
Key Product Details
Species Reactivity
Rat
Applications
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant rat Fas Ligand/TNFSF6
Leu104-Leu278
Accession # P36940
Leu104-Leu278
Accession # P36940
Specificity
Detects rat Fas Ligand/TNFSF6 in direct ELISAs and Western blots. In direct ELISAs and Western blots, approximately 20% cross-reactivity with recombinant mouse Fas Ligand is observed and 5% cross-reactivity with recombinant human Fas Ligand is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
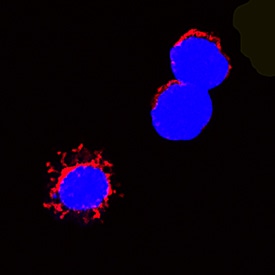
Scientific Data Images for Rat Fas Ligand/TNFSF6 Antibody
Fas Ligand/TNFSF6 in Rat Splenocytes.
Fas Ligand/TNFSF6 was detected in immersion fixed rat splenocytes using Goat Anti-Rat Fas Ligand/TNFSF6 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1858) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Rat Fas Ligand/TNFSF6 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed rat splenocytes
Sample: Immersion fixed rat splenocytes
Western Blot
0.1 µg/mL
Sample: Recombinant Rat Fas Ligand/TNFSF6 (Catalog # 1614-FL)
Sample: Recombinant Rat Fas Ligand/TNFSF6 (Catalog # 1614-FL)
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Fas Ligand/TNFSF6
References
- Lettau, M. et al. (2008) Curr. Med. Chem. 15:1684.
- Takahashi, T. et al. (1994) Int. Immunol. 6:1567.
- Seino, K-I. et al. (1998) J. Immunol. 161:4484.
- Suda, T. et al. (1993) Cell 75:1169.
- Pitti, R.M. et al. (1998) Nature 396:699.
- Schneider, P. et al. (1998) J. Exp. Med. 187:1205.
- Tanaka, M. et al. (1998) Nature Med. 4:31.
- Chen, J.-J. et al. (1998) Science 282:1714.
- Suzuki, I. and P.J. Fink (2000) Proc. Natl. Acad. Sci. 97:1707.
- Ferguson, T.A. and T.S. Griffith (2006) Immunol. Rev. 213:228.
- Ryan, A.E. et al. (2005) Cancer Res. 65:9817.
- Takahashi, T. et al. (1994) Cell 76:969.
- Lynch, D.H. et al. (1994) Immunity 1:131.
Alternate Names
CD178, CD95L, FASLG, TNFSF6
Gene Symbol
FASLG
UniProt
Additional Fas Ligand/TNFSF6 Products
Product Documents for Rat Fas Ligand/TNFSF6 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Rat Fas Ligand/TNFSF6 Antibody
For research use only
Customer Reviews for Rat Fas Ligand/TNFSF6 Antibody
There are currently no reviews for this product. Be the first to review Rat Fas Ligand/TNFSF6 Antibody and earn rewards!
Have you used Rat Fas Ligand/TNFSF6 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions
TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions

