Caspase-2 (Cysteine-aspartic acid protease 2/Casp2; also NEDD2 and ICH-1) is a 30‑32 kDa member of the peptidase C14A/IL-1 beta -converting family of enzymes (1‑3). It is widely expressed and is an integral component of the apoptotic cascade. Based on the length of its prodomain, caspase-2 has been considered to be an initiator caspase. However, studies have shown that other caspases (such as Casp 3) activate procaspase 2, and Caspase-2 likely acts on key cellular molecules such as BID, Golgin 160 and DFF45/ICAD (2, 4, 5). Thus, Caspase-2 is perhaps more likely to be a specialized executioner caspase. Human procaspase-2 is a 48‑51 kDa, 452 amino acid (aa) protein (4‑7). It is known to exist as a disulfide-linked homodimer via covalent linkage at Cys436 (2, 5). But this dimeric state may not be sufficient for (auto)activation. Actual activation may occur following oligomerization within the context of activating platforms such as DISC (death-inducing signaling complex) or the PIDDosome (8-10). Initially, procaspase-2 undergoes proteolytic cleavage to generate an N‑terminal 333 aa p34/34 kDa subunit, and a 119 aa C‑terminal p14/14 kDa subunit (5). The p34 and p14 subunits are further processed to generate the prodomain (aa 1-169), plus the mature p18 (aa 170-333) and p12 (aa 348-452) subunits (4-6). Notably, each p18:p12 noncovalent heterodimer demonstrates proteolytic activity around a catalytic site at aa 318-322, and, due to an nuclear localization signal within the prodomain, may be found in either nucleus or cytoplasm (11, 12). There are multiple potential isoform variants. Individually, or in combination, there is an alternative start site at Met18, a substitution of two aa for aa 107-452, a second substitution of 14 aa for aa 309-322, and a third substitution of 22 aa for aa 323-452 (6, 7, 13). The human and mouse procaspase 2 precursors are 90% aa identical, with the majority of differences lying in the prodomain.

Key Product Details
Validated by
Biological Validation
Species Reactivity
Human
Applications
Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 691233
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Caspase-2
Gly170-Asp333 & Ala348-Tyr452
Accession # P42575
Gly170-Asp333 & Ala348-Tyr452
Accession # P42575
Specificity
Detects human Caspase-2 in direct ELISAs. In direct ELISAs, no cross-reactivity with recombinant human Caspase-8 is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human Caspase‑2 Antibody
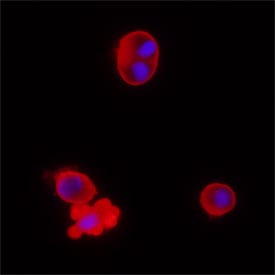
Caspase‑2 in Jurkat Human Cell Line.
Caspase-2 was detected in immersion fixed Jurkat human acute T cell leukemia cell line treated with with staurosporin using Mouse Anti-Human Caspase-2 Monoclonal Antibody (Catalog # MAB7228) at 25 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Human Caspase‑2 Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed Jurkat human acute T cell leukemia cell line treated with with staurosporin
Sample: Immersion fixed Jurkat human acute T cell leukemia cell line treated with with staurosporin
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Caspase-2
References
- Chowdhury, I. et al. (2008) Comp. Biochem. Physiol. B 151:10.
- Krumschnabel, G. et al. (2009) Cell Death Differ.16:195.
- Kitevska, T. et al. (2009) Apoptosis 14:829.
- Paroni, G. et al. (2001) J. Biol. Chem. 276:21907.
- Li, H. et al. (1997) J. Biol. Chem. 272:21010.
- SwissProt. Accession # P42575.
- Wang, L. et al. (1994) Cell 78:739.
- Chang, D.W. et al. (2003) J. Biol. Chem. 278:16466.
- Olsson, M. et al. (2009) Oncogene 28:1949.
- Tinel, A. & J. Tschopp (2004) Science 304:843.
- Schweizer, A. et al. (2003) J. Biol. Chem. 278:42441.
- Colussi, P.A. et al. (1998) J. Biol. Chem. 273:24535.
- Droin, N. et al. (2000) Cancer Res. 60:7039.
Alternate Names
CASP2, Caspase2, ICH-1, NEDD2
Gene Symbol
CASP2
UniProt
Additional Caspase-2 Products
Product Documents for Human Caspase‑2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Caspase‑2 Antibody
For research use only
Related Research Areas
Customer Reviews for Human Caspase‑2 Antibody
There are currently no reviews for this product. Be the first to review Human Caspase‑2 Antibody and earn rewards!
Have you used Human Caspase‑2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways

