Interleukin 12 (IL-12) and IL-23 are secreted heterodimeric glycoproteins belonging to the IL-12 cytokine family. The two cytokines share a common p40 (40 kDa) subunit, which is disulfide-linked with the p35 (35 kDa) subunit in IL-12, and with the p19 (19 kDa) subunit in IL-23. Feline p40 is synthesized as a 329 amino acid (aa) precursor with a 22 aa signal sequence and a 307 aa mature region. It contains a 90 aa fibronectin type III domain and a 75 aa Ig C2-like region. The expression of p40 is induced by substances such as LPS and CpG that activate antigen-presenting cells. Besides being found as a component of IL-12 or IL-23, free p40 monomers and homodimers are also secreted by cells expressing p40. Feline p40 shares 94%, 85%, 84%, 65%, and 65% aa sequence identity with canine, human, porcine, rat and mouse p40, respectively. Cells known to express p40 include macrophages, dendritic cells, monocytes, Langerhans cells, neutrophils, keratinocytes, plasmacytoid dendritic cells, and microglia. From cells that express both the p35 and p40 subunits (dendritic cells, monocytes, and CHO cells), the amount of free p40 secreted is 10‑1000 fold more than the heterodimeric IL-12. The high-affinity IL-12 receptor complex that transduces IL-12 signals is composed of a 100 kDa ligand-binding subunit (IL-12 R beta 1) and a 130 kDa signal transducing subunit (IL-12 R beta 2). Similarly, the high-affinity IL-23 signaling receptor complex is composed of the shared IL-12 R beta 1 and the unique IL-23 R, a novel gp130-like protein. Both the monomeric and the dimeric free p40 can bind to the IL-12 R beta 1 and function as antagonists of IL-12 or IL-23. However, the monomeric p40 binds IL-12 R beta 1 with lower affinity and is less potent as an IL-12 antagonist. Homodimeric mouse p40 has also been shown to have agonistic functions similar to IL-12, inducing nitric oxide expression and NF kappa B activation in mouse primary microglia and peritoneal macrophages. The molecular mechanism for the agonistic effects of homodimeric p40 has not been determined (1‑6).
Feline IL‑12/IL‑23 p40 Antibody
R&D Systems | Catalog # AF1954


Key Product Details
Species Reactivity
Validated:
Feline
Cited:
Avian - Chicken
Applications
Validated:
Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization, Immunocytochemistry
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant feline IL‑12/IL‑23 p40
Ile23-Ser329 (Glu167Gly)
Accession # O02744
Ile23-Ser329 (Glu167Gly)
Accession # O02744
Specificity
Detects feline IL‑12/IL‑23 p40 in ELISAs and Western blots. In sandwich immunoassays, approximately 65% cross‑reactivity with recombinant canine IL-12/23 p40 is observed, less than 3% cross-reactivity with recombinant mouse IL-12/23 p40 is observed, and less than 0.2% cross-reactivity with recombinant human IL‑12/23 p40 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Feline IL‑12/IL‑23 p40 Antibody
Cell Proliferation Induced by IL‑12 and Neutralization by Feline IL‑12/IL‑23 p40 Antibody.
Recombinant Feline IL-12 (Catalog # 1954-FL) stimulates proliferation in PHA-activated human peripheral blood mononuclear cells (PBMC) in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Feline IL-12 (25 ng/mL) is neutralized (green line) by increasing concen-trations of Goat Anti-Feline IL-12/IL-23 p40 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1954). The ND50 is typically 0.5-2.0 µg/mL.IL‑12/IL‑23 p40 in Feline PBMCs.
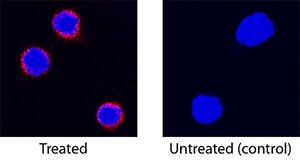
IL-12/IL-23 p40 was detected in immersion fixed feline peripheral blood mononuclear cells (PBMCs) stimulated with PMA and calcium ionomycin using Goat Anti-Feline IL-12/IL-23 p40 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1954) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Feline IL‑12/IL‑23 p40 Antibody
Application
Recommended Usage
Immunocytochemistry
10-25 µg/mL
Sample: Immersion fixed feline peripheral blood mononuclear cells treated with PMA and calcium ionomycin
Sample: Immersion fixed feline peripheral blood mononuclear cells treated with PMA and calcium ionomycin
Western Blot
0.1 µg/mL
Sample: Recombinant Feline IL‑12/IL‑23 p40 (Catalog # 2117-FL)
Sample: Recombinant Feline IL‑12/IL‑23 p40 (Catalog # 2117-FL)
Neutralization
Measured by its ability to neutralize IL‑12-induced proliferation in PHA-activated human peripheral blood mononuclear cells (PBMC). Yokota, T. et al. (1986) Proc. Natl. Acad. Sci. USA 83:5894. The Neutralization Dose (ND50) is typically 0.5-2.0 µg/mL in the presence of 25 ng/mL Recombinant Feline IL‑12.
Feline IL-12/IL-23 p40 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
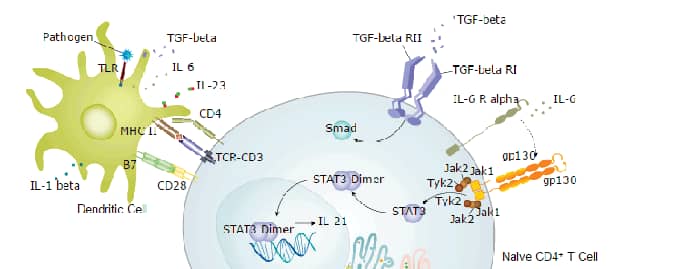
Background: IL-12/IL-23 p40
References
- Buttner, M. et al. (1997) Cytokine 10:241.
- Park, A.Y. and P. Scott (2001) Scand. J. Immunol. 53:529.
- Trinchieri, G. et al. (2003) Immunity 19:641.
- Brombacher, F. et al. (2003) Trends Immunol. 24:207.
- Lankford, C.S. and D.M. Frucht, 2003, J. Leukoc. Biol. 73:49.
- Abdi, K. (2002) Scand. J. Immunol. 5:1.
Long Name
Interleukin 12/Interleukin 23 p40
Alternate Names
CLMF2, IL-12B, IL12 p40, IL12B, NKSF2
Entrez Gene IDs
Gene Symbol
IL12B
UniProt
Additional IL-12/IL-23 p40 Products
Product Documents for Feline IL‑12/IL‑23 p40 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Feline IL‑12/IL‑23 p40 Antibody
For research use only
Citations for Feline IL‑12/IL‑23 p40 Antibody
Customer Reviews for Feline IL‑12/IL‑23 p40 Antibody
There are currently no reviews for this product. Be the first to review Feline IL‑12/IL‑23 p40 Antibody and earn rewards!
Have you used Feline IL‑12/IL‑23 p40 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars