinant Mouse CXCL13/BLC/BCA-1 (4 µg/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Mouse CXCL13/ BLC/BCA-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF470). The ND50 is typically 20-50 µg/mL.CXCL13/BLC/BCA‑1 in Mouse Intestine.
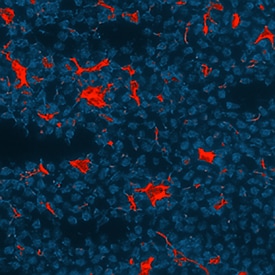
CXCL13/BLC/BCA-1 was detected in perfusion fixed frozen sections of mouse intestine (Peyer's patch) using Goat Anti-Mouse CXCL13/BLC/BCA-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF470) at 15 µg/mL overnight at 4 °C. Tissue was stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to endothelial cells. View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Detection of Mouse CXCL13/BLC/BCA-1 by Immunohistochemistry
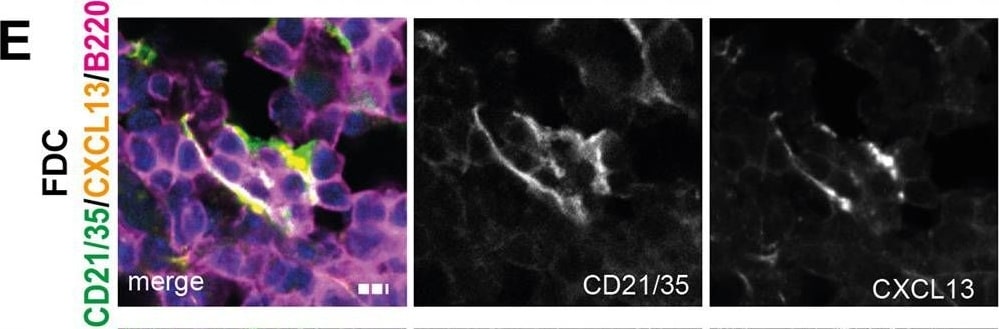
Molecular, cellular and structural composition of periarterial TLS.A–I Immunofluorescence staining and confocal laser scanning microscopy of representative Rbpj delta EC kidney samples, merged and single channels as indicated. “A” indicates artery. Optical Magnification 200x; different scan areas (see scale bars). Scale: solid bar 50 µm, dotted bar 10 µm (2E). Each micrograph is representative of at least 4 biological replicates. J Whole kidney mRNA expression, relative fold change to control gene Rps9, n = 10/group. Graphs: Scatter dot blot, mean, SD (whiskers). Mann–Whitney test, two-tailed, Exact p-values: Cxcl13, p = 0.0003; Cxcl12, p = 0.393; Cxcr5, p = 0.0433; Cxcr4, p = 0.0288; Ccl19, p = 0.0052; Baff, p = 0.0007; Rankl, p = 0.0005. Source data are provided as a Source Data file. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35440634), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Immunohistochemistry
Molecular, cellular and structural composition of periarterial TLS.A–I Immunofluorescence staining and confocal laser scanning microscopy of representative Rbpj delta EC kidney samples, merged and single channels as indicated. “A” indicates artery. Optical Magnification 200x; different scan areas (see scale bars). Scale: solid bar 50 µm, dotted bar 10 µm (2E). Each micrograph is representative of at least 4 biological replicates. J Whole kidney mRNA expression, relative fold change to control gene Rps9, n = 10/group. Graphs: Scatter dot blot, mean, SD (whiskers). Mann–Whitney test, two-tailed, Exact p-values: Cxcl13, p = 0.0003; Cxcl12, p = 0.393; Cxcr5, p = 0.0433; Cxcr4, p = 0.0288; Ccl19, p = 0.0052; Baff, p = 0.0007; Rankl, p = 0.0005. Source data are provided as a Source Data file. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35440634), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Immunocytochemistry/ Immunofluorescence
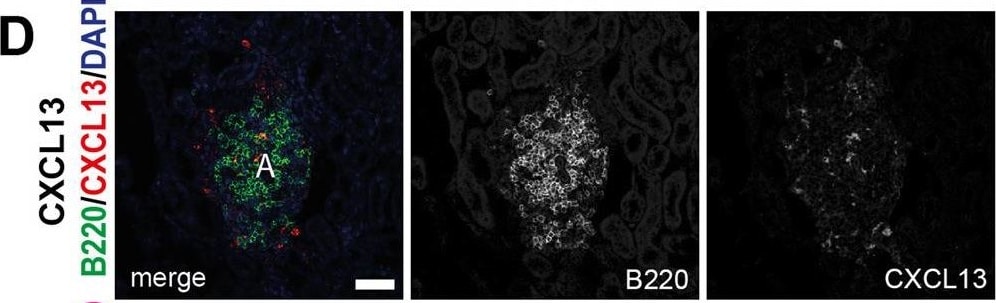
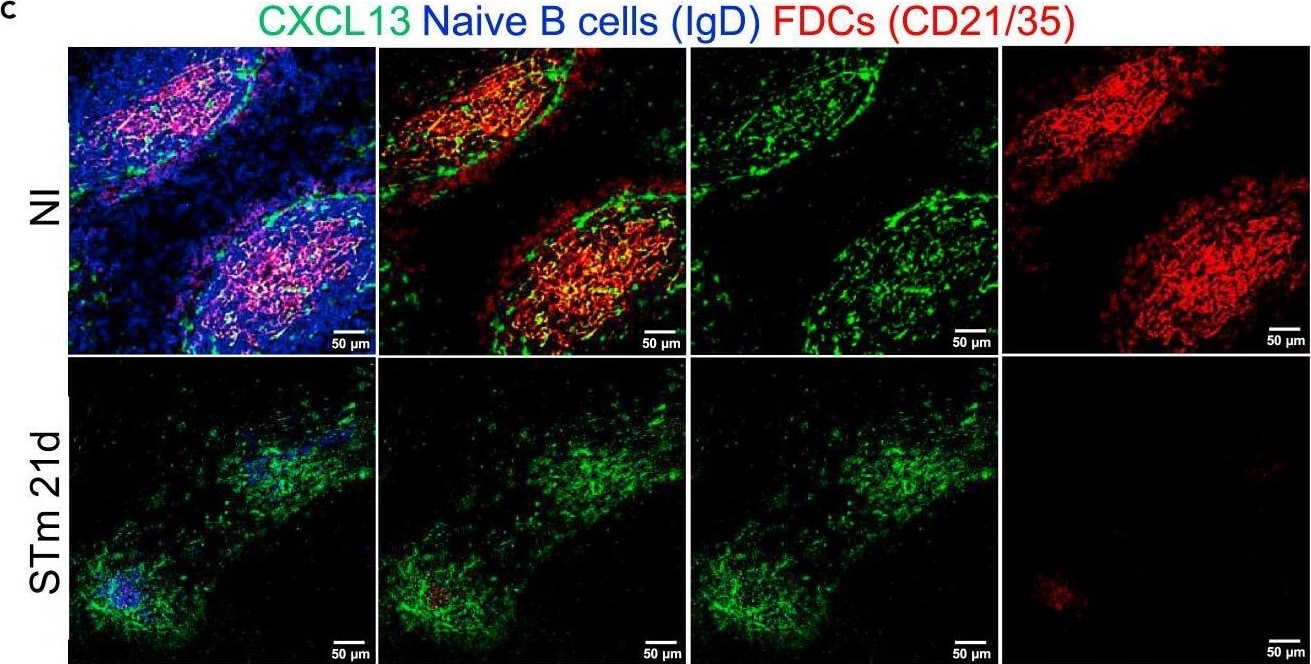
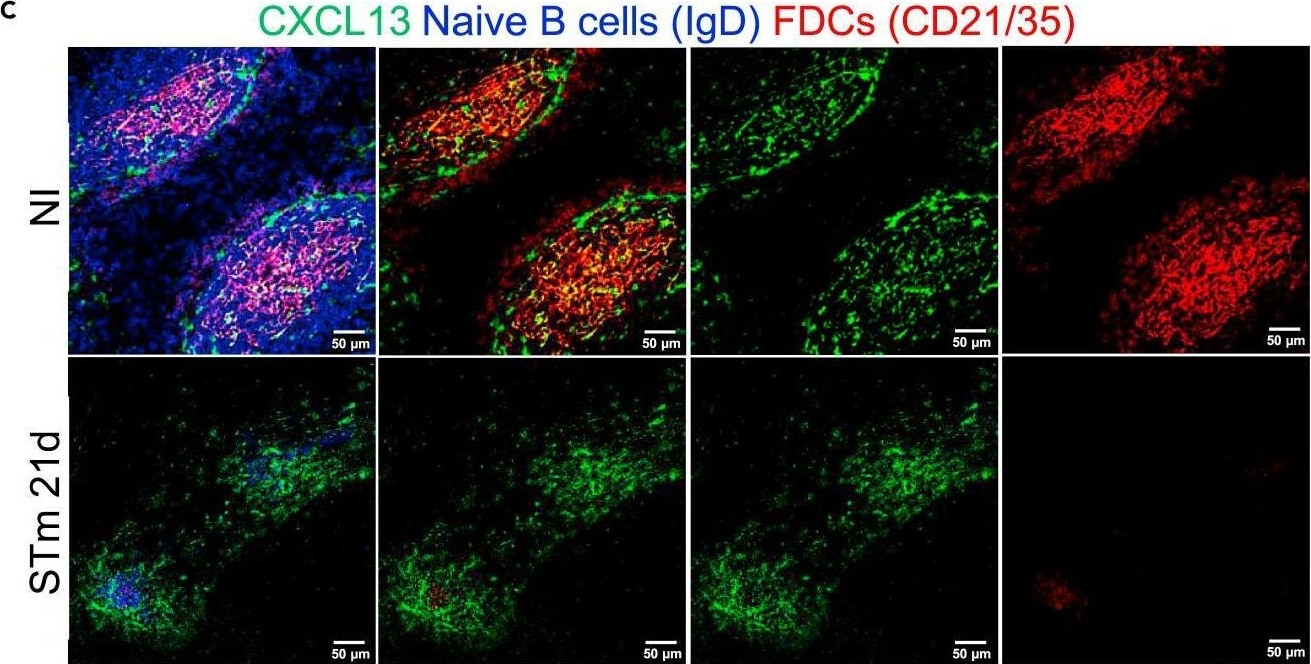
CXCL13 expression in the follicles is maintained after infection with STmMice were infected as per Figure 1.(A) Representative IF images show CCL21 (green) expression along with B220 (blue) and ER-TR-7 (red) staining in NI mice (top row) and mice infected for 21 days (bottom row). Left-hand panels show merged images, and single-colour panels are displayed in the right-hand panels.(B) Gene expression of Ccl21 in WP of NI mice and mice infected with STm for 21 days.(C) Representative IF images of spleen sections stained for CXCL13 (green), IgD (blue), and CD21/35 (red) in control mice (top row) and STm-infected mice for 21 days (bottom row). Merged three-color images (left), CXCL13 and CD21/35 merged images (second column), and single-color channels (two columns to the right) are displayed. Scale bar 50 μm.(D) Graph represents gene expression of Cxcl13 in WP from NI mice and mice infected with STm for 21 days. Each point in the graphs represents the gene expression detected in WP from one individual mouse, the bar height represents the median, and the error bars display the 95% CI. Two-tailed unpaired, t-test was used to compare groups. ∗∗p< 0.01, ns, nonsignificant.(E) Spleen sections were stained for CXCL13 (green), IgD (blue), MadCAM-1 (red) and CD3 (white). Representative IF images on top row show NI mice and images from STm-infected mice are shown in the bottom row. Images on the right displayed four-channel merged images, MadCAM-1 and CXCL13 merged images are presented in the second column and double-positive cells are yellow. Single-color images are shown to the left. Scale bar 50 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36950118), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Immunohistochemistry
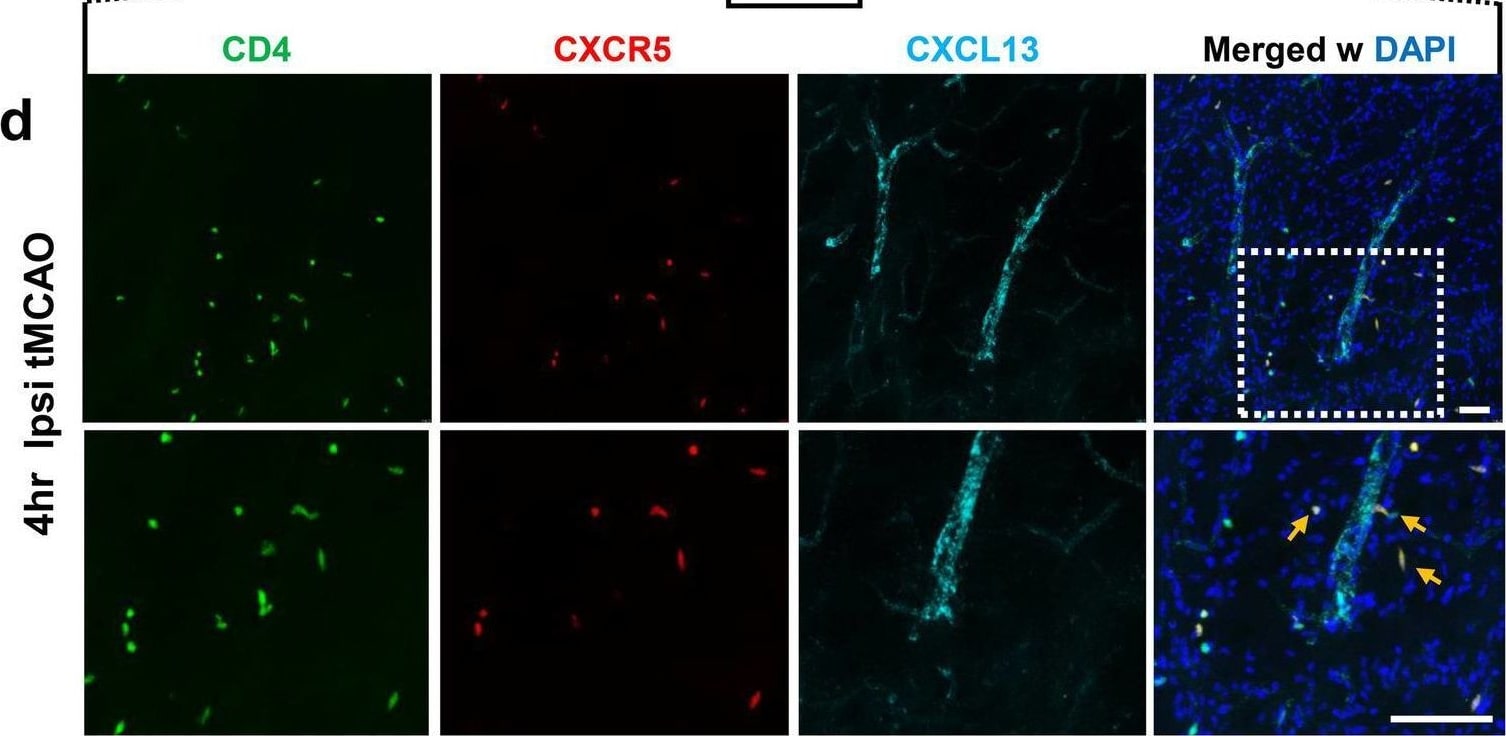
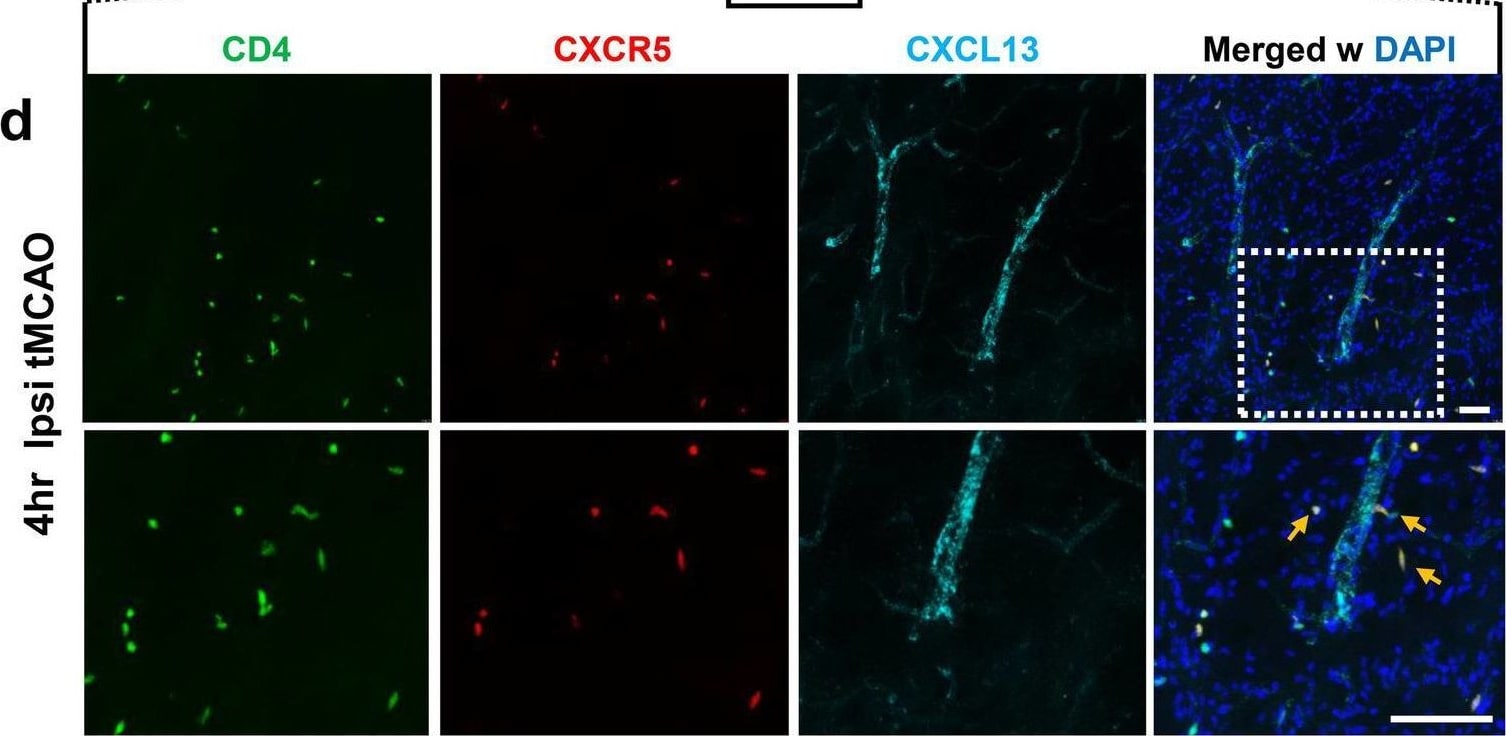
Increased expression of CXCL13 on ICAM-1+ cerebral blood vessels leads to accelerated CD4 + CXCR5 + cell infiltration in the ipsilateral brain hemisphere after tMCAO. a Representative images of immunohistochemistry (IHC) staining of mouse brains with anti-ICAM-1 (green), and anti-CXCL13 (red) antibodies, and merged image (right panel) of cerebral blood vessels in ipsilateral brain hemisphere following SHAM conditions, as well as at 4 h and 24 h post-tMCAO. b Quantification of ICAM-1+ area in ipsilateral brain hemisphere following SHAM conditions, as well as at 4 h and 24 h post-tMCAO (n = 5). c Quantification of percentage area of ICAM-1+ vessels that are also CXCL13+ in ipsilateral brain hemisphere following SHAM conditions, at 4 h post-tMCAO and at 24 h post-MCAO in mice (n = 5). d Representative low (top panels) and high (bottom panels) magnification images of CD4 (green), CXCR5 (red), and CXCL13 (cyan) IHC staining at the peri-infarct area, 4 h post-tMCAO. Bottom panels are a higher magnification image of the dotted box from the top panel to highlight double-positive CD4+ CXCR5+ T cells indicated by orange arrows. Scale bar = 30 μm. e–g Quantification of CD4+ T cells (e), percentage of CD4+ T cells positive for CXCR5 (f), and percentage of CXCR5+ cells positive for CD4 (g) in the contralateral caudate, ipsilateral caudate/thalamus and ipsilateral cortex. Data are combined from three independent experiments. Data represent mean ± s.e.m. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. n.s. = not significant. One-way ANOVA followed by Dunn’s post hoc test (b, c, e, f, g) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35624463), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Flow Cytometry
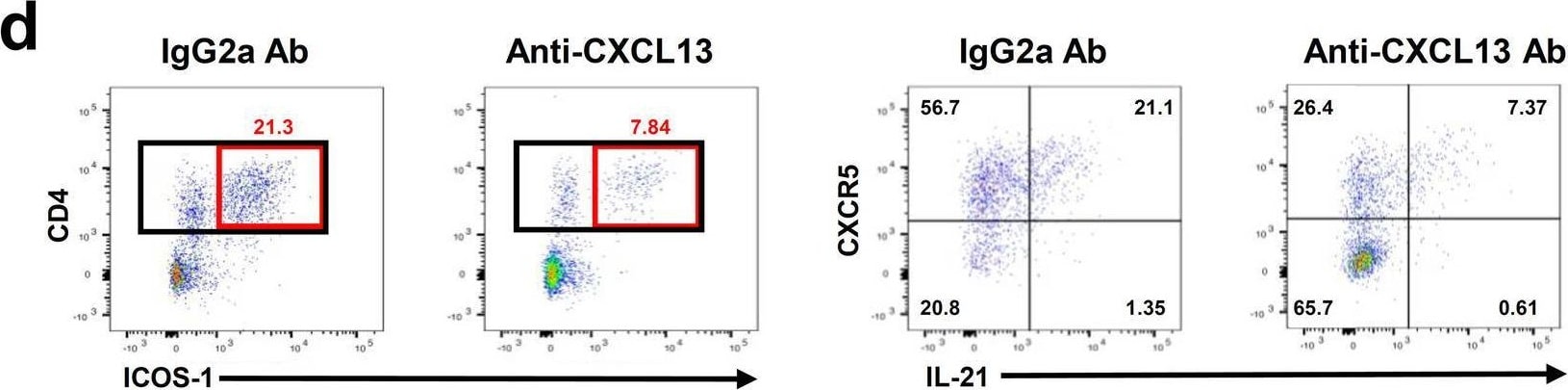
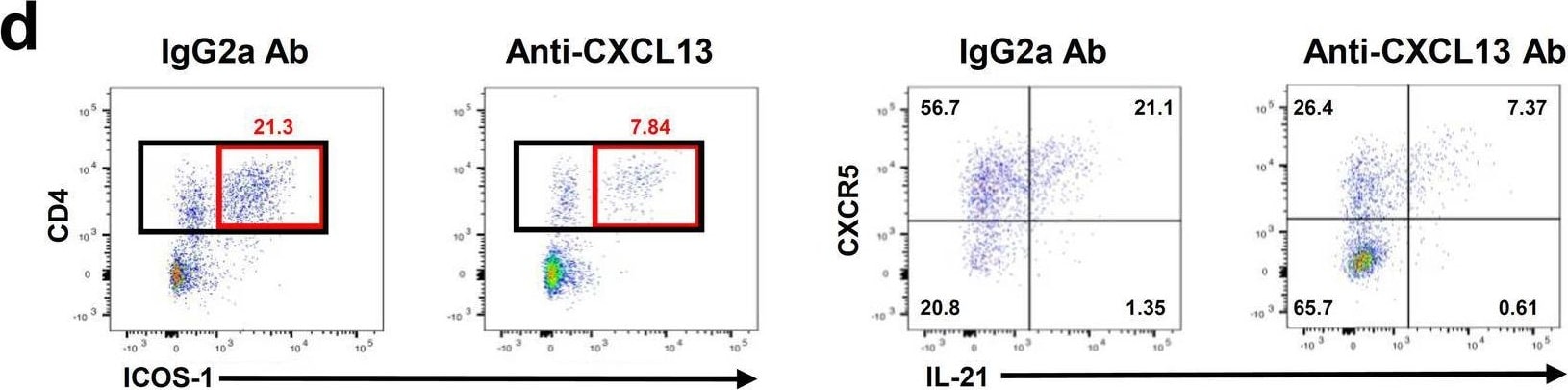
Administration of anti-CXCL13 antibody reduces infarct volume and inhibits the infiltration of TFH cells into the brain following tMCAO. a Experimental design for administration of IgG2a and anti-CXCL13 antibody (100 μg) i.p. b Representative cresyl violet images to assess ischemic brain damage at 24 h tMCAO in IgG2a-treated and anti-CXCL13 antibody (100 μg)-treated mice. c Quantification of infarct volumes (mm3) at 24 h tMCAO in IgG2a-treated and CXCL13 antibody-treated mice (n = 10). d Representative flow cytometry gating of CD4+ ICOS-1+ and subsequent CXCR5+ IL-21+ cells from the ipsilateral brain of IgG2a isotype control-treated or anti-CXCL13 antibody-treated mice at 24 h following tMCAO. e–g Quantification of CD4+ T cells (e), % of CD4 T cells+ for ICOS-1 (f), and % of CD4+ ICOS-1+ T cells+ for CXCR5+ and IL-21+ (g) from IgG2a isotype control-treated and anti-CXCL13 antibody-treated mice at 24 h tMCAO. (n = 6) Data are combined from three independent experiments. Data represent mean ± s.e.m. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. Student’s t test with Mann–Whitney U test (c, e–g) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35624463), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Immunocytochemistry/ Immunofluorescence
CXCL13 expression in the follicles is maintained after infection with STmMice were infected as per Figure 1.(A) Representative IF images show CCL21 (green) expression along with B220 (blue) and ER-TR-7 (red) staining in NI mice (top row) and mice infected for 21 days (bottom row). Left-hand panels show merged images, and single-colour panels are displayed in the right-hand panels.(B) Gene expression of Ccl21 in WP of NI mice and mice infected with STm for 21 days.(C) Representative IF images of spleen sections stained for CXCL13 (green), IgD (blue), and CD21/35 (red) in control mice (top row) and STm-infected mice for 21 days (bottom row). Merged three-color images (left), CXCL13 and CD21/35 merged images (second column), and single-color channels (two columns to the right) are displayed. Scale bar 50 μm.(D) Graph represents gene expression of Cxcl13 in WP from NI mice and mice infected with STm for 21 days. Each point in the graphs represents the gene expression detected in WP from one individual mouse, the bar height represents the median, and the error bars display the 95% CI. Two-tailed unpaired, t-test was used to compare groups. ∗∗p< 0.01, ns, nonsignificant.(E) Spleen sections were stained for CXCL13 (green), IgD (blue), MadCAM-1 (red) and CD3 (white). Representative IF images on top row show NI mice and images from STm-infected mice are shown in the bottom row. Images on the right displayed four-channel merged images, MadCAM-1 and CXCL13 merged images are presented in the second column and double-positive cells are yellow. Single-color images are shown to the left. Scale bar 50 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36950118), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Immunohistochemistry
Increased expression of CXCL13 on ICAM-1+ cerebral blood vessels leads to accelerated CD4 + CXCR5 + cell infiltration in the ipsilateral brain hemisphere after tMCAO. a Representative images of immunohistochemistry (IHC) staining of mouse brains with anti-ICAM-1 (green), and anti-CXCL13 (red) antibodies, and merged image (right panel) of cerebral blood vessels in ipsilateral brain hemisphere following SHAM conditions, as well as at 4 h and 24 h post-tMCAO. b Quantification of ICAM-1+ area in ipsilateral brain hemisphere following SHAM conditions, as well as at 4 h and 24 h post-tMCAO (n = 5). c Quantification of percentage area of ICAM-1+ vessels that are also CXCL13+ in ipsilateral brain hemisphere following SHAM conditions, at 4 h post-tMCAO and at 24 h post-MCAO in mice (n = 5). d Representative low (top panels) and high (bottom panels) magnification images of CD4 (green), CXCR5 (red), and CXCL13 (cyan) IHC staining at the peri-infarct area, 4 h post-tMCAO. Bottom panels are a higher magnification image of the dotted box from the top panel to highlight double-positive CD4+ CXCR5+ T cells indicated by orange arrows. Scale bar = 30 μm. e–g Quantification of CD4+ T cells (e), percentage of CD4+ T cells positive for CXCR5 (f), and percentage of CXCR5+ cells positive for CD4 (g) in the contralateral caudate, ipsilateral caudate/thalamus and ipsilateral cortex. Data are combined from three independent experiments. Data represent mean ± s.e.m. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. n.s. = not significant. One-way ANOVA followed by Dunn’s post hoc test (b, c, e, f, g) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35624463), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL13/BLC/BCA-1 by Flow Cytometry
Administration of anti-CXCL13 antibody reduces infarct volume and inhibits the infiltration of TFH cells into the brain following tMCAO. a Experimental design for administration of IgG2a and anti-CXCL13 antibody (100 μg) i.p. b Representative cresyl violet images to assess ischemic brain damage at 24 h tMCAO in IgG2a-treated and anti-CXCL13 antibody (100 μg)-treated mice. c Quantification of infarct volumes (mm3) at 24 h tMCAO in IgG2a-treated and CXCL13 antibody-treated mice (n = 10). d Representative flow cytometry gating of CD4+ ICOS-1+ and subsequent CXCR5+ IL-21+ cells from the ipsilateral brain of IgG2a isotype control-treated or anti-CXCL13 antibody-treated mice at 24 h following tMCAO. e–g Quantification of CD4+ T cells (e), % of CD4 T cells+ for ICOS-1 (f), and % of CD4+ ICOS-1+ T cells+ for CXCR5+ and IL-21+ (g) from IgG2a isotype control-treated and anti-CXCL13 antibody-treated mice at 24 h tMCAO. (n = 6) Data are combined from three independent experiments. Data represent mean ± s.e.m. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. Student’s t test with Mann–Whitney U test (c, e–g) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35624463), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse CXCL13/BLC/BCA‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse intestine (Peyer's patch)
Sample: Perfusion fixed frozen sections of mouse intestine (Peyer's patch)
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse CXCL13/BLC/BCA‑1 (Catalog # 470-BC)
Sample: Recombinant Mouse CXCL13/BLC/BCA‑1 (Catalog # 470-BC)
Neutralization
Measured by its ability to neutralize CXCL13/BLC/BCA‑1-induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human CXCR5. The Neutralization Dose (ND50) is typically 20-50 µg/mL in the presence of 4 µg/mL Recombinant Mouse CXCL13/BLC/BCA‑1.
Reviewed Applications
Read 2 reviews rated 4 using AF470 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CXCL13/BLC/BCA-1
References
- Gunn, M.D. et al. (1998) Nature, 391:799.
- Legler, D.F. et al. (1998) J. Exp. Med. 187:655.
- Forster, R. et al. (1996) Cell 87:1037.
Alternate Names
ANGIE2, BCA-1, BCA1, BLC, BLR1L, SCYB13
Gene Symbol
CXCL13
UniProt
Additional CXCL13/BLC/BCA-1 Products
Product Documents for Mouse CXCL13/BLC/BCA‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse CXCL13/BLC/BCA‑1 Antibody
For research use only
Citations for Mouse CXCL13/BLC/BCA‑1 Antibody
Customer Reviews for Mouse CXCL13/BLC/BCA‑1 Antibody (2)
4 out of 5
2 Customer Ratings
Have you used Mouse CXCL13/BLC/BCA‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: Immunohistochemistry-FrozenSample Tested: See PMID 20643338Species: MouseVerified Customer | Posted 01/08/2015
-
Application: ImmunofluorescenceSample Tested: See PMID 21593383Species: MouseVerified Customer | Posted 01/08/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways