Cathepsin C, also known as dipeptidyl-peptidase I (DPPI), is a cysteine protease of the papain family (1). Cathepsin C sequentially removes dipeptides from the free N-termini of proteins and peptides. It has broad specificity except that it does not cleave a basic amino acid (Arg or Lys) in the N-terminal position or Pro on either side of the scissle bond. It requires halide ions for activity. The pro form contains a pro peptide and a catalytic region, which can be further processed into heavy/ alpha and light/ beta chains that are linked by a disulfide bond. It is broadly distributed. Cathepsin C plays a role in the lysosomal degradation. It also functions as a key enzyme in the activation of granule serine proteases in cytotoxic T lymphocytes and natural killer cells (granzymes A and B), mast cells (tryptase and chymase), and neutrophils (Cathepsin G and elastase) by removing their N-terminal activation dipeptides (2). Loss of function mutations in the Cathepsin C gene result in periodontal disease and palmoplantar keratosis (3).
Human Cathepsin C/DPPI Antibody
R&D Systems | Catalog # AF1071

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human pro Cathepsin C/DPPI
Asp25-Leu463
Accession # P53634
Asp25-Leu463
Accession # P53634
Specificity
Detects human Cathepsin C/DPPI in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Cathepsin C/DPPI Antibody
Detection of Human Cathepsin C/DPPI by Western Blot.
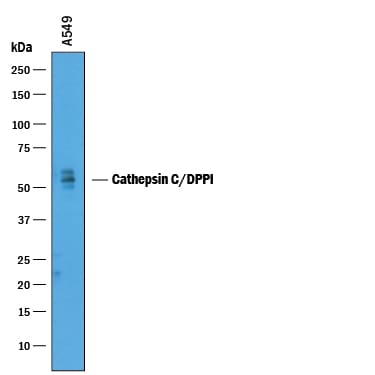
Western blot shows lysates of A549 human lung carcinoma cell line. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human Cathepsin C/DPPI Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1071) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for Cathepsin C/DPPI at approximately 55 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human Cathepsin C/DPPI by Simple WesternTM.
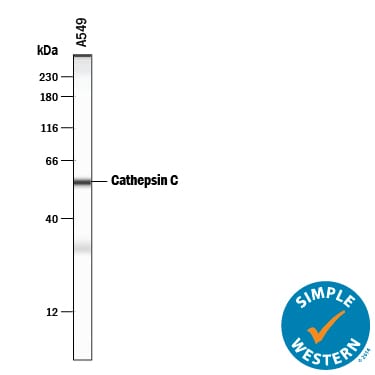
Simple Western lane view shows lysates of A549 human lung carcinoma cell line, loaded at 0.2 mg/mL. A specific band was detected for Cathepsin C/DPPI at approximately 56 kDa (as indicated) using 50 µg/mL of Goat Anti-Human Cathepsin C/DPPI Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1071) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system. Non-specific interaction with the 230 kDa Simple Western standard may be seen with this antibody.Cathepsin C/DPPI in Human Lung Cancer Tissue.
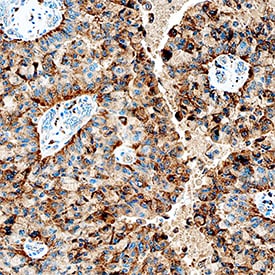
Cathepsin C/DPPI was detected in immersion fixed paraffin-embedded sections of human lung cancer tissue using Goat Anti-Human Cathepsin C/DPPI Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1071) at 1 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in cancer cells. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Cathepsin C/DPPI by Western Blot
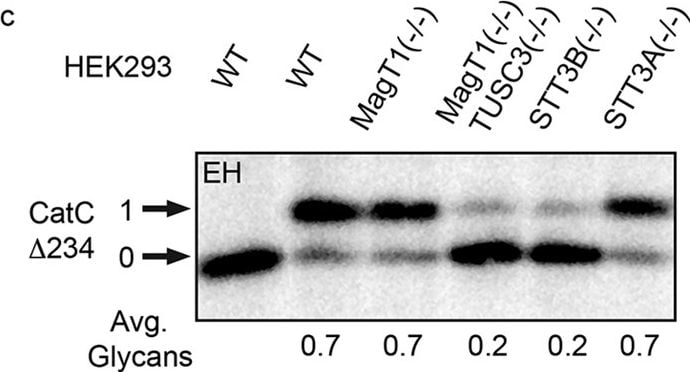
Hypoglycosylation of proteins in knockout HEK293 cell lines.Cell lines were transfected with expression vectors for the following human glycoproteins: (a) SHBG, (b) SHBG derivatives (see diagram) with single glycosylation sites (SHBG N380Q and N396Q), (c) a cathepsin C derivative with a single glycosylation site (CatC delta 234-HA), (d) prosaposin (pSAP-DDKHis), (e) haptoglobin (Hp-DDKHis), (f) hemopexin (Hpx-DDKHis). Endogenous progranulin (pGran) (d) was analyzed using non-transfected cells. The cells were pulse labeled for 10 min and chased for 10 min (pSAP, pGran and CatC delta 234-HA) or pulse labeled for 5 min and chased for 20 min (Hp, SHBG and Hpx). Glycoproteins precipitated with anti-DDK, anti-HA or anti-SHBG were resolved by PAGE in SDS. EH designates treatment with endoglycosidase H. Quantified values below gel lanes (a–f) are for the displayed image, which is representative of two or more experiments. (b) The vertical line indicates the excision of three intervening gel lanes. (d) Resolution of pGran glycoforms is not sufficient for quantification. Nonglycosylated forms of pSAP, Hp, and Hpx that comigrate with the EH-digested form of the substrate in HEK293 cells correspond to a non-translocated precursor and were not used to calculate the average number of glycans. (a–f) Phosphorimages were cropped to display the region of interest. Full-length phosphorimages are shown in Supplemental Fig. S5. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/26864433), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Cathepsin C/DPPI Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lung subjected to Antigen Retrieval Reagent-Basic (Catalog # CTS013) and immersion fixed paraffin-embedded sections of human lung cancer tissue
Sample: Immersion fixed paraffin-embedded sections of human lung subjected to Antigen Retrieval Reagent-Basic (Catalog # CTS013) and immersion fixed paraffin-embedded sections of human lung cancer tissue
Simple Western
50 µg/mL
Sample: A549 human lung carcinoma cell line
Sample: A549 human lung carcinoma cell line
Western Blot
1 µg/mL
Sample: A549 human lung carcinoma cell line
Sample: A549 human lung carcinoma cell line
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cathepsin C/DPPI
References
- Turk, B. et al. (2004) in Handbook of Proteolytic Enzymes (ed. Barrett, A.J. et al.) p. 1192, Academic Press, San Diego.
- Dahl, S.W. et al. (2001) Biochemistry 40:1671.
- Toomes, A.J. et al. (1999) Nat. Genet. 23:421.
Alternate Names
CTSC, DPPI, PALS, PLS
Gene Symbol
CTSC
UniProt
Additional Cathepsin C/DPPI Products
Product Documents for Human Cathepsin C/DPPI Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Cathepsin C/DPPI Antibody
For research use only
Related Research Areas
Citations for Human Cathepsin C/DPPI Antibody
Customer Reviews for Human Cathepsin C/DPPI Antibody
There are currently no reviews for this product. Be the first to review Human Cathepsin C/DPPI Antibody and earn rewards!
Have you used Human Cathepsin C/DPPI Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...