CD155 [also known as PVR (poliovirus receptor) and Necl-5 (nectin-like molecule-5)] is a 70 kDa type I transmembrane (TM) glycoprotein that is a member of the nectin-like (Necl) family of nectin-related molecules (1). Like nectins, Necl molecules are Ig superfamily members that contain three Ig-like extracellular domains, a TM segment, and a cytoplasmic tail. Unlike nectins, Necl molecules cannot interact with cytoplasmic afadin (1). While Nectins serve as cell adhesion molecules, the actual functions of most Necls are yet-to-be determined. CD155/PVR was originally isolated based on its ability to mediate polio virus attachment to host cells (2, 3). The full-length (or CD155 alpha isoform) is synthesized as a 417 amino acid (aa) precursor that contains a 20 aa signal sequence, a 323 aa extracellular region, a 24 aa TM segment and a 50 aa cytoplasmic tail. The extracellular region contains one N-terminal V-type and two C2-type Ig-like domains (2, 3). The V‑type domain mediates polio virus binding (4). Three other isoforms exist, all of which retain the Ig-like domains. CD155 delta is transmembrane with a shortened cytoplasmic tail of 25 aa. CD155 beta (352 aa) and CD155 gamma (344 aa) are 60‑65 kDa soluble forms that show removal of the TM segment and surrounding amino acids (2, 5). The soluble forms will bind the polio virus (due to the presence of the V-type Ig domain) but afford no protection against polio infection because of low circulating levels (5). CD155 has been demonstrated to bind vitronectin, nectin-3, and DNAM-1 (6‑8). DNAM-1 binding promotes monocyte migration and NK cell killing. CD155 is expressed in all normal tissues and is highly expressed in tumor cells of epithelial and neuronal origin.
Human CD155/PVR Antibody
R&D Systems | Catalog # AF2530

Key Product Details
Validated by
Knockout/Knockdown
Species Reactivity
Human
Applications
Knockout Validated, Western Blot, Immunocytochemistry, Simple Western
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human CD155/PVR
Gly27-Asn343
Accession # AAH15542
Gly27-Asn343
Accession # AAH15542
Specificity
Detects human CD155/PVR in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human CD155/PVR Antibody
Detection of Human CD155/PVR by Western Blot.
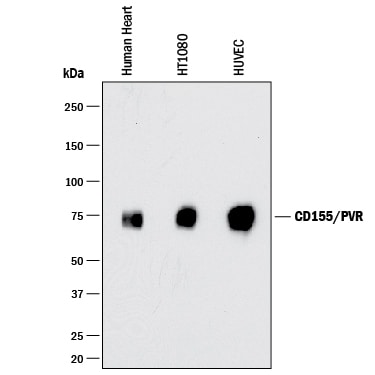
Western blot shows lysates of human heart tissue, HT1080 human fibrosarcoma cell line, and HUVEC human umbilical vein endothelial cells. PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Human CD155/PVR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2530) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for CD155/PVR at approximately 75 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.CD155/PVR in U937 human histiocytic lymphoma cell line.
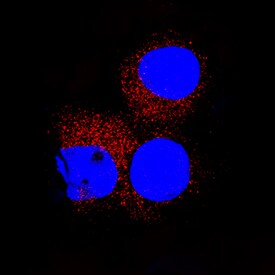
CD155/PVR was detected in immersion fixed U937 human histiocytic lymphoma cell line using Goat Anti-Human CD155/PVR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2530) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of Human CD155/PVR by Simple WesternTM.
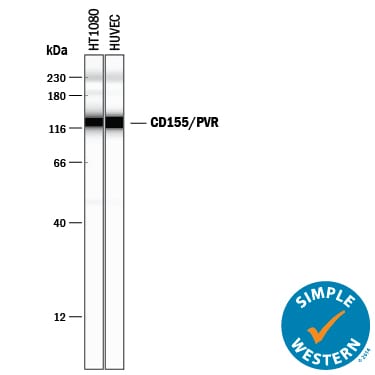
Simple Western lane view shows lysates of HT1080 human fibrosarcoma cell line and HUVEC human umbilical vein endothelial cells, loaded at 0.2 mg/mL. A specific band was detected for CD155/PVR at approximately 128 kDa (as indicated) using 2.5 µg/mL of Goat Anti-Human CD155/PVR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2530) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Western Blot Shows Human CD155/PVR Specificity by Using Knockout Cell Line.
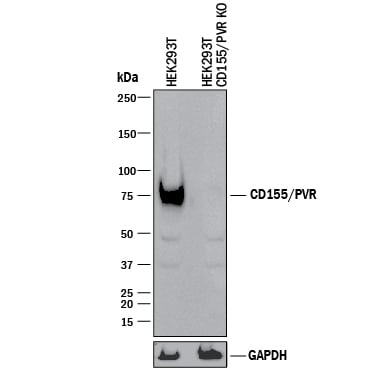
Western blot shows lysates of HEK293T human embryonic kidney parental cell line and CD155/PVR knockout HEK293T cell line (KO). PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Human CD155/PVR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2530) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for CD155/PVR at approximately 75 kDa (as indicated) in the parental HEK293T cell line, but is not detectable in knockout HEK293T cell line. GAPDH (Catalog # AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Human CD155/PVR Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed U937 human histiocytic lymphoma cell line
Sample: Immersion fixed U937 human histiocytic lymphoma cell line
Knockout Validated
CD155/PVR
is specifically detected in HEK293T human embryonic kidney parental cell line but is not detectable in
CD155/PVR knockout HEK293T cell line.
Simple Western
2.5 µg/mL
Sample: HT1080 human fibrosarcoma cell line and HUVEC human umbilical vein endothelial cells
Sample: HT1080 human fibrosarcoma cell line and HUVEC human umbilical vein endothelial cells
Western Blot
0.25 µg/mL
Sample: Human heart tissue, HT1080 human fibrosarcoma cell line, and HUVEC human umbilical vein endothelial cells
Sample: Human heart tissue, HT1080 human fibrosarcoma cell line, and HUVEC human umbilical vein endothelial cells
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CD155/PVR
References
- Takai, Y. et al. (2003) Cancer Sci. 94:655.
- Mendelsohn, C.L. et al. (1989) Cell 56:855.
- Koike, H. et al. (1990) EMBO J. 9:3217.
- Koike, S. et al. (1991) Proc. Natl. Acad. Sci. USA 88:4104.
- Baury, B. et al. (2003) Biochem. Biophys. Res. Commun. 309:175.
- Mueller, S. and E. Wimmer (2003) J. Biol. Chem. 278:31251.
- Reymond, N. et al. (2004) J. Exp. Med. 199:1331.
- Lange, R. et al. (2001) Virology 285:218.
Long Name
Poliovirus Receptor
Alternate Names
CD155, HVED, Necl-5, PVR, PVS
Entrez Gene IDs
Gene Symbol
PVR
UniProt
Additional CD155/PVR Products
Product Documents for Human CD155/PVR Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CD155/PVR Antibody
For research use only
Citations for Human CD155/PVR Antibody
Customer Reviews for Human CD155/PVR Antibody
There are currently no reviews for this product. Be the first to review Human CD155/PVR Antibody and earn rewards!
Have you used Human CD155/PVR Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...