Klotho beta, a divergent structural member of the glycosidase I superfamily, is expressed primarily in the liver and pancreas, with lower expression in adipose tissue (1, 2). Like Klotho, Klotho beta facilitates binding between FGF19 subfamily members and their receptors via formation of a ternary complex (3). The Klotho beta mediated interaction of human FGF19 (mouse FGF15) with FGF Receptor 4 in the liver negatively regulates bile acid synthesis by controlling the secretion of two key bile acid synthase genes, cholesterol 7-alpha hydroxylase (Cyp7a1) and sterol 12-alpha hydroxylase (Cyp8b1) (2-5). Klotho beta is also a cofactor for the interaction of FGF21 with FGF Receptor 1c in adipocytes, which allows FGF21 to stimulate GLUT1 expression, upregulating adipocyte insulin-dependent glucose uptake (2-4, 6). The 1043 amino acid (aa) type I transmembrane protein is composed of a 51 aa signal sequence, a 943 aa extracellular domain (ECD) containing two glycosidase-like regions, a 21 aa transmembrane domain, and 28 aa intracellular tail. Since Klotho-related proteins lack critical active site Glu residues present in beta -glycosidases, it was initially unclear whether they were functional enzymes (1, 7). However, glucuronidase activity has since been demonstrated for Klotho, indicating that physiologically relevant enzymatic activity for Klotho beta is also possible (8). The extracellular domain shares 79%, 87%, 87% and 67% identity with mouse, equine, canine and rat Klotho beta, respectively. The low identity with rat reflects aa discordance within rodent ECD.
Human Klotho beta Antibody
R&D Systems | Catalog # AF5889


Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Immunocytochemistry
Cited:
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Klotho beta
Phe53-Leu997
Accession # Q86Z14
Phe53-Leu997
Accession # Q86Z14
Specificity
Detects human Klotho beta in direct ELISAs and Western blots. In direct ELISAs, approximately 35% cross-reactivity with recombinant mouse Klotho beta is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Klotho beta Antibody
Detection of Human Klotho beta by Western Blot.
Western blot shows lysates of Hep3B human hepatocellular carcinoma cell line. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Human Klotho beta Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5889) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for Klotho beta at approximately 130 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Klotho beta in HepG2 Human Cell Line.
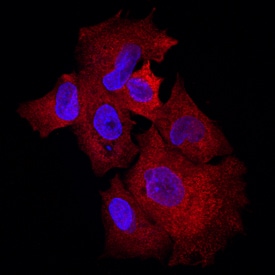
Klotho beta was detected in immersion fixed HepG2 human hepatocellular carcinoma cell line using Goat Anti-Human Klotho beta Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5889) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Klotho beta by Immunocytochemistry/ Immunofluorescence
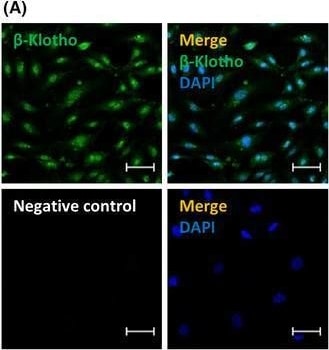
Effects of FGF21 on beta ‐klotho expression and oxidative stress in EPCs. After FGF21 treatment, cells were (A) stained with an beta ‐klotho antibody, and (B) protein expression was analysed by Western blotting. (C) The effects of FGF21 on EPC viability were analysed by the CCK‐8 assay. (D) EPCs were treated with the indicated concentration of FGF21 for 12 h and 600 µM H2O2 for 1 h. Intracellular ROS levels were measured using a Fluorometric Intracellular ROS Kit. The results are expressed as the mean ± SEM of five separate experiments run in triplicate (*p < 0.01 vs. control, #p < 0.01 vs. H2O2 only) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35307922), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Klotho beta Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed HepG2 human hepatocellular carcinoma cell line
Sample: Immersion fixed HepG2 human hepatocellular carcinoma cell line
Western Blot
0.5 µg/mL
Sample: Hep3B human hepatocellular carcinoma cell line
Sample: Hep3B human hepatocellular carcinoma cell line
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Klotho beta
References
- Mian, I. S. (1998) Blood Cells Mol. Dis. 24:83.
- Kurosu, H. and M. Kuro-o (2009) Mol. Cell. Endocrinol. 299:72.
- Ito, S. et al. (2005) J. Clin. Invest. 115:2202.
- Kurosu, H. et al. (2007) J. Biol. Chem. 282:26687.
- Lin, B. C. et al. (2007) J. Biol. Chem. 282:27277.
- Ogawa, Y. et al. (2007) Proc. Natl. Acad. Sci USA 104:7432.
- Chang, Q. et al. (2005) Science 310:490.
- Goetz, R. et al. (2007) Mol. Cell. Biol. 27:3417.
Alternate Names
KLB
Gene Symbol
KLB
UniProt
Additional Klotho beta Products
Product Documents for Human Klotho beta Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Klotho beta Antibody
For research use only
Related Research Areas
Citations for Human Klotho beta Antibody
Customer Reviews for Human Klotho beta Antibody
There are currently no reviews for this product. Be the first to review Human Klotho beta Antibody and earn rewards!
Have you used Human Klotho beta Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...