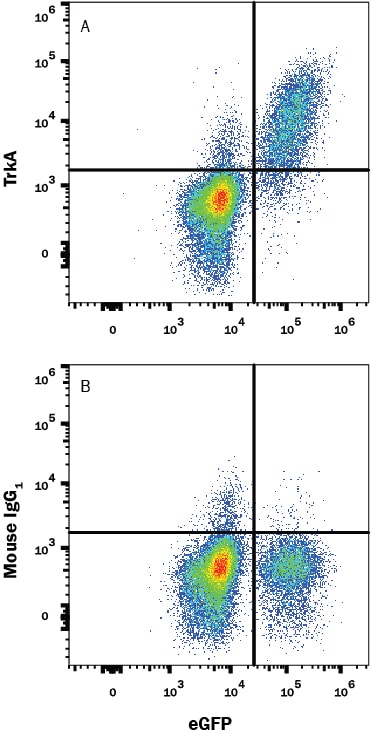
367 results for "TrkA" in Products
TrkA Products
TrkA, the product of the protooncogene trk, is a member of the neurotrophic tyrosine kinase receptor family that has three members. TrkA, TrkB and TrkC preferentially bind NGF, NT-4 and BDNF, and NT-3, respectively. All Trk family proteins share a conserved complex subdomain organization consisting of a signal peptide, two cysteine-rich domains, a cluster of three leucine-rich motifs, and two immunoglobulin-like domains in the extracellular region, as well as an intracellular region that ...
| Reactivity: | Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
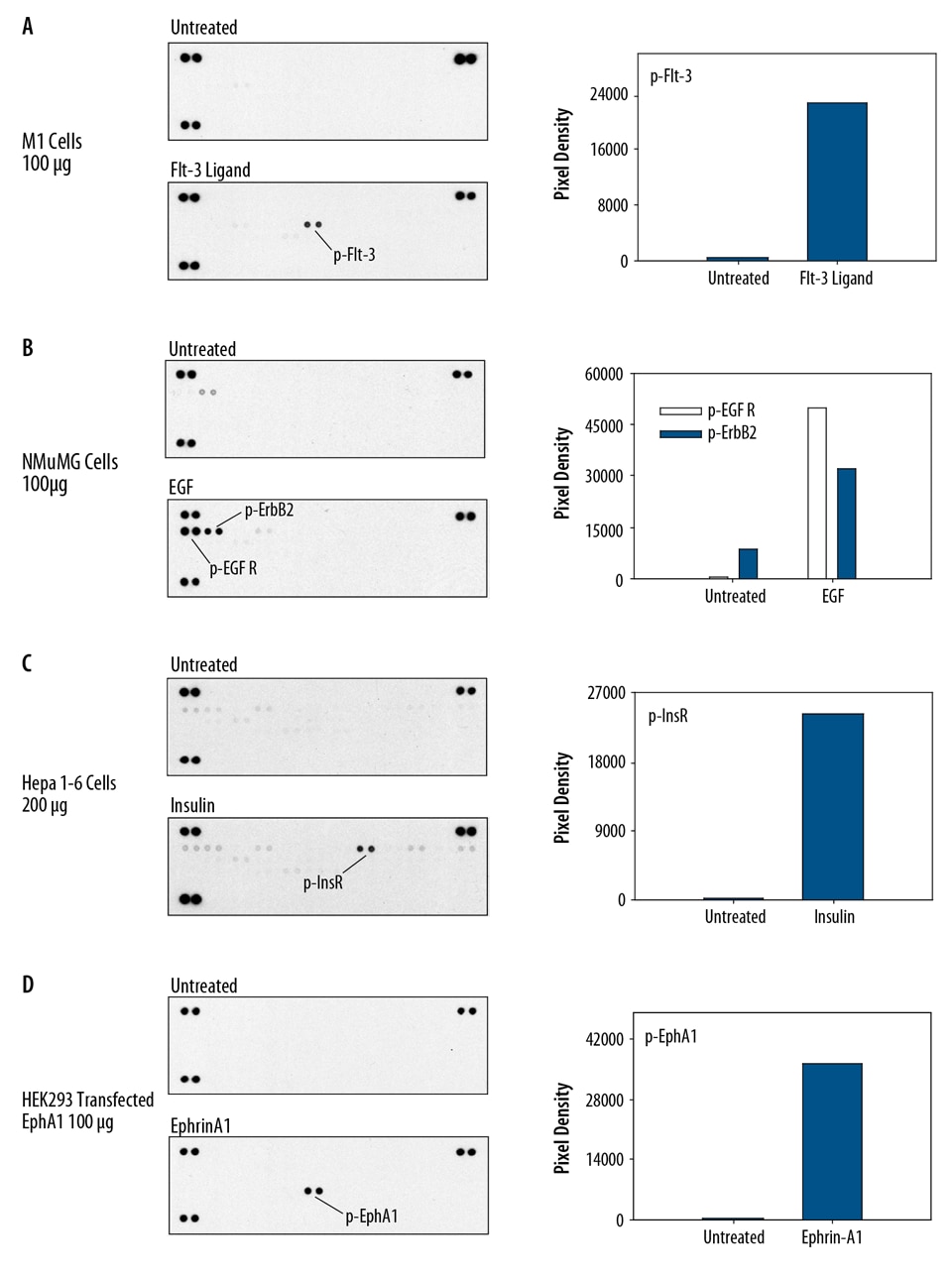
Contains 4 membranes - each spotted in duplicate with 49 different RTK antibodies
| Reactivity: | Human |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB, B/N, Flow, CyTOF-ready |
| Reactivity: | Human, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |
| Reactivity: | Human |
| Source: | NS0 |
| Accession #: | AAA36770 |
| Applications: | Bioactivity |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #JJ084-04 |
| Applications: | IHC, WB, Flow, ICC/IF, IP |
Recombinant Monoclonal Antibody.
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #165131R |
| Applications: | KO, Flow, CyTOF-ready |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #994307 |
| Applications: | B/N, Flow, ICC/IF, CyTOF-ready |
| Reactivity: | Rat |
| Details: | Mouse IgG2B Monoclonal Clone #315104 |
| Applications: | WB |
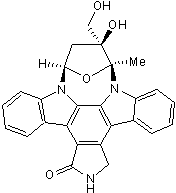
JAK2, FLT3 and TrkA inhibitor; also inhibits AurA and AurB
| Alternate Names: | CEP-701 |
| Chemical Name: | (9S,10S,12R)-2,3,9,10,11,12-Hexahydro-10-hydroxy-10-(hydroxymethyl)-9-methyl-9,12-epoxy-1H-diindolo[1,2,3-fg:3',2',1'-kl]pyrrolo[3,4-i][1,6]benzodiazocin-1-one |
| Purity: | ≥98% |
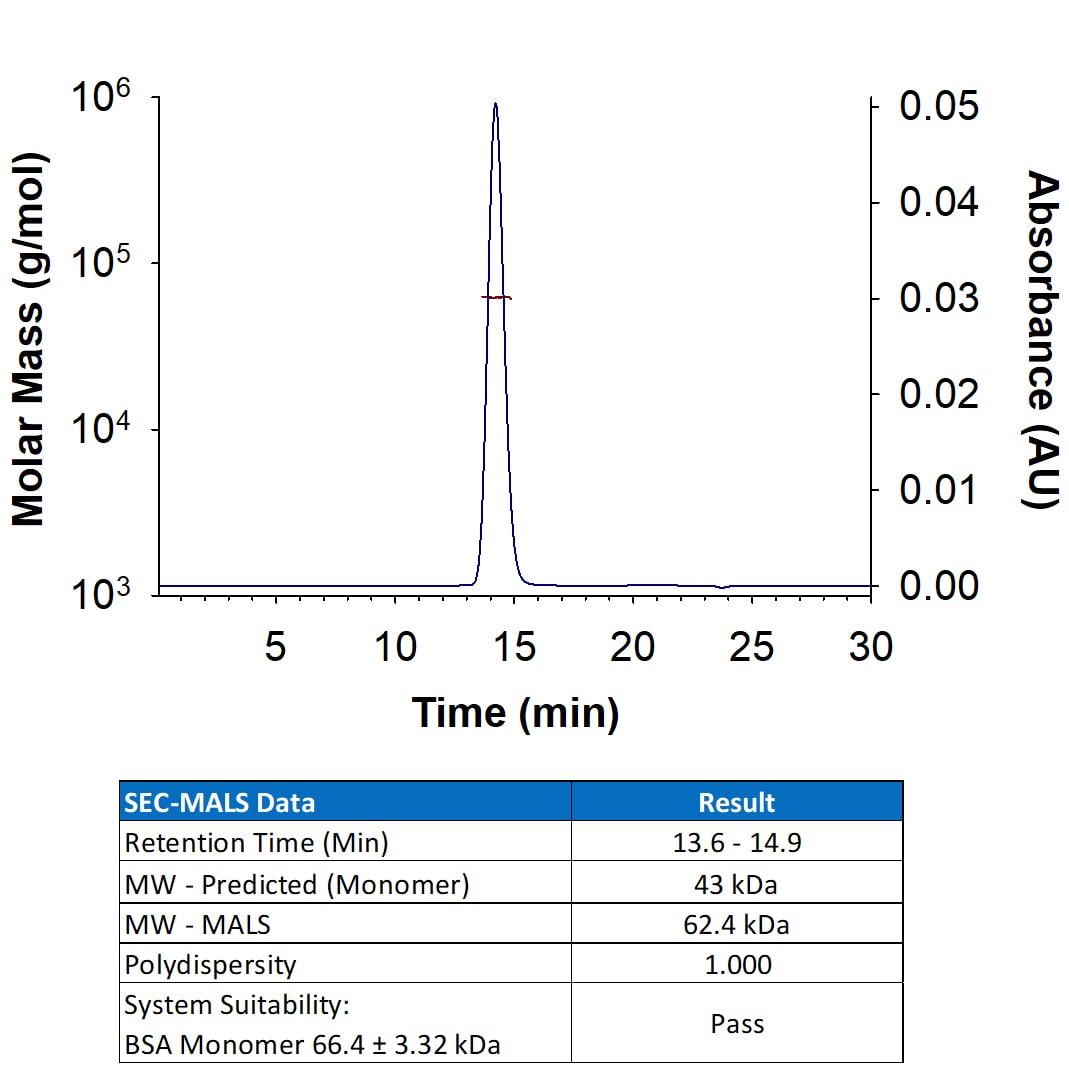
Analyzed by SEC-MALS
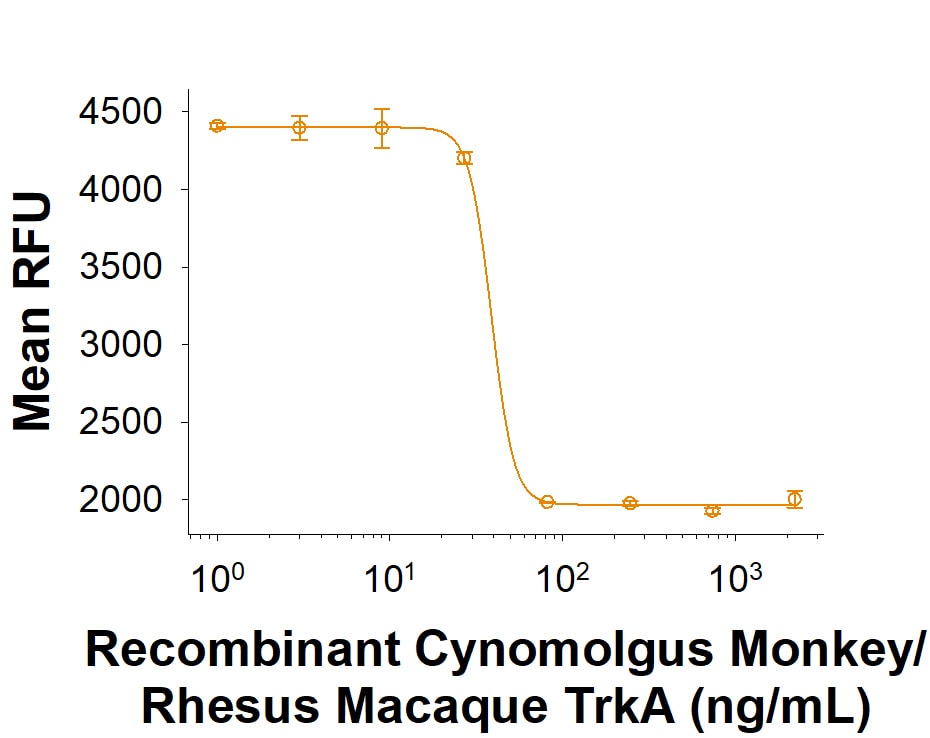
| Reactivity: | Cynomolgus Monkey, Rhesus Macaque |
| Source: | CHO |
| Accession #: | XP_005541454.3 |
| Applications: | Bioactivity |
| Reactivity: | Rat |
| Source: | NS0 |
| Accession #: | P35739 |
| Applications: | Bioactivity |
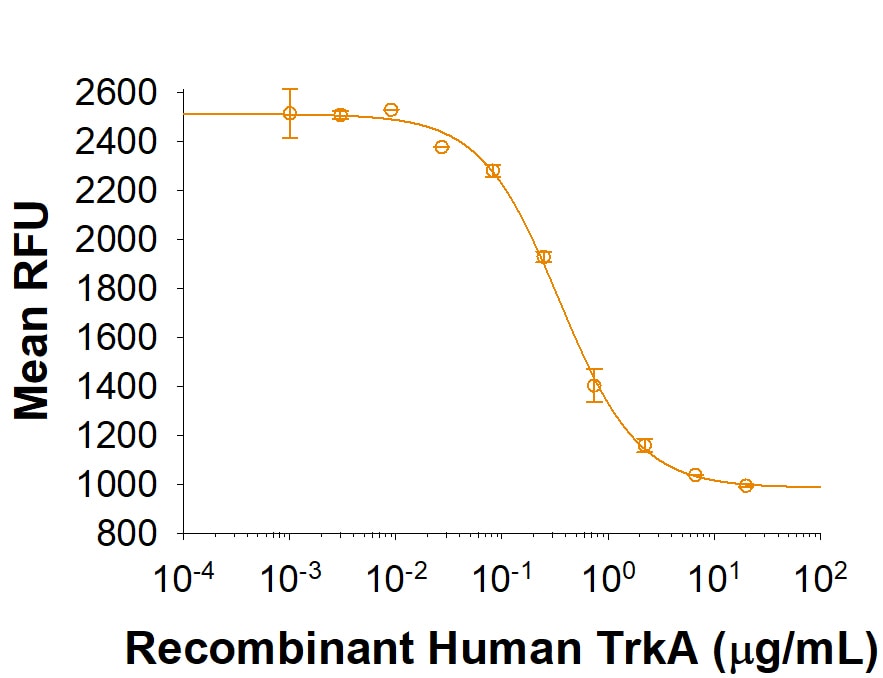
| Reactivity: | Human |
| Source: | HEK293 |
| Accession #: | P04629-1 |
| Applications: | Bioactivity |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
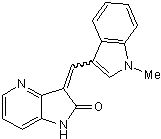
Potent, selective TrkA inhibitor
| Chemical Name: | 1,3-Dihydro-3-[(1-methyl-1H-indol-3-yl)methylene]-2H-pyrrolo[3,2-b]pyridin-2-one |
| Purity: | ≥99% |
| Reactivity: | Cynomolgus Monkey, Rhesus Macaque |
| Source: | CHO |
| Accession #: | XP_005541454.3 |
| Applications: | Bioactivity |
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #165126 |
| Applications: | WB |
| Reactivity: | Mouse |
| Source: | NS0 |
| Accession #: | Q3UFB7 |
| Applications: | Bioactivity |
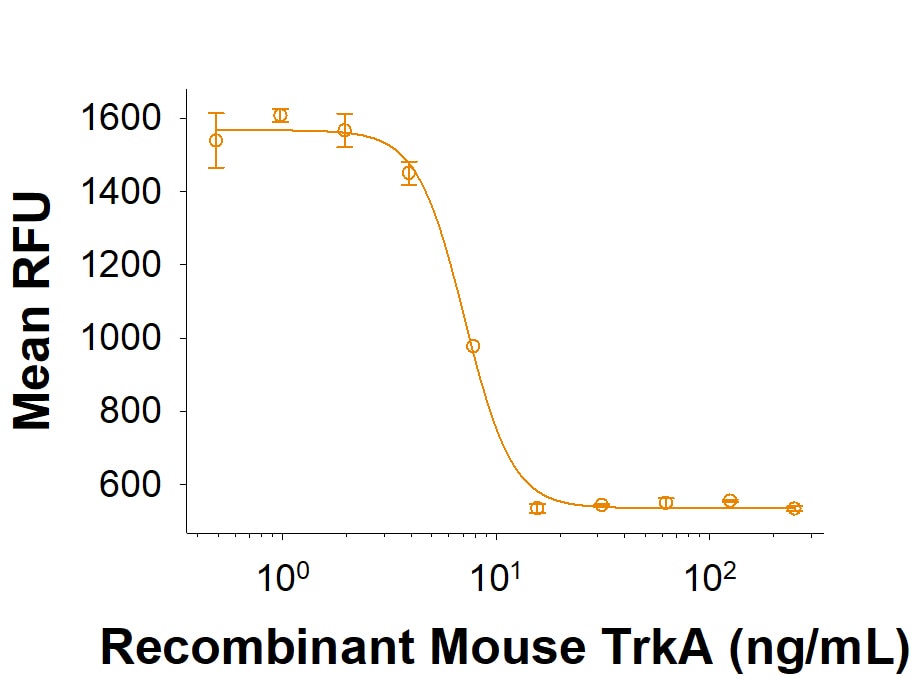
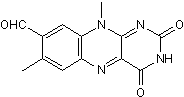
Inhibits NGF binding to p75NTR and TrkA
| Chemical Name: | 2,3,4,10-Tetrahydro-7,10-dimethyl-2,4-dioxobenzo[g]pteridine-8-carboxaldehyde |
| Purity: | ≥95% |
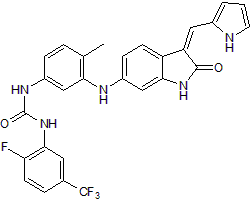
Potent Trk inhibitor; inhibits TrkA, TrkB and TrkC
| Chemical Name: | N-[3-[[2,3-Dihydro-2-oxo-3-(1H-pyrrol-2-ylmethylene)-1H-indol-6-yl]amino]-4-methylphenyl]-N'-[2-fluoro-5-(trifluoromethyl)phenyl]urea |
| Purity: | ≥98% |
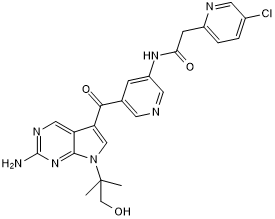
Potent and selective pan-Trk inhibitor; peripherally restricted
| Chemical Name: | N-[5-[[2-Amino-7-(2-hydroxy-1,1-dimethylethyl)-7H-pyrrolo[2,3-d]pyrimidin-5-yl]carbonyl]-3-pyridinyl]-5-chloro-2-pyridineacetamide |
| Purity: | ≥98% |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #OTI5B6 |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #OTI5B6 |
| Applications: | IHC, WB |
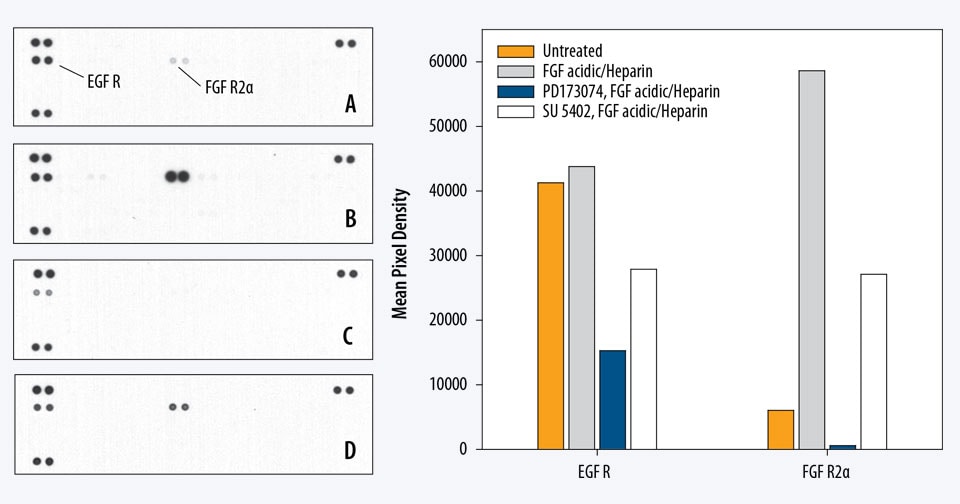
Contains 4 membranes - each spotted in duplicate with 39 different RTK antibodies
| Reactivity: | Mouse |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #165131R |
| Applications: | KO, Flow |


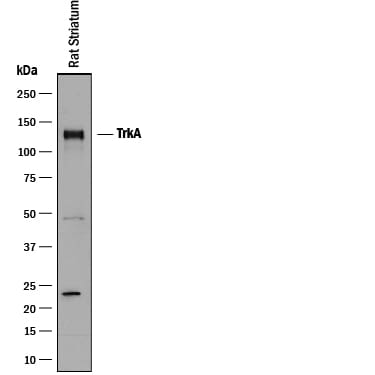
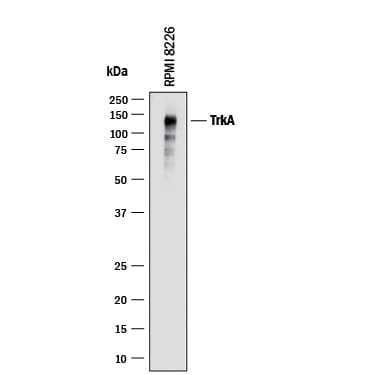



![Western Blot: TrkA Antibody (JJ084-04) [NBP2-67473] Western Blot: TrkA Antibody (JJ084-04) [NBP2-67473]](https://resources.rndsystems.com/images/products/TrkA-Antibody-JJ084-04-Western-Blot-NBP2-67473-img0010.jpg)
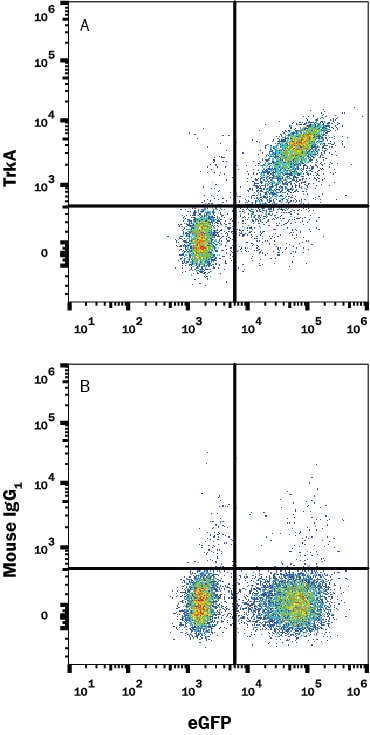
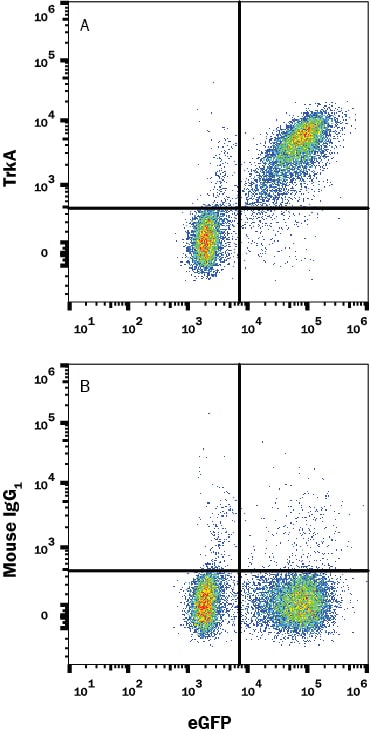

![Immunohistochemistry-Paraffin: TrkA Antibody [NBP2-38265] Immunohistochemistry-Paraffin: TrkA Antibody [NBP2-38265]](https://resources.rndsystems.com/images/products/TrkA-Antibody-Immunohistochemistry-Paraffin-NBP2-38265-img0005.jpg)
![Western Blot: TrkA Antibody (OTI5B6) [NBP2-46266] Western Blot: TrkA Antibody (OTI5B6) [NBP2-46266]](https://resources.rndsystems.com/images/products/TrkA-Antibody-5B6-Western-Blot-NBP2-46266-img0002.jpg)
![Western Blot: TrkA Antibody (OTI5B6)Azide and BSA Free [NBP2-74623] Western Blot: TrkA Antibody (OTI5B6)Azide and BSA Free [NBP2-74623]](https://resources.rndsystems.com/images/products/TrkA-Antibody-OTI5B6-Azide-and-BSA-Free-Western-Blot-NBP2-74623-img0001.jpg)