Lectin-like oxidized low-density-lipoprotein receptor-1 (LOX-1), also known as oxidized low-density-lipoprotein receptor-1 (OLR-1), is a type II transmembrane receptor belonging to the C-type lectin family (1). It also belongs to the functionally defined scavenger receptor (SR) superfamily, whose members share the common ability to bind and internalize modified forms of Low Density Lipoproteins (LDL) (2 - 4). LOX-1 is the first member of the class E scavenger receptor subfamily (SR-E). It binds and supports the internalization of multiple structurally unrelated macromolecules including oxidized LDL, advanced glycation end products (AGE), activated platelets, bacteria, apoptotic or aged cells, and heat shock proteins (5 - 7). LOX-1 has also been implicated as an intestinal receptor involved in the transcytosis of pancreatic bile salt-dependent lipase (8). The human LOX-1 gene encodes a 273 amino acid (aa) residue protein with a short N-terminal intracellular domain, a transmembrane domain, an extracellular stalk/neck region followed by a C-type lectin-like domain (CTLD) (1, 6). The CTLD, which is required for ligand recognition, contains the six conserved cysteine residues present in all C-type lectins, but lacks the Ca2+-binding residues found in classical C-type lectins. LOX-1 can be detected on activated endothelial cells, vascular smooth muscle cells, macrophages, intestinal cells and dendritic cells (6 - 8). The expression of LOX-1 is induced by proinflammatory or proatherogenic stimuli, as well as by oxidized LDL itself and hemodynamic or oxidative stress. Human LOX-1 exists on the cell surface as covalent homodimers, which can further associate into non-covalent-linked oligomers (9). Cell surface LOX-1 can also be cleaved by yet unidentified proteases to release the soluble LOX-1 extracellular domain (6). Binding and endocytosis of oxidized LDL by LOX-1 induces oxidative stress, activates NF kappa B, and upregulates the expression of monocyte chemoattractant protein-1 and matrix metalloproteases (5 - 9). LOX-1-dependent oxidized LDL uptake also induces apoptosis by inducing the expression of the pro-apoptotic Bax and downregulation of the anti-apoptotic Bcl-2 (10). Oxidized LDL plays a key role in the pathogenesis of atherosclerosis and endothelial dysfunction. Blockade of LOX-1 functions may turn out to be a suitable target for the therapeutic intervention of atherosclerosis.

Key Product Details
Validated by
Knockout/Knockdown
Species Reactivity
Validated:
Human
Cited:
Human, Porcine
Applications
Validated:
Immunohistochemistry, Western Blot, Blockade of Receptor-ligand Interaction
Cited:
Western Blot, Neutralization, Immunocytochemistry, ELISA Detection
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human LOX-1 (R&D Systems, Catalog # 1798-LX)
Ser61-Gln273
Accession # P78380
Ser61-Gln273
Accession # P78380
Specificity
Selected for its ability to block receptor-ligand interaction in a functional ELISA assay. In direct ELISAs, less than 5% cross‑reactivity with recombinant mouse LOX-1 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human LOX‑1/OLR1 Antibody
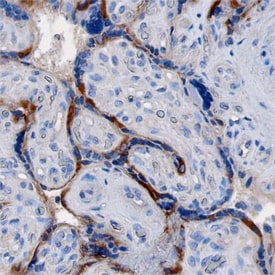
LOX‑1/OLR1 in Human Placenta.
LOX-1/OLR1 was detected in immersion fixed paraffin-embedded sections of human placenta using Goat Anti-Human LOX-1/OLR1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1798) at 1 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to cytotrophoblasts. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human LOX-1/OLR1 by Immunocytochemistry/Immunofluorescence
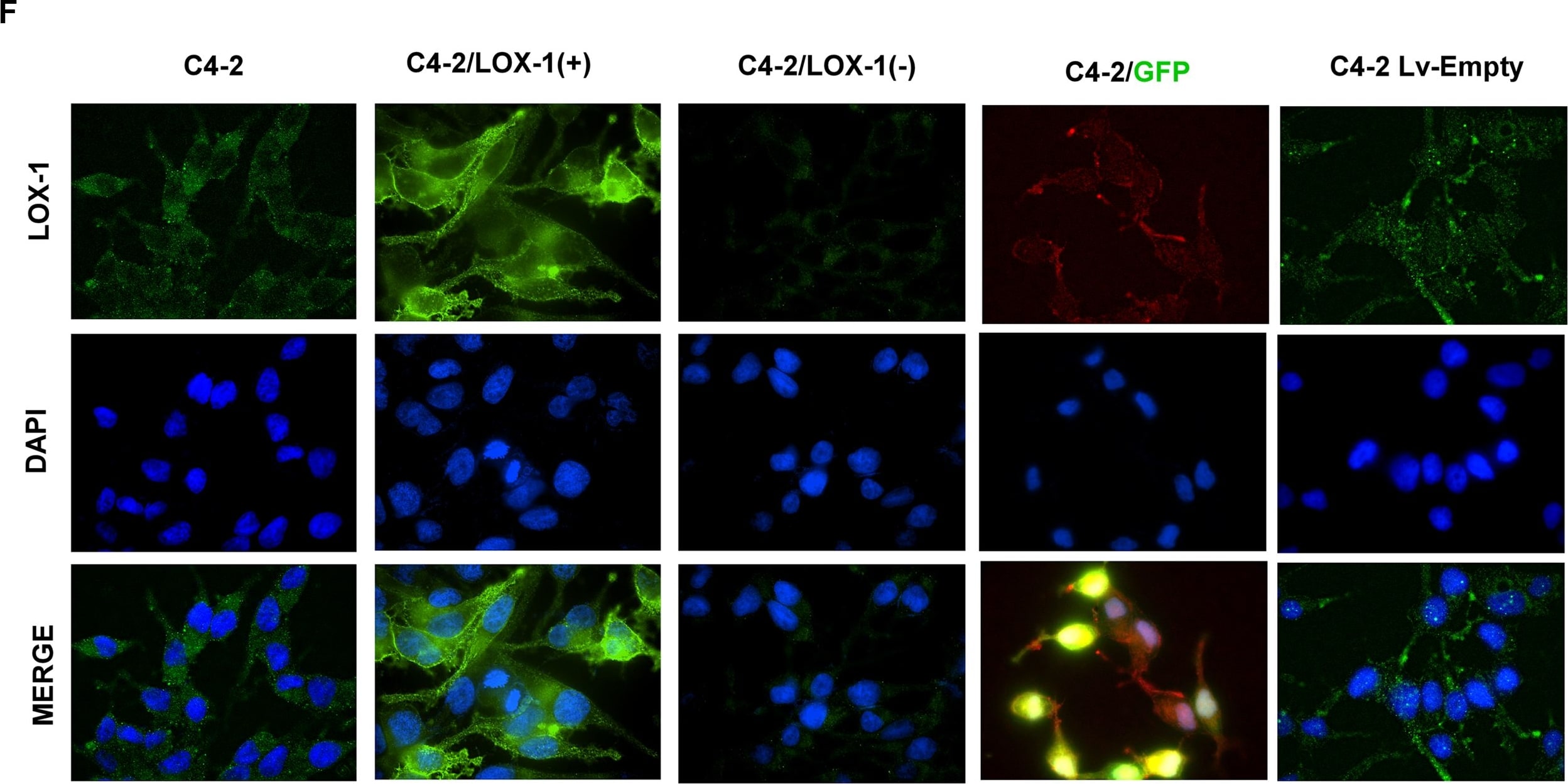
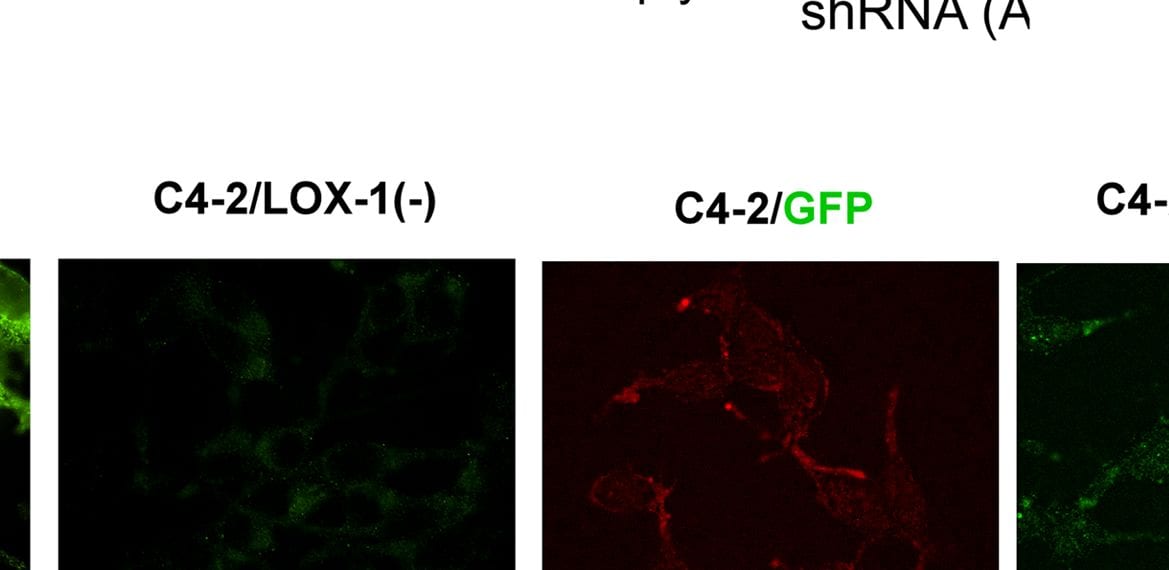
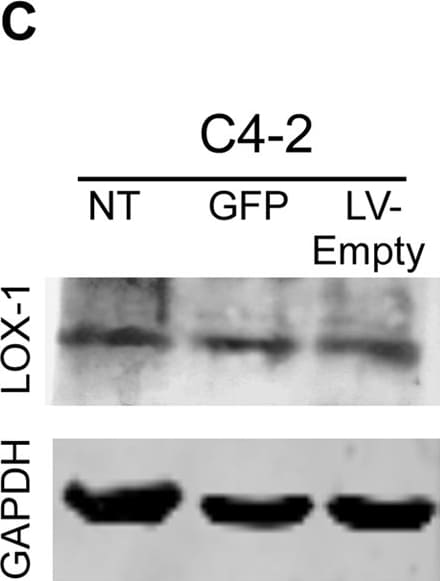
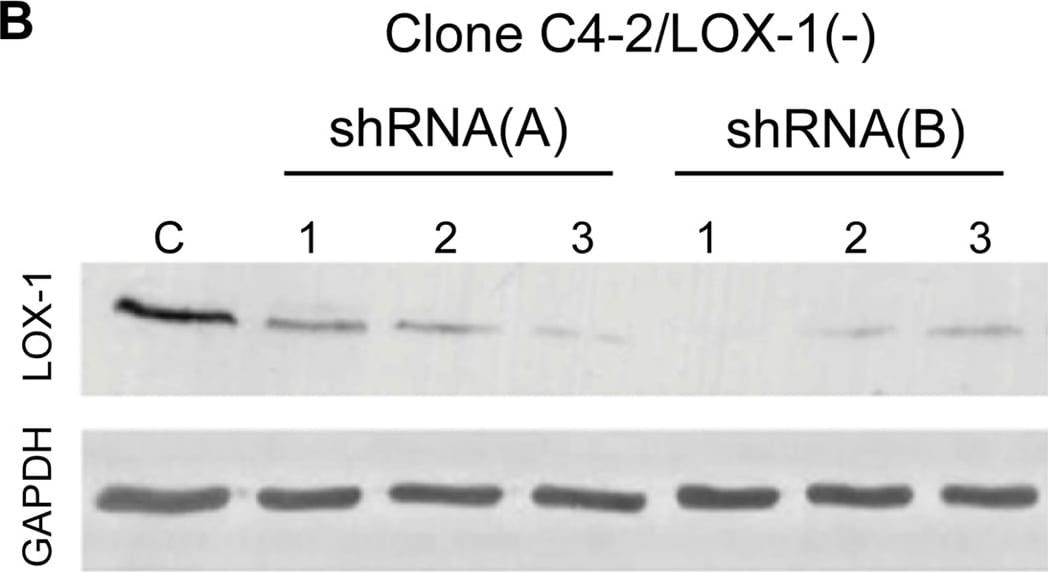
Generation of stable prostate cancer cell lines with LOX-1 over-expression and shRNA against olr1.A) Western blot for LOX-1 (40 kDa) expression in human CaP clones with overexpression of LOX-1. B) Western blot for LOX-1 (40 kDa) expression in human prostate cancer cell clones with LOX-1 knockdown C) Real-time PCR for LOX-1 expression in three clones with overexpression of LOX-1. D) Real-time PCR for LOX-1 expression was determined in three clones that express shRNA/LOX-1(A), and three clones that express shRNA/LOX-1(B). The data represent the means ± S.D. of three independent experiments performed in triplicate, and statistically analyzed using one-way analysis of variance and Dunnett’s post-test; (***p≤0.001, **p≤0.01, *p≤0.05). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25170920), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human LOX-1/OLR1 by Western Blot
Generation of stable prostate cancer cell lines with LOX-1 over-expression and shRNA against olr1.A) Western blot for LOX-1 (40 kDa) expression in human CaP clones with overexpression of LOX-1. B) Western blot for LOX-1 (40 kDa) expression in human prostate cancer cell clones with LOX-1 knockdown C) Real-time PCR for LOX-1 expression in three clones with overexpression of LOX-1. D) Real-time PCR for LOX-1 expression was determined in three clones that express shRNA/LOX-1(A), and three clones that express shRNA/LOX-1(B). The data represent the means ± S.D. of three independent experiments performed in triplicate, and statistically analyzed using one-way analysis of variance and Dunnett’s post-test; (***p≤0.001, **p≤0.01, *p≤0.05). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25170920), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human LOX-1/OLR1 by Western Blot
Generation of stable prostate cancer cell lines with LOX-1 over-expression and shRNA against olr1.A) Western blot for LOX-1 (40 kDa) expression in human CaP clones with overexpression of LOX-1. B) Western blot for LOX-1 (40 kDa) expression in human prostate cancer cell clones with LOX-1 knockdown C) Real-time PCR for LOX-1 expression in three clones with overexpression of LOX-1. D) Real-time PCR for LOX-1 expression was determined in three clones that express shRNA/LOX-1(A), and three clones that express shRNA/LOX-1(B). The data represent the means ± S.D. of three independent experiments performed in triplicate, and statistically analyzed using one-way analysis of variance and Dunnett’s post-test; (***p≤0.001, **p≤0.01, *p≤0.05). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25170920), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human LOX-1/OLR1 by Knockdown Validated
Generation of stable prostate cancer cell lines with LOX-1 over-expression and shRNA against olr1.A) Western blot for LOX-1 (40 kDa) expression in human CaP clones with overexpression of LOX-1. B) Western blot for LOX-1 (40 kDa) expression in human prostate cancer cell clones with LOX-1 knockdown C) Real-time PCR for LOX-1 expression in three clones with overexpression of LOX-1. D) Real-time PCR for LOX-1 expression was determined in three clones that express shRNA/LOX-1(A), and three clones that express shRNA/LOX-1(B). The data represent the means ± S.D. of three independent experiments performed in triplicate, and statistically analyzed using one-way analysis of variance and Dunnett’s post-test; (***p≤0.001, **p≤0.01, *p≤0.05). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25170920), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human LOX‑1/OLR1 Antibody
Application
Recommended Usage
Blockade of Receptor-ligand Interaction
In a functional ELISA, 0.5 - 2 μg/mL of this antibody will block 50% of the binding of 1 μg/mL of biotinylated AGE-BSA to immobilized recombinant human LOX-1 coated at 5 μg/mL (100 µL/well). At 10 μg/mL, this antibody will block >95% of the binding.
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human placenta
Sample: Immersion fixed paraffin-embedded sections of human placenta
Western Blot
0.1 µg/mL
Sample: Recombinant Human LOX‑1/OLR1 (Catalog # 1798-LX)
Sample: Recombinant Human LOX‑1/OLR1 (Catalog # 1798-LX)
Reviewed Applications
Read 3 reviews rated 4 using AF1798 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: LOX-1/OLR1
References
- Sawamura, T. et al. (1997) Nature 386:73.
- Daugherty, A. et al. (2000) Curr. Opin. Cardiovasc. Pulm. Ren. Invest. Drugs. 2:223.
- Platt, N. and S. Gordon (2001) J. Clin. Invest. 108:649.
- Platt, N. and S. Gordon (1998) Chem. Biol. 5:R193.
- Jono, T. et al. (2002) FEBS Lett. 511:170.
- Kume, N. et al. (2001) Curr. Opin. Lipidol. 12:419.
- Delneste, Y. et al. (2002) Immunity 17:353.
- Bruneau, N. et al. (2003) Mol. Biol. Cell 14:2861.
- Xie, Q. et al. (2004) DNA and Cell Biol. 23:111.
- Chen, J. et al. (2003) Circ. Res. 94:370.
Long Name
Lectin-like Oxidized Low-density Lipoprotein Receptor 1
Alternate Names
CLEC8A, LOX1, OLR1, SCARE1, SR-E1
Gene Symbol
OLR1
UniProt
Additional LOX-1/OLR1 Products
Product Documents for Human LOX‑1/OLR1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human LOX‑1/OLR1 Antibody
For research use only
Related Research Areas
Citations for Human LOX‑1/OLR1 Antibody
Customer Reviews for Human LOX‑1/OLR1 Antibody (3)
4 out of 5
3 Customer Ratings
Have you used Human LOX‑1/OLR1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: ELISASample Tested: Recombinant protein and Human recombinant antibodySpecies: HumanVerified Customer | Posted 07/09/2018
-
Application: Western BlotSample Tested: CHO Chinese hamster ovary cell line and Recombinant proteinSpecies: HumanVerified Customer | Posted 07/09/2018
-
Application: Western BlotSample Tested: See PMID 23950953Species: OtherVerified Customer | Posted 01/05/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...