GITR (glucocorticoid-induced tumor necrosis factor receptor; also named AITR) is a member of the co‑stimulatory subset of the TNF receptor superfamily (1, 2). In mouse, the GITR gene is composed of five exons and encodes multiple length isoforms that arise from alternate splicing. The “standard”, or first reported isoform is a type I transmembrane protein, 228 amino acids (aa) in length that contains a 19 aa signal sequence, a 134 aa extracellular region, a 21 aa transmembrane segment, and a 54 aa cytoplasmic domain. The extracellular region contains four potential N-linked glycosylation sites plus three cysteine-rich pseudorepeats of about 40 aa each (3, 4). The extracellular regions of mouse and human are 57% aa identical. The cytoplasmic domain has a P-x-Q/E-E motif that is known to associate with TRAF2. This is a common characteristic of TNFRSF members with co‑stimulatory functions (4). Three other mouse GITR isoforms (B, C and D) have been reported (5). All share the same N-terminal 101 of 134 aa in the extracellular region (including pseudorepeats #1, #2 and one-half of #3). Isoform D diverges at aa #101 and continues for another 12 aa for a total length of 113 aa. This is a naturally-occurring soluble form. Isoforms B and C show splicing in their cytoplasmic tails that creates cytoplasmic domains of 118 aa and 46 aa, respectively. In both the B and C isoforms, the TRAF2 binding site is spliced out, with a p56lck binding site inserted in isoform B (4). Given its membership in the TNFRSF, it likely functions as a trimer on the cell surface (2). GITR is predominantly expressed on CD4+CD25+ regulatory T cells (Treg) and naïve CD8+ and CD4+ CD25- T cells, where its expression is up-regulated after antigen-driven activation. GITR activation provides co‑stimulatory signals for activated CD4+ CD25- T cells to enhance cell proliferation and augment cytokine production (IL-2, IL-4, IFN-gamma ). On CD4+ CD25+ Treg cells, GITR activation provides co‑stimulatory signals to induce proliferation, setting Treg cells in an active/hyperproliferactive state (6‑8).
Mouse GITR/TNFRSF18 Antibody
R&D Systems | Catalog # MAB52412

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Accession # O35714
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse GITR/TNFRSF18 Antibody
Detection of GITR/TNFRSF18 in HEK293 Human Cell Line Transfected with Mouse GITR/TNFRSF18 and eGFP by Flow Cytometry.
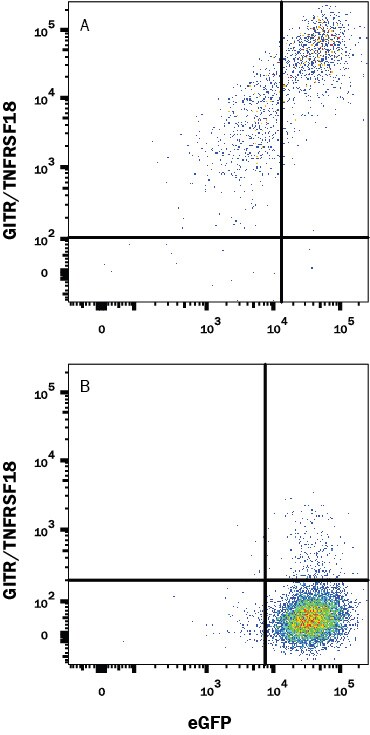
HEK293 human embryonic kidney cell line transfected with (A) mouse GITR/TNFRSF18 or (B) irrelevant transfectants and eGFP was stained with Rabbit Anti-Mouse GITR/TNFRSF18 Monoclonal Antibody (Catalog # MAB52412) followed by APC-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # F0111). Quadrant markers were set based on control antibody staining (Catalog # MAB1050). View our protocol for Staining Membrane-associated Proteins.GITR/TNFRSF18 in Mouse Splenocytes and NIH-3T3 Cell Line.
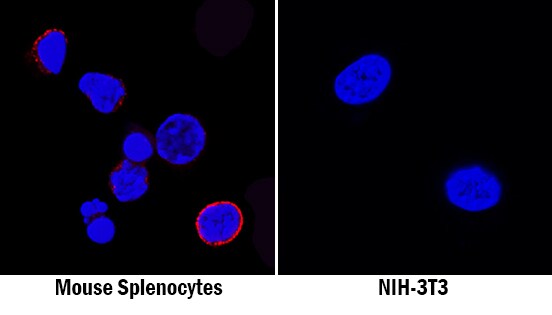
GITR/TNFRSF18 was detected in immersion fixed mouse splenocytes (left panel; positive staining) and NIH-3T3 mouse embryonic fibroblast cell line (right panel; negative staining) using Rabbit Anti-Mouse GITR/TNFRSF18 Monoclonal Antibody (Catalog # MAB52412) at 8 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rabbit IgG Secondary Antibody (red; Catalog # NL004) and counterstained with DAPI (blue). Specific staining was localized to cell surfaces. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.GITR/TNFRSF18 in Mouse Spleen.
GITR/TNFRSF18 was detected in immersion fixed frozen sections of mouse spleen using Rabbit Anti-Mouse GITR/TNFRSF18 Monoclonal Antibody (Catalog # MAB52412) at 3 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Rabbit IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC003). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to lymphocytes. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Applications for Mouse GITR/TNFRSF18 Antibody
CyTOF-ready
Flow Cytometry
Sample: HEK293 Human Cell Line Transfected with Mouse GITR/TNFRSF18 and eGFP
Immunocytochemistry
Sample: Immersion fixed mouse splenocytes and NIH-3T3 mouse embryonic fibroblast cell line
Immunohistochemistry
Sample: Immersion fixed frozen sections of mouse spleen
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: GITR/TNFRSF18
References
- Kwon, B. et al. (2003) Exp. Mol. Med. 35:8.
- Croft, M. (2003) Nat. Rev. Immunol. 3:609.
- Nocentini, G. et al. (1997) Proc. Natl. Acad. Sci. USA 94:6216.
- Nocentini, G. et al. (2000) DNA Cell Biol. 19:205.
- Nocentini, G. et al. (2000) Cell Death Differ. 7:408.
- Tone, M. et al. (2003) Proc. Natl. Acad. Sci. USA 100:15059.
- Ji, H. et al. (2004) J. Immunol. 172:5823.
- Stephens, G.L. et al. (2004) 173:5008.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional GITR/TNFRSF18 Products
Product Documents for Mouse GITR/TNFRSF18 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse GITR/TNFRSF18 Antibody
For research use only
Customer Reviews for Mouse GITR/TNFRSF18 Antibody
There are currently no reviews for this product. Be the first to review Mouse GITR/TNFRSF18 Antibody and earn rewards!
Have you used Mouse GITR/TNFRSF18 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways