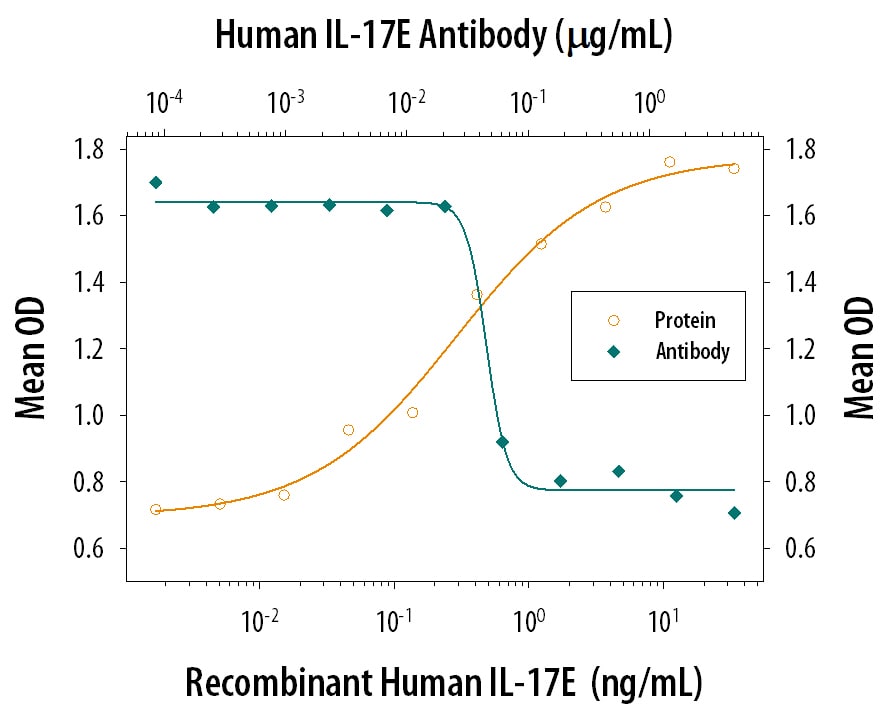
The Interleukin 17 (IL-17) family proteins, comprising six members (IL-17, IL-17B through IL-17F), are secreted, structurally related proteins that share a conserved cysteine-knot fold near the C-terminus, but have considerable sequence divergence at the N-terminus. With the exception of IL-17B, which exists as a non-covalently linked dimer, all IL-17 family members are disulfide-linked dimers. IL-17 family proteins are pro-inflammatory cytokines that induce local cytokine production and are involved in the regulation of immune functions (1, 2).
Human IL-17E cDNA encodes a 177 amino acid (aa) residues precursor protein with a putative 32 aa signal peptide (3). A second isoform of human IL-17E encoding a 161 aa precursor protein also exists (4). The two isoforms differ in their signal peptide sequences. Mature human IL-17E shares 76% aa sequence identity with mature mouse IL-17E. Human IL-17E also shares from 25% to 36% aa sequence identity with the other human IL-17 family members. IL-17E expression was detected at very low levels by PCR in various peripheral tissues including brain, kidney, lung, prostate, testis, adrenal gland spinal cord and trachea (3). IL-17E binds and activates IL‑17 B Receptor (IL-17B R) (alternatively known as IL-17 Rh1, IL-17E R, and EVI27) (3), which is expressed in kidney and liver, and at lower levels in brain, testis and other endocrine tissues. The expression of IL-17B R is up regulated under inflammatory conditions. Ligation of IL-17E to IL-17 RB induces activation of nuclear factor kappa-B and stimulates the production of the proinflamatory cytokine IL-8 (3). IL-17 has also been found to promote the expression of the prototypical Th2 genes (4, 5).