Human GFI‑1 Antibody
R&D Systems | Catalog # AF3540

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Pro2-Leu250
Accession # Q99684
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human GFI‑1 Antibody
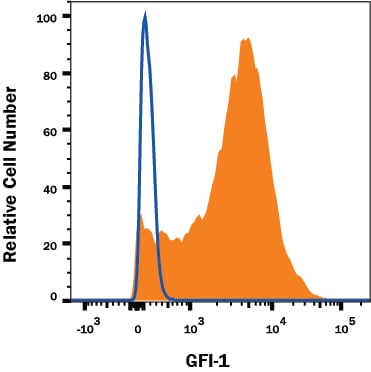
Detection of GFI-1 in THP-1 Human Cell Line by Flow Cytometry.
THP-1 monocytic leukemia cell line was stained with Goat Anti-Human GFI-1SOX2 Affinity Purified Polyclonal Antibody (Catalog # AF3540, filled histogram) or control antibody (Catalog # AB-108-C, open histogram), followed by APC-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0108). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005). View our protocol for Staining Intracellular Molecules.Detection of Human GFI-1 by Western Blot
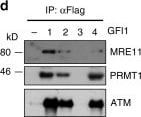
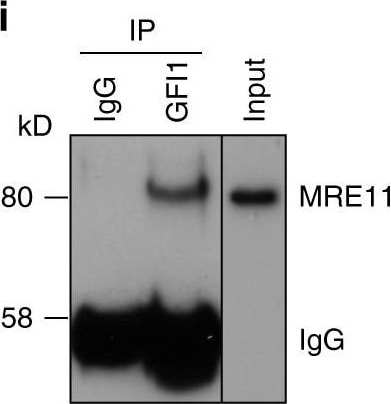
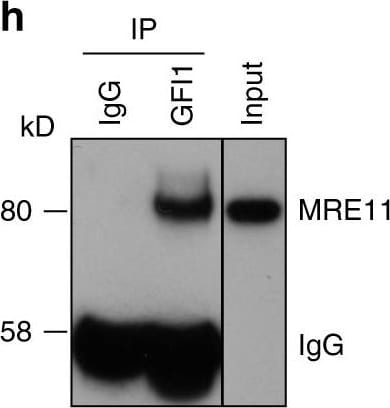
GFI1 interacts with proteins involved in the DDR. a GFI1-Flag fusion protein was immunoprecipitated from 293T cells. Co-precipitated proteins were run on polyacrylamide gel and stained with Coomassie blue. b Peptide sequence of MRE11 with peptides identified through mass spectrometry highlighted and corresponding spectra below. c Peptide sequence of PRMT1 with peptides identified through mass spectrometry highlighted and corresponding spectra below. d Variants of the GFI1-Flag fusion protein were immunoprecipitated from 293T cells. Extracts were separated by SDS–PAGE and blotted for the indicated proteins. e Diagram of the different GFI1 fusion proteins used in d and g. f GFI1-Flag fusion protein was immunoprecipitated in 293T cells in the presence or absence of benzonase. Extracts were separated by SDS–PAGE and blotted for MRE11. g GFI1 KO Jurkat cells were electroporated with plasmids expressing GFI1 variant constructs as shown in e. GFI1 KO and parental control Jurkat cells were used as controls. After 24 h, cells were exposed to 5 Gy IR and allowed to recover for the indicated time. Cells were lysed and analyzed by alkaline Comet assay. Comet tail moment averages are shown. One of three replicate experiments is shown. Error bars represent s.d. h Endogenous GFI1 protein was immunoprecipitated in SupT1 cells. Co-precipitated proteins were separated by SDS–PAGE and blotted for MRE11 and PRMT1. i Endogenous GFI1 protein was immunoprecipitated in Jurkat cells. Co-precipitated proteins were separated by SDS–PAGE and blotted for MRE11 and PRMT1 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/s41467-018-03817-5), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GFI-1 by Western Blot
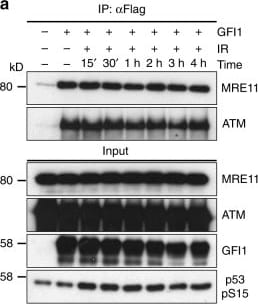
GFI1 activities are independent of DNA damage. a GFI1-Flag fusion protein was immunoprecipitated in 293T cells treated with 5 Gy IR and allowed to recover for the indicated amount of time. Extracts were separated by SDS–PAGE and blotted for the indicated proteins. b SupT1 cells were spread on glass slides 15 min and 1 h after irradiation using a Cytospin, stained for endogenous Gfi1 and gamma -H2AX and visualized for immunofluorescence by confocal microscopy. Control cells stained without primary antibody but with secondary antibodies are shown. c U2OS cells carrying a LacO array and expressing a LacR-Fok1-mCherry endonuclease were transfected with a vector expressing the GFI1-GFP fusion protein. These cells were plated on cover glass, stained for gamma -H2AX and visualized for immunofluorescence by confocal microscopy. d U2OS cells expressing a GFI1-GFP fusion protein were exposed to 405 nm UV micro-irradiation and the recruitment of the GFI1-GFP fusion protein to the site of damage was quantified by confocal microscopy. Average signal intensity is shown with error bars representing s.d. Recruitment of Ku80-mRuby2 fusion protein and GFP protein are shown as controls. Representative images of selected time points are shown on the right. Scale bar represents 10 μm Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/s41467-018-03817-5), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human GFI-1 Antibody by Immunoprecipitation
GFI1 interacts with proteins involved in the DDR. a GFI1-Flag fusion protein was immunoprecipitated from 293T cells. Co-precipitated proteins were run on polyacrylamide gel and stained with Coomassie blue. b Peptide sequence of MRE11 with peptides identified through mass spectrometry highlighted and corresponding spectra below. c Peptide sequence of PRMT1 with peptides identified through mass spectrometry highlighted and corresponding spectra below. d Variants of the GFI1-Flag fusion protein were immunoprecipitated from 293T cells. Extracts were separated by SDS–PAGE and blotted for the indicated proteins. e Diagram of the different GFI1 fusion proteins used in d and g. f GFI1-Flag fusion protein was immunoprecipitated in 293T cells in the presence or absence of benzonase. Extracts were separated by SDS–PAGE and blotted for MRE11. g GFI1 KO Jurkat cells were electroporated with plasmids expressing GFI1 variant constructs as shown in e. GFI1 KO and parental control Jurkat cells were used as controls. After 24 h, cells were exposed to 5 Gy IR and allowed to recover for the indicated time. Cells were lysed and analyzed by alkaline Comet assay. Comet tail moment averages are shown. One of three replicate experiments is shown. Error bars represent s.d. h Endogenous GFI1 protein was immunoprecipitated in SupT1 cells. Co-precipitated proteins were separated by SDS–PAGE and blotted for MRE11 and PRMT1. i Endogenous GFI1 protein was immunoprecipitated in Jurkat cells. Co-precipitated proteins were separated by SDS–PAGE and blotted for MRE11 and PRMT1 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29651020), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human GFI-1 Antibody by Immunoprecipitation
GFI1 interacts with proteins involved in the DDR. a GFI1-Flag fusion protein was immunoprecipitated from 293T cells. Co-precipitated proteins were run on polyacrylamide gel and stained with Coomassie blue. b Peptide sequence of MRE11 with peptides identified through mass spectrometry highlighted and corresponding spectra below. c Peptide sequence of PRMT1 with peptides identified through mass spectrometry highlighted and corresponding spectra below. d Variants of the GFI1-Flag fusion protein were immunoprecipitated from 293T cells. Extracts were separated by SDS–PAGE and blotted for the indicated proteins. e Diagram of the different GFI1 fusion proteins used in d and g. f GFI1-Flag fusion protein was immunoprecipitated in 293T cells in the presence or absence of benzonase. Extracts were separated by SDS–PAGE and blotted for MRE11. g GFI1 KO Jurkat cells were electroporated with plasmids expressing GFI1 variant constructs as shown in e. GFI1 KO and parental control Jurkat cells were used as controls. After 24 h, cells were exposed to 5 Gy IR and allowed to recover for the indicated time. Cells were lysed and analyzed by alkaline Comet assay. Comet tail moment averages are shown. One of three replicate experiments is shown. Error bars represent s.d. h Endogenous GFI1 protein was immunoprecipitated in SupT1 cells. Co-precipitated proteins were separated by SDS–PAGE and blotted for MRE11 and PRMT1. i Endogenous GFI1 protein was immunoprecipitated in Jurkat cells. Co-precipitated proteins were separated by SDS–PAGE and blotted for MRE11 and PRMT1 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29651020), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human GFI‑1 Antibody
CyTOF-ready
Intracellular Staining by Flow Cytometry
Sample: THP-1 Human Cell Line fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005)
Western Blot
Sample: Recombinant Human GFI‑1
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: GFI-1
Long Name
Alternate Names
Gene Symbol
UniProt
Additional GFI-1 Products
Product Documents for Human GFI‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human GFI‑1 Antibody
For research use only
Citations for Human GFI‑1 Antibody
Customer Reviews for Human GFI‑1 Antibody
There are currently no reviews for this product. Be the first to review Human GFI‑1 Antibody and earn rewards!
Have you used Human GFI‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 Th2 Differentiation Pathway
Th2 Differentiation Pathway