Podoplanin, also known as T1 alpha (T1 alpha ), is a mucin type transmembrane glycoprotein with extensive O-glycosylation. It is specifically expressed by lymphatic endothelial cells but not blood vascular endothelial cells. In addition, non-endothelial cells in numerous normal tissues also express the protein. Within the region used as the immunogen, mouse Podoplanin shares 73.5% and 29% amino acid sequence homology with rat and human Podoplanin, respectively.

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Canine, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunofluorescence, Immunocytochemistry, IF/IHC
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Podoplanin
Gln21-Lys133
Accession # Q546R8
Gln21-Lys133
Accession # Q546R8
Specificity
Detects mouse Podoplanin in direct ELISAs and Western blots. In direct ELISAs, approximately 5% cross-reactivity with recombinant human Podoplanin is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse Podoplanin Antibody
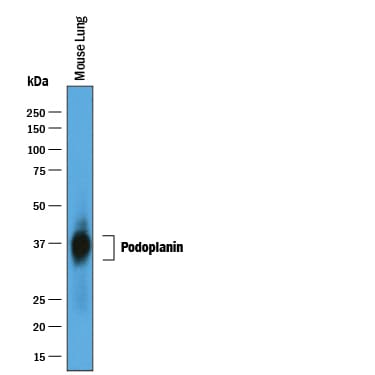
Detection of Mouse Podoplanin by Western Blot.
Western blot shows lysates of mouse lung tissue. PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Mouse Podoplanin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3244) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for Podoplanin at approximately 35-42 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Podoplanin in Mouse Kidney.
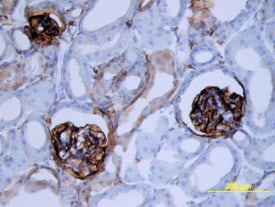
Podoplanin was detected in perfusion fixed frozen sections of mouse kidney using Goat Anti-Mouse Podoplanin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3244) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of Mouse Podoplanin by Simple WesternTM.
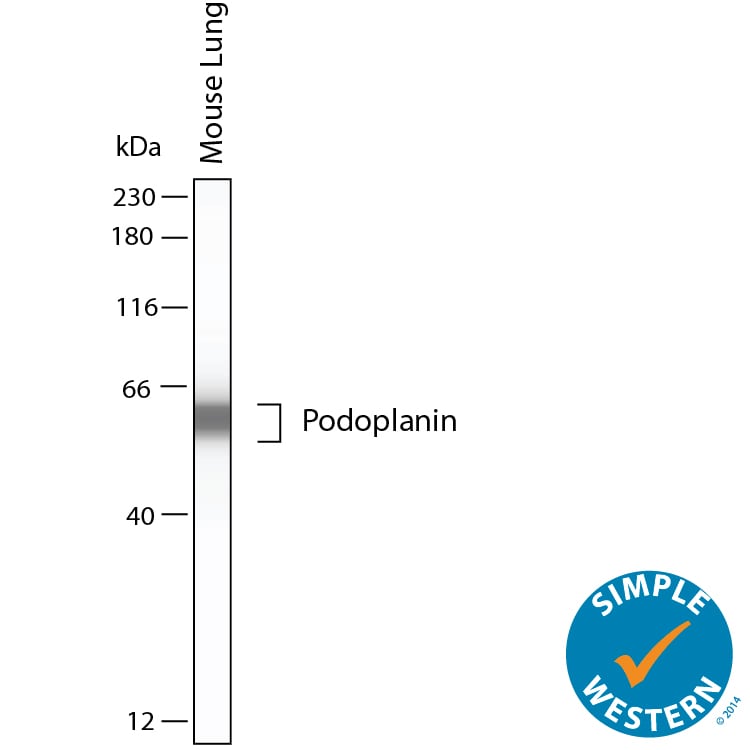
Simple Western shows lysates of mouse lung, loaded at 0.5 mg/ml. A specific band was detected for Podoplanin at approximately 60 kDa (as indicated) using 10 µg/mL of Goat Anti-Mouse Podoplanin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3244). This experiment was conducted under reducing conditions and using the 12-230kDa separation system.Detection of Mouse Podoplanin by Immunocytochemistry/Immunofluorescence
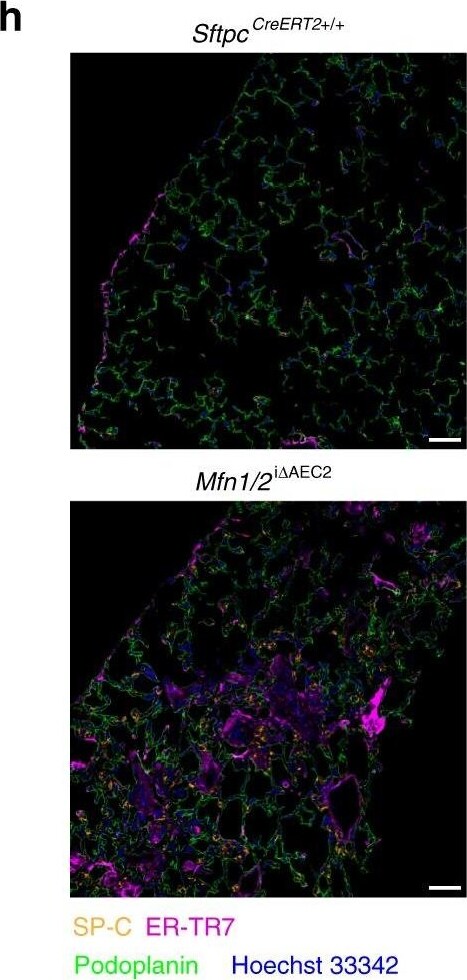
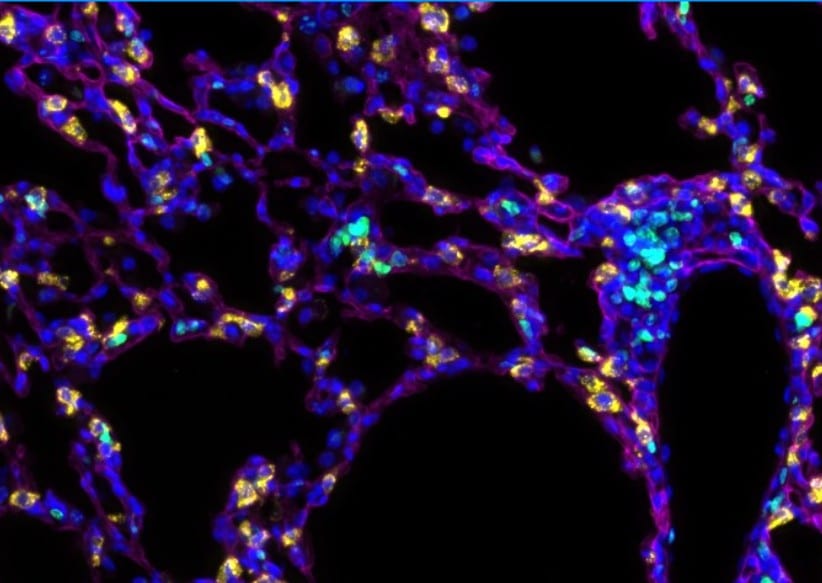
Mfn1/2i delta AEC2 mice develop spontaneous lung fibrosis. a Schema demonstrating the generation of mice with AEC2 cell-specific tamoxifen-inducible deletion of Mfn1/2. SftpcCreERT2+/+ or SftpcCreERT2+/− mice were used as controls. b Genotyping of CD45(-)EpCAM(+) cells isolated from Mfn1/2i delta AEC2 mice (n = 3 mice; lane 1 to lane 3 serves as the positive control). c Representative immunoblots of AEC2 cell lysates obtained 6 weeks after tamoxifen-induced deletion, showing decreased protein levels of both MFN1 and MFN2 in the Mfn1/2−/− AEC2 cells (n = 3 mice per group). d Representative TEM images (upper row, ×12,000; lower row, ×50,000) show mitochondrial ultrastructural changes in SftpcCreERT2+/− and Mfn1/2−/− AEC2 cells (n = 3 mice per group) with disrupted cristae marked with white arrowheads (scale bar, upper row 2 μm, lower row 500 nm. e Kaplan–Meier survival curves of Mfn1/2i delta AEC2 (n = 22) and SftpcCreERT2+/+ (n = 23) mice (p < 0.01 by log-rank test). f Representative Masson’s trichrome-stained lung sections (upper panel, ×100 magnification; lower panel, ×200 magnification) 17 weeks post tamoxifen-induced deletion (SftpcCreERT2+/+ mice n = 6; Mfn1/2i delta AEC2 mice n = 11; scale bar, upper panel 4 mm, lower panel 200 μm). g Representative IHC staining of vimentin, alpha-smooth muscle actin ( alpha -SMA), and collagen III (Col-III) (×200 magnification; n = 3 mice per group; scale bar 200 μm). h Representative immunofluorescent staining of 5x5 tiled confocal images (using ×40 objective) of frozen murine lung sections stained for podoplanin (green), surfactant protein-C (SP-C) (yellow), ER-TR7 (magenta), and Hoechst 33342 stain (blue) (n = 3 mice per group; scale bar 50 μm). i Representative immunofluorescence staining confocal images of podoplanin (green), SP-C (yellow), ER-TR7 (magenta) and Hoechst 33342 nuclear stain (blue) using lung sections of SftpcCreERT2+/−, Mfn1i delta AEC2 and Mfn2i delta AEC2 mice (n = 2 mice per group; scale bar 20 μm). Source data (c, e) are provided as a Source Data file Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31358769), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Podoplanin by Western Blot
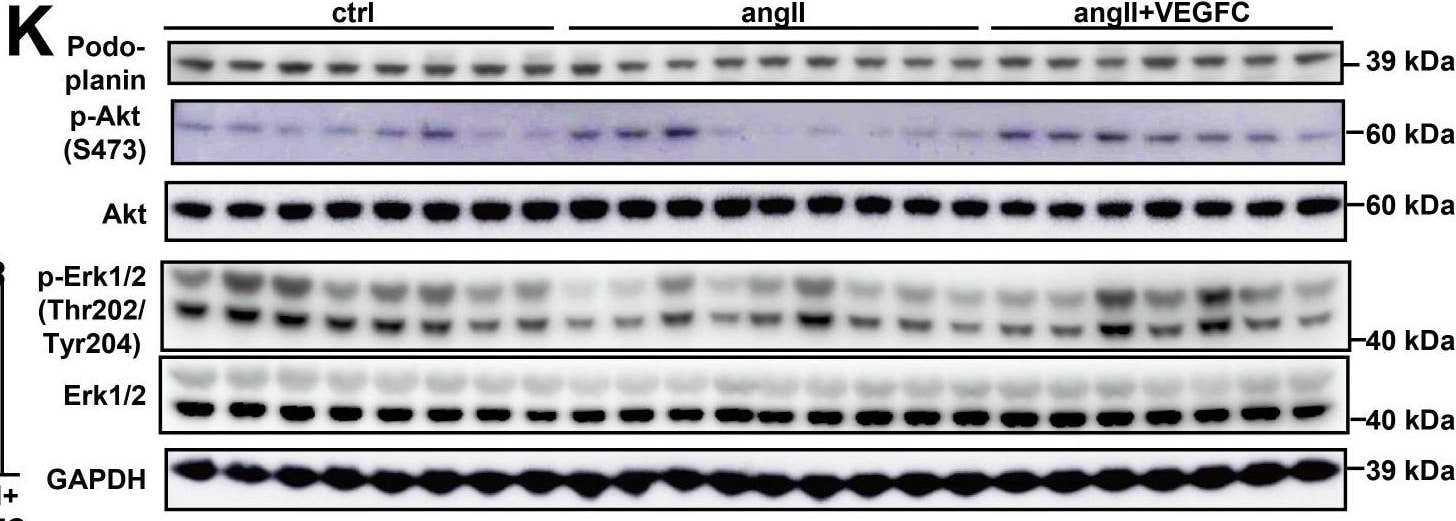
Effect of VEGFCC156S on angiotensin II-induced cardiac dysfunction, cardiac lymphatics, and skin lymphatics.(A) Pharmacokinetic analysis of VEGFCC156S in mice (n = 2 per dose group). (B) Plasma VEGFC concentration measured by ELISA at euthanasia (n = 10–17/group). (C–S) Mice were infused with saline + BSA, angiotensin II + BSA, or angiotensin II + VEGFCC156S as described in Figure 2A. (C) Representative M-mode echocardiography images for each group. (D–G) Echocardiography parameters cardiac output (D), stroke volume (E), Left Ventricular Posterior Wall thickness at diastole (LVPWd) (F) and heart rate (G) are shown (n = 15–24/group). (H–J) Hearts were arrested in diastole, fixed, sectioned, and stained with wheat germ agglutinin (WGA). (H) Representative images of WGA stain for each group. Scale bar, 100 μm. (I) Cardiomyocyte size was assessed (n = 7–9/group). (J) Analysis of cell size variance coefficient from WGA-stained mouse heart sections (n = 21–27/group; a linear mixed model was used for statistics). (K–N) Protein lysates were prepared from mouse hearts and immunoblotted for podoplanin, p-Akt, Akt, p-Erk1/2, Erk1/2, and GAPDH. (K) Representative immunoblots. (L–N) Densitometric quantification of podoplanin (L), p-Akt to Akt ratio (M) and p-Erk to Erk ratio (N) (n = 7–9/group). (O) Representative images of the whole mount stain of lyve1-positive lymphatic vessels in ear skin for each group. Scale bar, 100 μm. (P) Quantification of lyve1-positive lymphatic vessel diameter in ear skin (n = 737–880 vessel/group; a linear mixed model was used for statistics). (Q) Ear skin samples were fixed, sectioned, and stained for lyve1(green) and DAPI (blue). Representative images of lyve1-positive lymphatic vessels in ear skin cross-section for each group. Scale bar, 200 μm. (R) Quantification of lyve1-positive lymphatic vessel density (normalized to total area) in ear skin (n = 6–8 animals/group, n = 6–8 sections/animal; a linear mixed model was used for statistics). Ctrl, control. AngII, angiotensin II. VEGFC, VEGFCc156s. Data are mean ± s.d. One-way ANOVA with Bonferroni posthoc was used for statistics for all figures except 2J, 2P and 2R (a linear mixed model was used for statistics). *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001, n.s. not significant. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/33200983), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Mouse Podoplanin Antibody by Immunohistochemistry-Paraffin
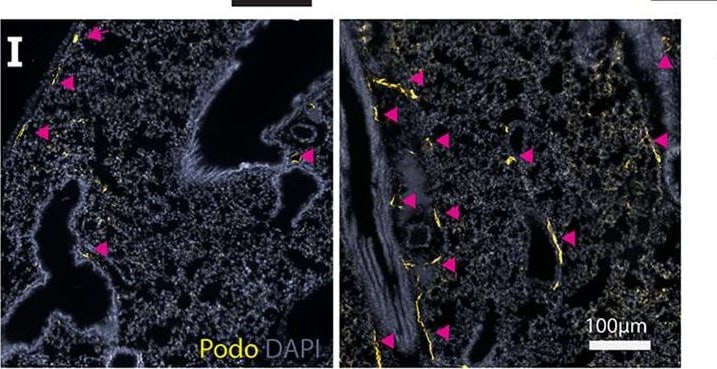
Carboplatin induces lymphangiogenesis in healthy tissues (A) Schematic of rat mesentery culture model. (B) Vehicle-treated lymphatic vessels from mesentery cultures stained with LYVE-1 (grey). (i) High magnification image of boxed area in (B). (C) Carboplatin-treated lymphatic vessels from mesentery cultures stained with LYVE-1 (grey). (ii) High magnification image of boxed area in (C) Scale bar=100µm. (D) Number of sprouts per lymphatic vessel area (n=3/group). (E) Lymphatic vessel density (podoplanin+ vessels per mm2 stroma) in whole mammary fat pads of healthy mice treated with systemic carboplatin (8 mg/kg/dose) or vehicle by IV(n=3-4/group). (F) Lymphatic vessel density measured in mammary fat pads of healthy mice 2 months after treatment with 3 doses of carboplatin or vehicle, (n=3/group). (G) Lymph nodes from healthy, tumor-naïve mice treated with vehicle and stained H&E. (H) LEC number in vehicle-treated and carboplatin-treated lymph nodes in vivo (n=6/group). (I) Representative images of lungs from mice treated with 3 doses of vehicle (left) and carboplatin (right). Podoplanin+ lymphatic vessels noted by arrowheads. (J) Lymphatic vessel density in stromal tissue of lungs of mice pre-treated with carboplatin (n=3/group). *p < 0.05, **p < 0.01, ****p < 0.001. Each data point represents one mouse. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35372032), licensed under a CC-BY license. Not internally tested by R&D Systems.Podoplanin in Mouse Small Intestine and Lung.
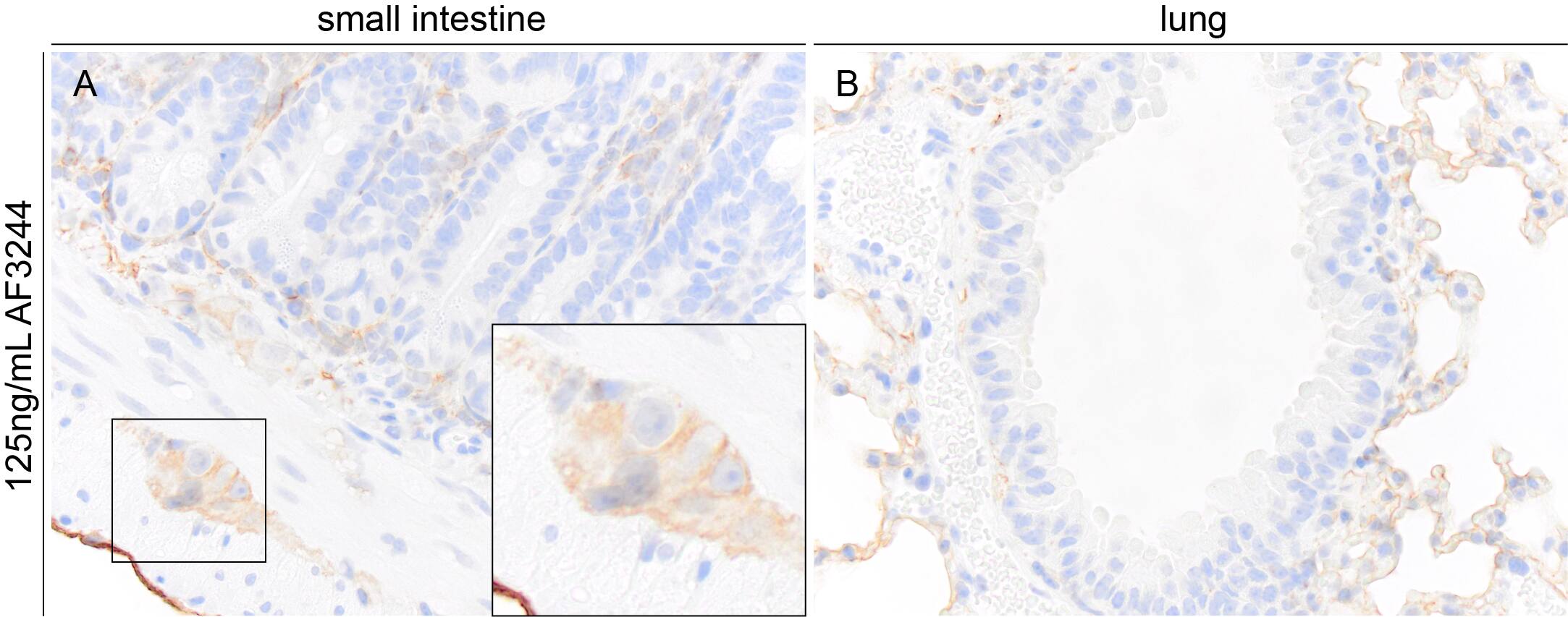
Podoplanin immunoreactivity in FFPE sections of mouse small intestine and lung. AF3244 was used at 125ng/mL and was left on tissue sections for 30m at room temperature. Formalin fixed paraffin embedded sections were deparaffinized and rehydrated. Sections then underwent heat induced epitope retrieval in a citrate-based solution for 20min in a food steamer. Endogenous peroxidase activity was blocked by incubating slides for 15min in tris buffered saline containing 3% (volume/volume) hydrogen peroxide. Tissue sections were then washed, blocked with normal horse serum for 20 min, and then incubated with the primary antibody (125ng/mL) for 30min at room temperature. Sections were then washed and incubated with a horse anti-goat HRP polymer for 30m at room temperature. Sections were washed and incubated with DAB chromogen for approximately 2.5min. Sections were then washed, counterstained with hematoxylin, dehydrated, cleared and mounted with a coverslip. Image from a verified customer review.Podoplanin in Mouse Lung.
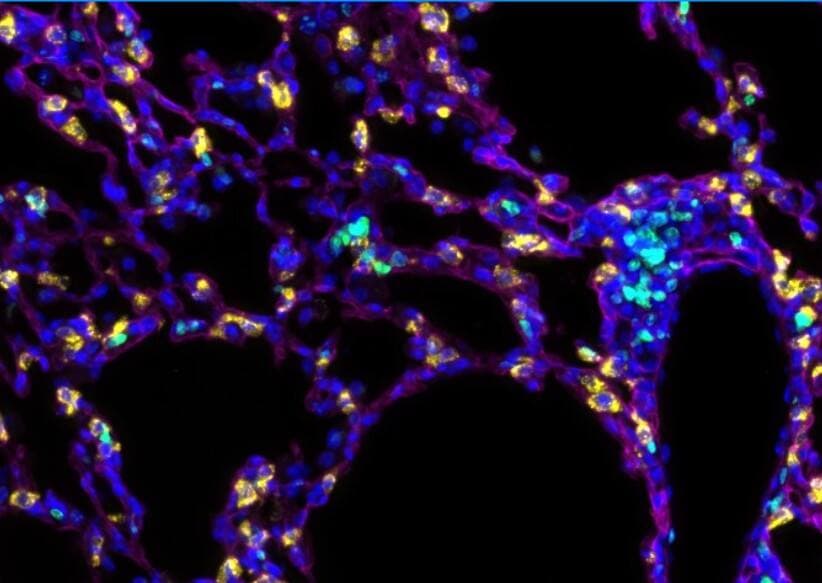
Podoplanin IF staining of mouse lung tissue (Cy5, magenta). Image from a verified customer review.Detection of Mouse Podoplanin by Western Blot
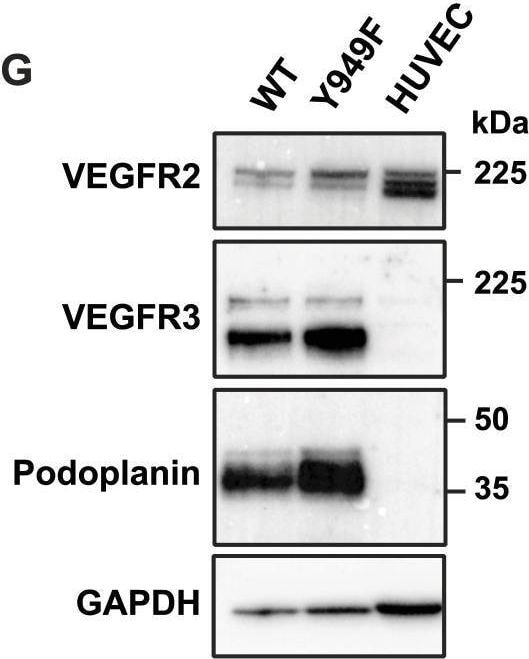
Characterization of dermal lymphatic vessels in WT and Vegfr2Y949F/Y949F embryos and adult mice.(A) Dermal lymphatic vessels in E14.5 WT and Vegfr2Y949F/Y949F (Y949F) embryos shown by immunostaining of Neuropilin2 (Nrp2) in the back skin. Scale bar, 500 μm. (B) Quantification of dermal lymphatic sprouting tips/mm2 in the E14.5 embryos. WT, n = 13; Y949F, n = 10. NS, not significant, t test. (C) Lymphatic vessel density measured by NRP2 staining and normalized to tissue area/field of view in the E14.5 embryos. WT, n = 7; Y949F, n = 6. NS, not significant, t test. (D) Representative images of LYVE1 immunofluorescence showing ear dermal lymphatic vasculature in 8–10-wk-old mice. Scale bar, 1 mm. (E) Quantification of LYVE1+ lymphatic vessel width in 8–10-wk-old mouse ear dermis. n = 4 mice/genotype. NS, not significant, t test. (F) Quantification of LYVE1+ lymphatic vessel density in the 8–10-wk-old mouse ear dermis. WT, n = 4; Y949F, n = 3. P = 0.0087, t test. (G) Expressions of VEGFR2, VEGFR3, and Podoplanin in isolated LYVE1+ cells from lungs of WT and Vegfr2Y949F/Y949F mice at postnatal day 10 and in HUVECs, detected by immunoblotting. (H) Quantification of expression levels normalized to GAPDH in (G).Source data are available for this figure. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/38148112), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Podoplanin Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse kidney
Sample: Perfusion fixed frozen sections of mouse kidney
Simple Western
10 µg/mL
Sample: Mouse lung tissue
Sample: Mouse lung tissue
Western Blot
0.25 µg/mL
Sample: Mouse lung tissue
Sample: Mouse lung tissue
Reviewed Applications
Read 3 reviews rated 4.7 using AF3244 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Podoplanin
Long Name
Lung Type-I Cell Membrane-associated Glycoprotein, Isoform A
Alternate Names
Aggrus, Gp38, PDPN, RANDAM-2, T1A-2
Gene Symbol
PDPN
UniProt
Additional Podoplanin Products
Product Documents for Mouse Podoplanin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Podoplanin Antibody
For research use only
Related Research Areas
Citations for Mouse Podoplanin Antibody
Customer Reviews for Mouse Podoplanin Antibody (3)
4.7 out of 5
3 Customer Ratings
Have you used Mouse Podoplanin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: ImmunofluorescenceSample Tested: Lung tissueSpecies: MouseVerified Customer | Posted 09/08/2025Podoplanin IF staining of mouse lung tissue (Cy5, magenta)IF staining of mouse lung tissue with podoplanin
-
Application: Immunohistochemistry-ParaffinSample Tested: Small Intestine & LungSpecies: MouseVerified Customer | Posted 04/09/2025Podoplanin immunoreactivity in FFPE sections of mouse small intestine and lung. AF3244 was used at 125ng/mL and was left on tissue sections for 30m at room temperature.Formalin fixed paraffin embedded sections were deparaffinized and rehydrated. Sections then underwent heat induced epitope retrieval in a citrate-based solution for 20min in a food steamer. Endogenous peroxidase activity was blocked by incubating slides for 15min in tris buffered saline containing 3% (volume/volume) hydrogen peroxide. Tissue sections were then washed, blocked with normal horse serum for 20 min, and then incubated with the primary antibody (125ng/mL) for 30min at room temperature. Sections were then washed and incubated with a horse anti-goat HRP polymer for 30m at room temperature. Sections were washed and incubated with DAB chromogen for approximately 2.5min. Sections were then washed, counterstained with hematoxylin, dehydrated, cleared and mounted with a coverslip.
-
Application: ImmunocytochemistrySample Tested: See PMID 23892627Species: MouseVerified Customer | Posted 01/07/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

%20IHC-P%20Protocol_bf6cb300-cc3b-471e-881b-a6cda9d78ecf.jpg)