Human DC‑SIGN+DC‑SIGNR Antibody
R&D Systems | Catalog # MAB1621

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Primate - Macaca mulatta (Rhesus Macaque)
Applications
Validated:
Immunohistochemistry, Flow Cytometry, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Neutralization, Flow Cytometry, Immunocytochemistry, Functional Assay, Imaging
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2A Clone # 120612
Loading...
Product Specifications
Immunogen
NIH-3T3 mouse embryonic fibroblast cell line transfected with human DC-SIGNR
Accession # Q9H2X3
Accession # Q9H2X3
Specificity
Recognizes both human DC-SIGN and human DC-SIGNR on transfected cells. Does not react with parental mouse cells or irrelevant transfectants.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2A
Scientific Data Images for Human DC‑SIGN+DC‑SIGNR Antibody
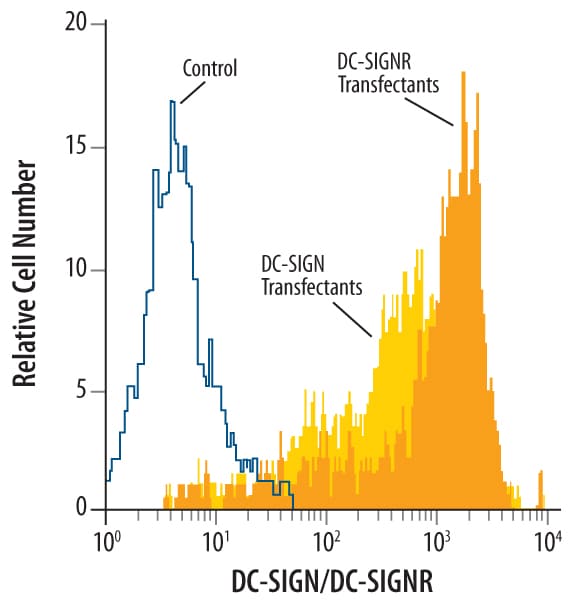
Detection of DC‑SIGN+DC‑SIGNR in Human DC‑SIGN or DC-SIGNR Transfected 3T3 Mouse Cell Line by Flow Cytometry.
Human DC-SIGN and DC-SIGNR transfected 3T3 mouse embryonic fibroblast cell line were stained with Mouse Anti-Human DC-SIGN+ DC-SIGNR Monoclonal Antibody (Catalog # MAB1621, filled histograms) or isotype control antibody (Catalog # MAB003, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG F(ab')2Secondary Antibody (Catalog # F0102B).Detection of DC‑SIGN+DC‑SIGNR in Human Monocyte Derived Dendritic Cells by Flow Cytometry.
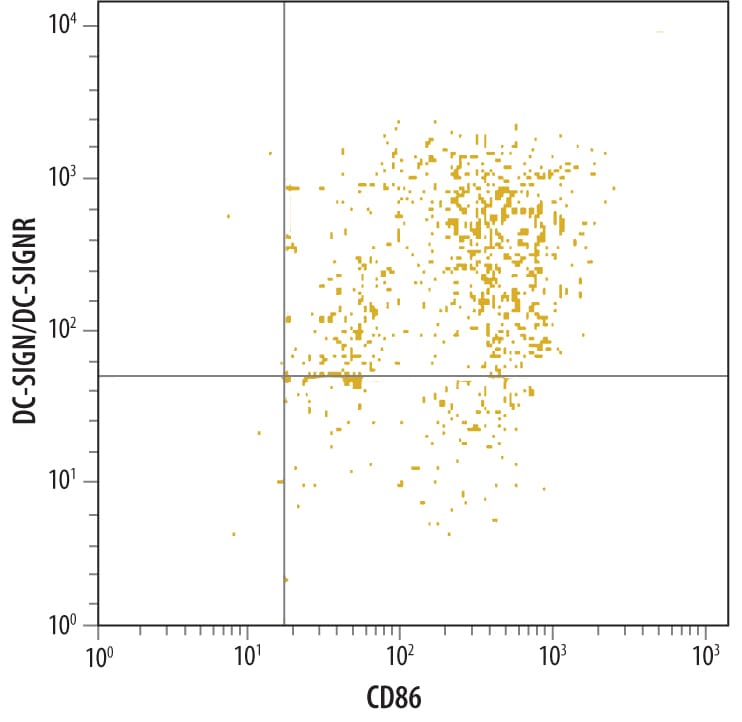
Human monocyte derived dendritic cells were stained with Mouse Anti-Human DC-SIGN+ DC-SIGNR Monoclonal Antibody (Catalog # MAB1621), followed by PE-conjugated anti-mouse secondary antibody (Catalog # F0102B) and Human B7-2/CD86 Fluorescein-conjugated Monoclonal Antibody (Catalog # FAB141F).Quadrant markers were set based on isotype control antibody staining (Catalog # MAB003).DC‑SIGN+DC‑SIGNR in Human Lymphoma.
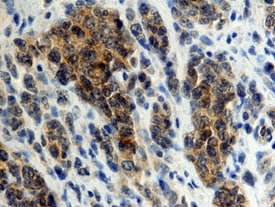
DC-SIGN+DC-SIGNR was detected in immersion fixed paraffin-embedded sections of human lymphoma using 25 µg/mL Mouse Anti-Human DC-SIGN+ DC-SIGNR Monoclonal Antibody (Catalog # MAB1621) overnight at 4 °C. Tissue was stained with the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counter-stained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human DC-SIGN+DC-SIGNR by Block/Neutralize
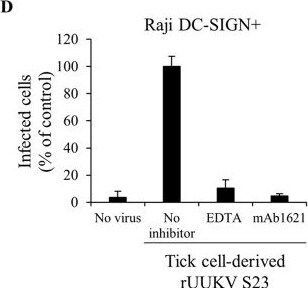
The C type lectin DC-SIGN enhances infection of human cells by tick cell-derived rUUKV S23. (A) BHK-21 cells were infected (at an MOI of 0.1) with rUUKV S23 derived from IRE/CTVM19 cells for 18 h and immunostained for N, GN, and GC proteins prior to flow cytometry analysis. (B) Parental (Raji) and DC-SIGN-expressing Raji cells (Raji DC-SIGN+) were infected with IRE/CTVM19 cell-derived rUUKV S23 and analyzed by flow cytometry 16 h after immunostaining against the viral nucleoprotein. (C) Parental (HeLa) and DC-SIGN-expressing HeLa cells (HeLa DC-SIGN+) were exposed to various MOIs of IRE/CTVM19 cell-derived rUUKV S23. The next day, infected cells were immunostained for the intracellular virus nucleoprotein N using the anti-N primary mouse monoclonal antibody 8B11A3 and an AF488-coupled anti-mouse secondary monoclonal antibody (green). Nuclei were stained with Hoechst (blue), and samples were analyzed by wide-field microscopy. (D) Raji DC-SIGN-expressing cells were exposed to IRE/CTVM19 cell-derived rUUKV S23 (MOI of ∼1) in the presence of inhibitors blocking DC-SIGN, namely, EDTA (5 mM) or the neutralizing mouse monoclonal antibody mAb1621 (25 μg · ml−1). Intracellular viral antigens were detected by immunostaining with an anti-UUKV rabbit polyclonal antibody, followed by incubation with AF647-conjugated secondary antibodies. Infection was analyzed by flow cytometry 18 h later and normalized to infection of DC-SIGN-expressing Raji cells in the absence of inhibitor (as a percentage of the control). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27194760), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human DC‑SIGN+DC‑SIGNR Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
2.5 µg/106 cells
Sample: Human DC‑SIGN or DC-SIGNR transfected 3T3 mouse embryonic fibroblast cell line, and human monocyte derived dendritic cells
Sample: Human DC‑SIGN or DC-SIGNR transfected 3T3 mouse embryonic fibroblast cell line, and human monocyte derived dendritic cells
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lymphoma
Sample: Immersion fixed paraffin-embedded sections of human lymphoma
Reviewed Applications
Read 1 review rated 5 using MAB1621 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: DC-SIGN+DC-SIGNR
References
- Geijtenbeek, T.B.H. et al. (2000) Cell 100:575.
- Geijtenbeek, T.B.H. et al. (2000) Cell 100:587.
- Yokoyama-Kobayashi, M.T. et al. (1999) Gene 228:161.
- Soilleux, E.J. et al. (2000) J. Immunol. 165:2937.
- Bashirova, A.A. et al. (2001) J. Exp. Med. 193:671.
- Mummidi, S. et al. (2001) J. Biol. Chem. 276:33196..
- Pohlman, S. et al. (2001) Proc. Natl. Acad. Sci. USA 98:2670.
- Geijtenbeek, T.B.H. et al. (2000) Nature Immunol. 1:353.
Long Name
Dendritic Cell-specific ICAM-3-grabbing Non-integrin
Alternate Names
DCSIGN+DCSIGNR
Entrez Gene IDs
30835 (Human)
Gene Symbol
CD209
UniProt
Additional DC-SIGN+DC-SIGNR Products
Product Documents for Human DC‑SIGN+DC‑SIGNR Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human DC‑SIGN+DC‑SIGNR Antibody
For research use only
Related Research Areas
Citations for Human DC‑SIGN+DC‑SIGNR Antibody
Customer Reviews for Human DC‑SIGN+DC‑SIGNR Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human DC‑SIGN+DC‑SIGNR Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
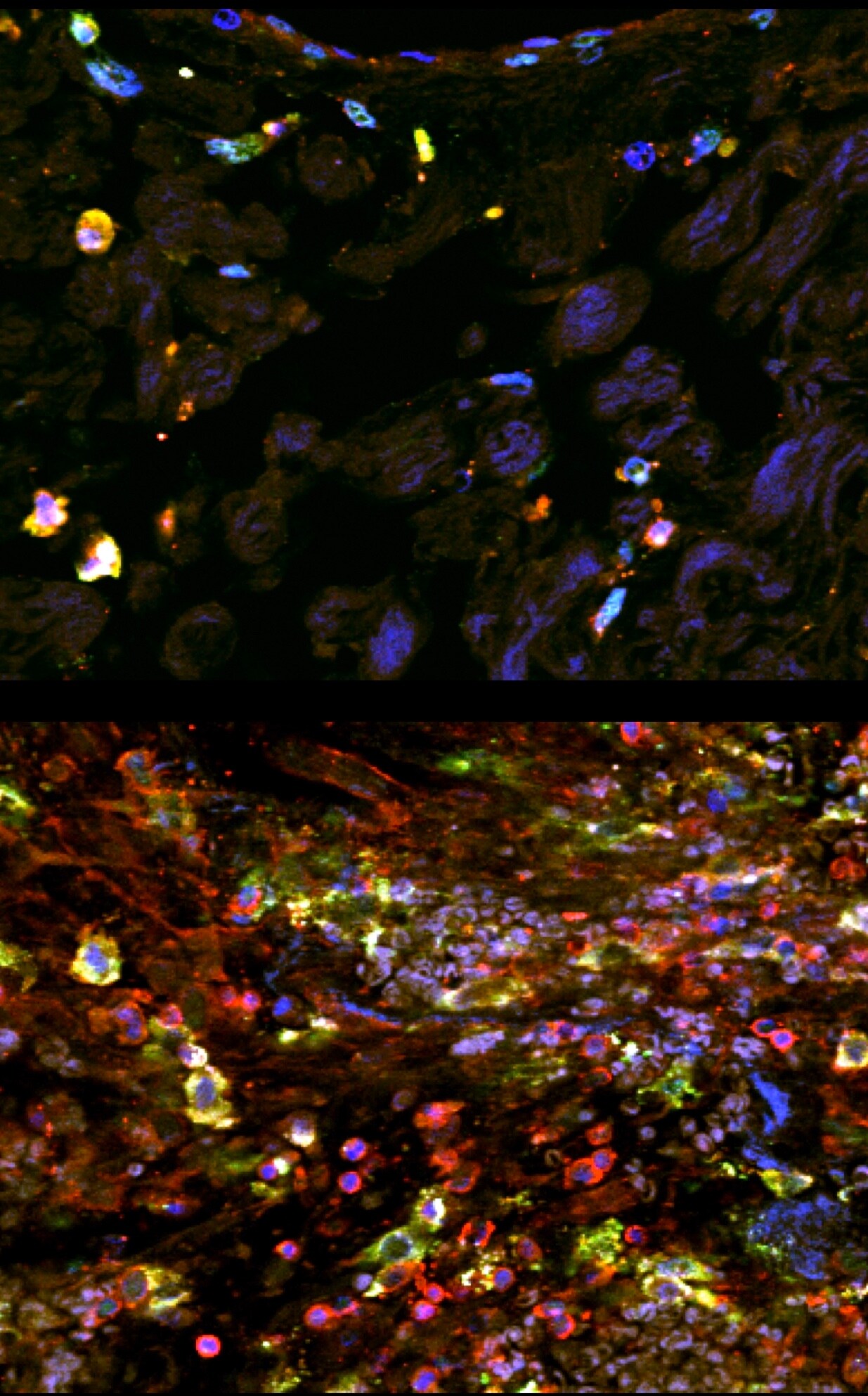
Application: ImmunohistochemistrySample Tested: Paraffin-embedded tissue and Paraffin-embeddedSpecies: HumanVerified Customer | Posted 12/10/2019Immunostaining of human paraffin-embedded tissue showing DC-sign (green) costained with CD45 to label immune cells (red) and Dapi to label nuclei (blue). Tissue underwent antigen retrieval with Tris-EDTA (pH8) and DC-sign was used at 1:100. Top panel is from normal tissue, bottom panel is after injury.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...