DDR2, also known as TYR010 and TKT, is a widely expressed 130 kDa type I transmembrane glycoprotein belonging to the discoidin-like domain containing subfamily of receptor tyrosine kinases (1). Mature human DDR2 consists of a 378 amino acid (aa) extracellular domain (ECD) that includes the discoidin-like domain, a 22 aa transmembrane segment, and a 434 aa cytoplasmic domain that includes the kinase domain (2). Within the ECD, human DDR2 shares 53% aa sequence identity with DDR1 and 97% aa sequence identity with mouse DDR2. The discoidin-like domain mediates DDR2 interactions with collagens I, III, and X (3‑5). Collagens II and V are less efficacious ligands (3). DDR2 selectively recognizes the triple helical structure of collagen compared to monomeric or denatured collagen (3, 5, 6). Within collagen II, the D2 period is required for DDR2 binding, and the D1 period is additionally required to trigger DDR2 autophosphorylation (6). The ECD of DDR2 exists as a non-covalent dimer in solution, and dimerization of the receptor greatly enhances collagen binding (4, 7). DDR2 interaction with collagen I inhibits collagen fibrillogenesis and alters collagen fiber morphology (7). Ligand binding induces DDR2 autophosphorylation in the cytoplasmic domain (3, 5, 8), which promotes associations with Shc and Src (9). In addition to the above mechanism, DDR2 exhibits a distinct interaction with collagen X. A region other than the discoidin-like domain of DDR2 recognizes the non-helical NC1 domain of collagen X, and this interaction does not lead to receptor autophosphorylation (5). Activation of DDR2 by collagen induces upregulation of MMP-1, -2, and -13 as well as DDR2 itself (3, 8, 10). DDR2 is implicated in collagenous matrix destruction and cell invasiveness (8, 10). DDR2 is also upregulated in several pathological conditions, including hepatic fibrosis following injury, rheumatoid and osteoarthritis, and smooth muscle cell hyperplasia (8, 10‑12).
Human DDR2 Antibody
R&D Systems | Catalog # MAB25381

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gln24-Arg399
Accession # Q16832
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human DDR2 Antibody
Detection of Human DDR2 in HEK293 Human Cell Line
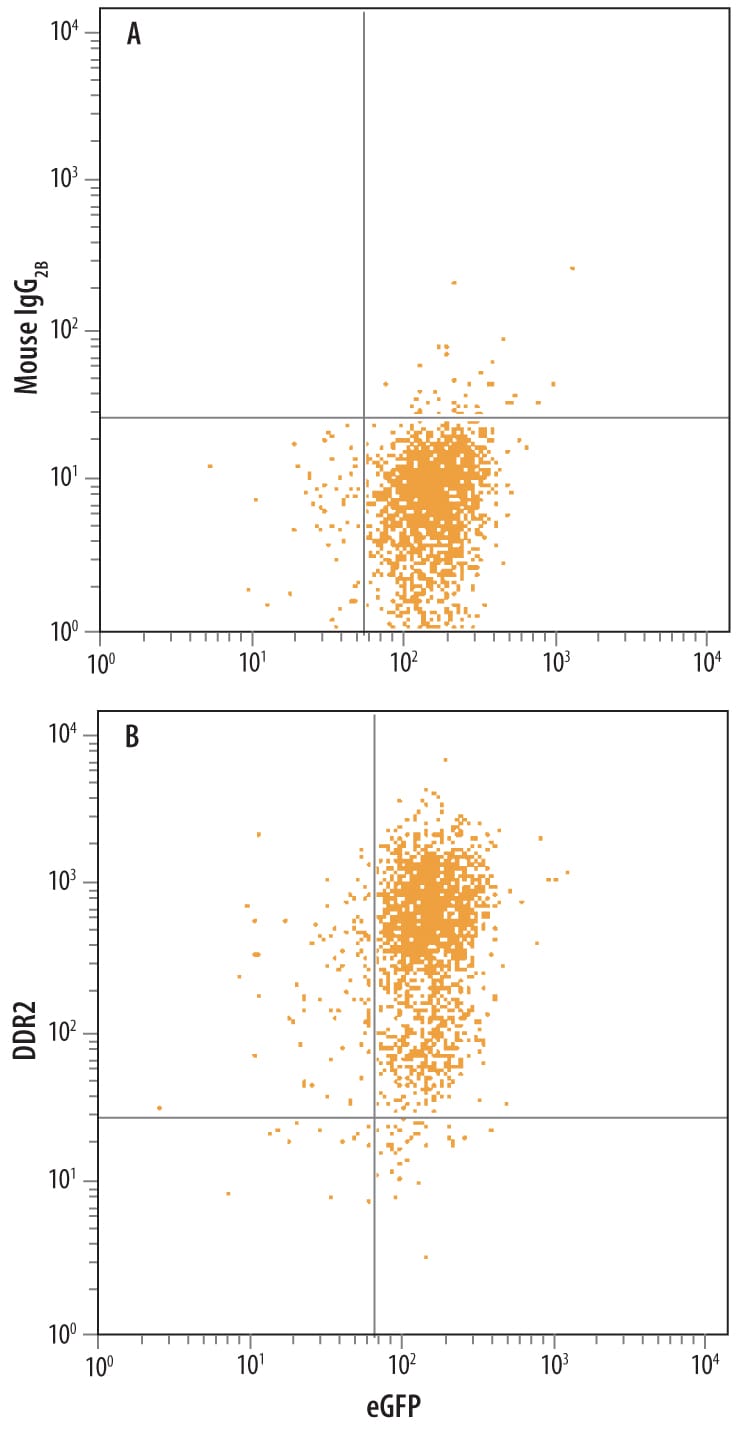
HEK293 human embryonic kidney cell line transfected with human DDR2 and Enhanced Green Fluorescent Protein (eGFP) was stained with either (A) Mouse IgG2BIsotype Control (Catalog # MAB00401) or (B) Mouse Anti-Human DDR2 Monoclonal Antibody (Catalog # MAB25381) followed by Allophycocyanin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0101B).DDR2 in Human Lung Cancer Tissue.
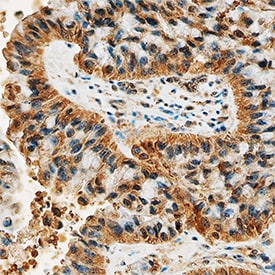
DDR2 was detected in immersion fixed paraffin-embedded sections of human lung cancer tissue using Mouse Anti-Human DDR2 Monoclonal Antibody (Catalog # MAB25381) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human DDR2 Antibody
CyTOF-reported
Flow Cytometry
Sample: HEK293 human embryonic kidney cell line transfected with human DDR2 and Enhanced Green Fluorescent Protein (eGFP)
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human lung cancer tissue
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: DDR2
References
- Vogel, W.F. et al. (2006) Cell. Signal. 18:1108.
- Karn, T. et al. (1993) Oncogene 8:3433.
- Vogel, W. et al. (1997) Mol. Cell 1:13.
- Leitinger, B. (2003) J. Biol. Chem. 278:16761.
- Leitinger, B. and A.P.L Kwan (2006) Matrix Biol. 25:355.
- Leitinger, B. et al. (2004) J. Mol. Biol. 344:993.
- Mihai, C. et al. (2006) J. Mol. Biol. 361:864.
- Olaso, E. et al. (2001) J. Clin. Invest. 108:1369.
- Ikeda, K. et al. (2002) J. Biol. Chem. 277:19206.
- Xu, L. et al. (2005) J. Biol. Chem. 280:548.
- Wang, J. et al. (2002) J. Autoimmun. 19:161.
- Ferri, N. et al. (2004) Am. J. Pathol. 164:1575.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional DDR2 Products
Product Documents for Human DDR2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human DDR2 Antibody
For research use only
Related Research Areas
Citations for Human DDR2 Antibody
Customer Reviews for Human DDR2 Antibody
There are currently no reviews for this product. Be the first to review Human DDR2 Antibody and earn rewards!
Have you used Human DDR2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways