The human Macrophage Mannose Receptor (MMR), also known as CD206 and MRC1 (mannose receptor C, type 1), is a 190 kDa scavenger receptor that is expressed on tissue macrophages, myeloid dendritic cells, and liver and lymphatic endothelial cells (1). It belongs to a family of receptors sharing similar protein structure that also includes DEC205, phospholipase A2 receptor, and Endo180 (2, 3). The human MMR protein is synthesized as a 1456 amino acid (aa) precursor that contains an 18 aa signal sequence, a 1371 aa extracellular region, a 21 aa transmembrane segment and a 46 aa cytoplasmic domain (4). Its extracellular region is composed of an N‑terminal cysteine-rich domain, followed by a single fibronectin type II repeat, and eight C-type lectin carbohydrate recognition domains (CRD) (3, 4). Human to mouse, the extracellular region is 82% aa identical. The cysteine-rich domain mediates recognition of sulfated N‑acetylgalactosamine, which occurs on some extracellular matrix proteins and is the terminal sugar of the unusual oligosaccharides present on pituitary hormones such as lutropin and thyrotropin (5). Several of the CRDs participate in the Ca2+-dependent recognition of carbohydrates showing a preference for branched sugars with terminal mannose, fucose or N‑acetylglucosamine (6). The cytoplasmic domain of MMR includes a tyrosine-based motif for internalization in clathrin-coated vesicles. Once internalized, ligands are released following acidification of phagosomes or endosomes, and the receptor is recycled to the cell surface (3, 7). MMR mediates phagocytosis upon binding to target structures that occur on a variety of pathogenic microorganisms including Gram-negative and Gram-positive bacteria, yeasts, parasites, and mycobacteria. MMR also functions to maintain homeostasis through the endocytosis of potentially harmful glycoproteins associated with inflammation (2, 3).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Primate - Macaca mulatta (Rhesus Macaque), Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry, Simple Western
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Bioassay, ELISA Capture, Functional Assay
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human MMR/CD206
Leu19-Lys1383 (Thr399Ala) & (Leu407Phe)
Accession # P22897
Leu19-Lys1383 (Thr399Ala) & (Leu407Phe)
Accession # P22897
Specificity
Detects human MMR/CD206 in direct ELISAs and Western blots. In direct ELISAs, approximately 20% cross-reactivity with recombinant mouse MMR is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human MMR/CD206 Antibody
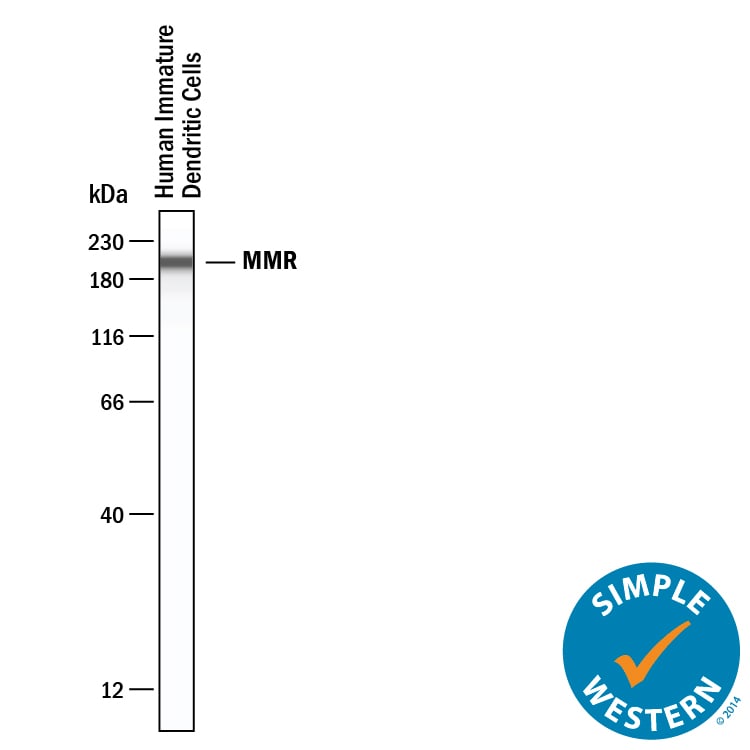
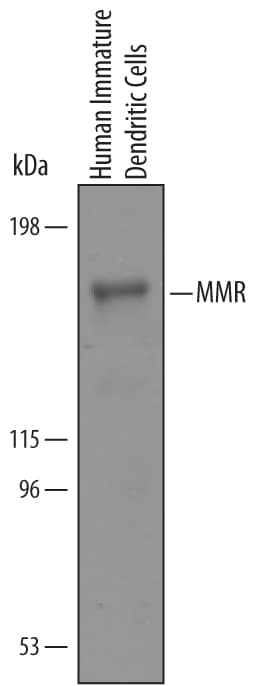
Detection of Human MMR/CD206 by Western Blot.
Western blot shows lysates of human immature dendritic cells. PVDF Membrane was probed with 1 µg/mL of Goat Anti-Human MMR/CD206 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2534) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for MMR/CD206 at approximately 185 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.MMR/CD206 in Human Liver.
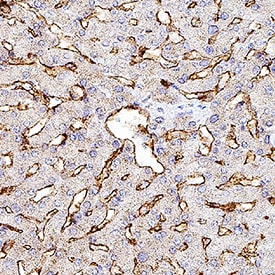
MMR/CD206 was detected in immersion fixed paraffin-embedded sections of human liver using Goat Anti-Human MMR/CD206 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2534) at 3 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to sinusoids. Staining was performed using our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Human MMR/CD206 by Simple WesternTM.
Simple Western lane view shows lysates of human immature dendritic cells, loaded at 0.2 mg/mL. A specific band was detected for MMR/CD206 at approximately 201 kDa (as indicated) using 10 µg/mL of Goat Anti-Human MMR/CD206 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2534). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Human MMR/CD206/Mannose Receptor by Immunohistochemistry-Paraffin
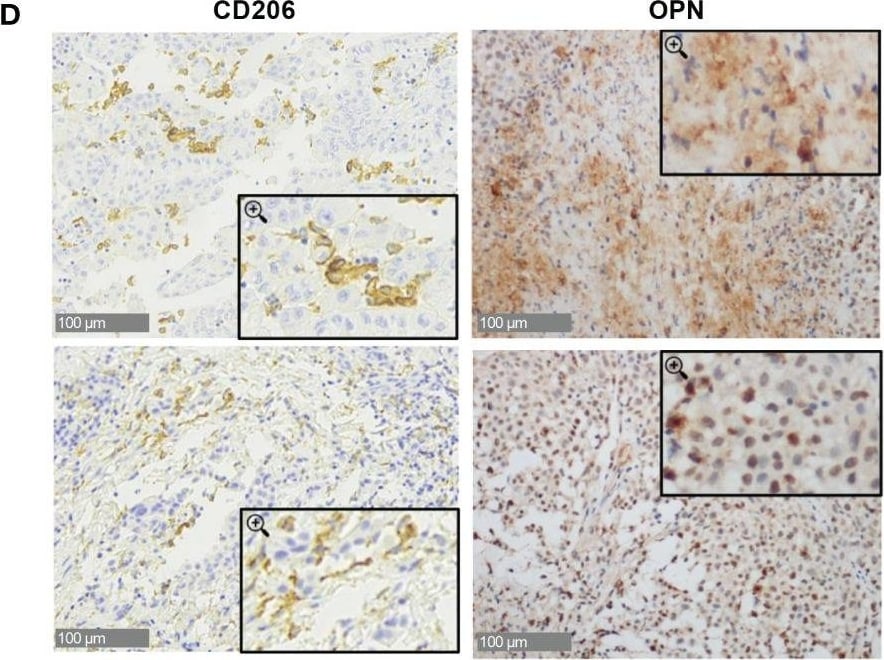
Osteopontin is overexpressed in human MPM patients. (A) Real Time PCR for the Spp1 gene (osteopontin, OPN) in surgical human MPM samples. Comparison between tumor and undiseased adjacent tissues. Data are shown as mean ± SEM (Unpaired t test with Welch’s correction). (B) ELISA quantification of hOPN on plasma samples from 99 MPM patients and 101 healthy subjects. (ROUT, identify outlier and Unpaired t test with Welch’s correction). (C) Kaplan-Meier curves of overall survival according to OPN levels categorized based on CART analysis cut-off (n=61 MPM patients). (D–F) Representative images of immunohistochemistry in MPM tumor tissues stained for OPN or CD206 (40x, insert 100x) and semi-quantitative analysis in 28 cases (0=negative, 1 = 1-25% positivity, 2 = 26-50% positivity, 3= >50% positivity). Data are shown as mean ± SEM. (Unpaired t test with Welch’s correction). (G, H) ELISA quantification of hOPN on plasma samples from 18 MPM patients treated with immunotherapy. Blood was collected at baseline and after revalutation at 4-6 months. Patients with progressive disease (PD): 6 patients; stable disease (SD): 6 patients; partial response (PR): 6 patients. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37398648), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MMR/CD206 by Immunohistochemistry
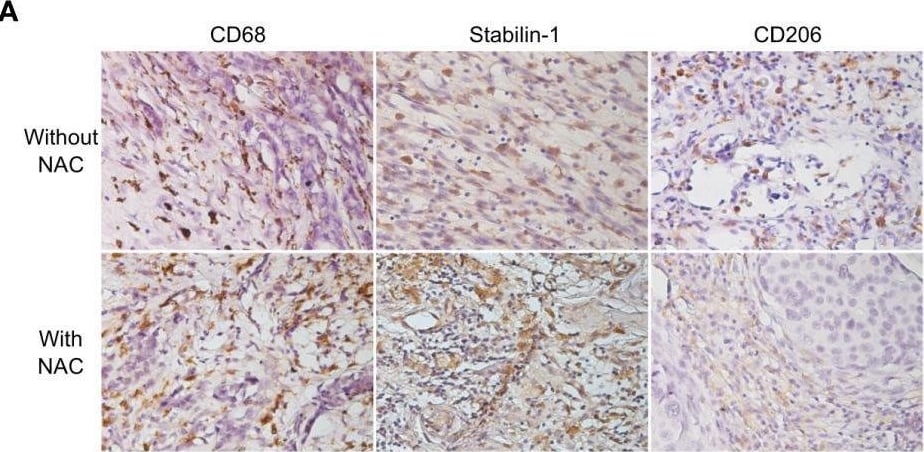
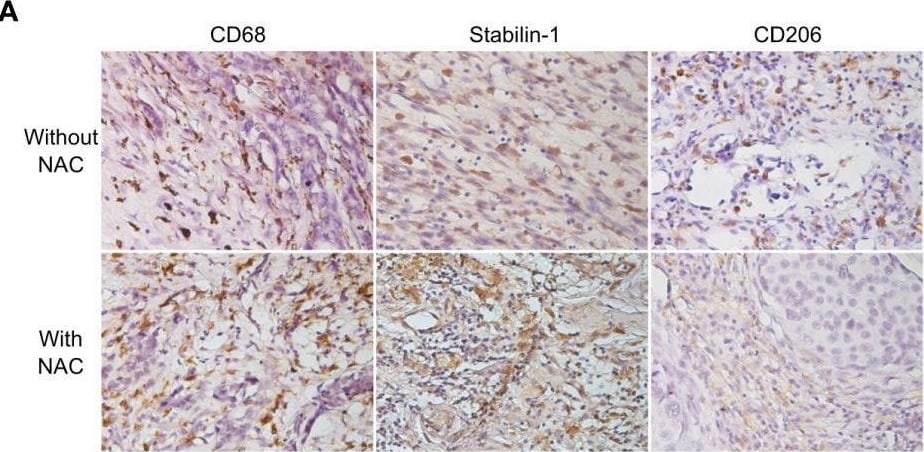
Cisplatin does not affect TAM viability and M2 phenotype. (A) IHC analysis of CD68, stabilin-1 and CD206 expression in human breast cancer tissue treated and not treated with cisplatin-based therapy. (B) Quantitative analysis of CD68, stabilin-1 and CD206 expression. Tumor tissue was obtained after surgery for all patients. (C) Schematic description of ex vivo TAM generation which were stimulated with MCF-7 supernatants and treated by cisplatin. (D) Immunofluorescent staining/confocal microscopy of CD206 and stabilin-1 expression in CD68+ ex vivo TAMs. Scale bars are equal to 11µM. (E) Viability assay for ex vivo TAMs treated with cisplatin. Viability was estimated on day 6 before cisplatin treatment and day 9 after cisplatin-treatment (n=6 independent experiments). Data information: In (B), the level of protein expression is presented as Me (Q1;Q3). In (E), OD means defined in the percentage of NS control are presented as mean ± SD. The Manna-Whitney test was applied to compare two independent groups. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36960065), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MMR/CD206 by Immunohistochemistry
Cisplatin does not affect TAM viability and M2 phenotype. (A) IHC analysis of CD68, stabilin-1 and CD206 expression in human breast cancer tissue treated and not treated with cisplatin-based therapy. (B) Quantitative analysis of CD68, stabilin-1 and CD206 expression. Tumor tissue was obtained after surgery for all patients. (C) Schematic description of ex vivo TAM generation which were stimulated with MCF-7 supernatants and treated by cisplatin. (D) Immunofluorescent staining/confocal microscopy of CD206 and stabilin-1 expression in CD68+ ex vivo TAMs. Scale bars are equal to 11µM. (E) Viability assay for ex vivo TAMs treated with cisplatin. Viability was estimated on day 6 before cisplatin treatment and day 9 after cisplatin-treatment (n=6 independent experiments). Data information: In (B), the level of protein expression is presented as Me (Q1;Q3). In (E), OD means defined in the percentage of NS control are presented as mean ± SD. The Manna-Whitney test was applied to compare two independent groups. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36960065), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human MMR/CD206 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed human peripheral blood mononuclear cells
Sample: Immersion fixed human peripheral blood mononuclear cells
Immunohistochemistry
3-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human liver
Sample: Immersion fixed paraffin-embedded sections of human liver
Simple Western
10 µg/mL
Sample: Human immature dendritic cells
Sample: Human immature dendritic cells
Western Blot
1 µg/mL
Sample: Human immature dendritic cells
Sample: Human immature dendritic cells
Reviewed Applications
Read 4 reviews rated 3.8 using AF2534 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: MMR/CD206
References
- East, L. and C. Isake (2002) Biochim. Biophys. Acta 1572:364.
- Chieppa, M. et al. (2003) J. Immunol. 171:4552.
- Figdor, C. et al. (2002) Nat. Rev. Immunol. 2:77.
- Taylor, M. et al. (1990) J. Biol. Chem. 265:12156.
- Leteux, C. et al. (2000) J. Exp. Med. 191:1117.
- Martinez-Pomares, L. et al. (2001) Immunobiology 204:527.
- Feinberg, H. et al. (2000) J. Biol. Chem. 275:21539.
Long Name
Macrophage Mannose Receptor
Alternate Names
CD206, CLEC13D, MRC1
Gene Symbol
MRC1
UniProt
Additional MMR/CD206 Products
Product Documents for Human MMR/CD206 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human MMR/CD206 Antibody
For research use only
Citations for Human MMR/CD206 Antibody
Customer Reviews for Human MMR/CD206 Antibody (4)
3.8 out of 5
4 Customer Ratings
Have you used Human MMR/CD206 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
4 of
4 reviews
Showing All
Filter By:
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 09/23/2019I used this antibody for developing a sandwich ELISA in combination with mAb (cat.MAB25341) and protein (cat.2534-MR). This combination gives good standard curve but unfortunately did not detect any MMR in our samples.
-
Application: Immunofluorescence - paraffinSample Tested: Human spleen, FFPESpecies: HumanVerified Customer | Posted 04/12/2019FFPE human spleen, CD206, 1:100pH9 antigen retrieval
-
Application: ImmunofluorescenceSample Tested: See PMID 22516068Species: HumanVerified Customer | Posted 01/07/2015
-
Application: ImmunofluorescenceSample Tested: See PMID 22500753Species: HumanVerified Customer | Posted 01/07/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...