LRRC4 (Leucine rich repeat/LRR-containing protein 4), also called NGL-2 (netrin-G ligand-2) or NAG14 (nasopharyngeal carcinoma-associated gene 14) is a 55 kDa (predicted, unglycosylated) type I transmembrane protein that is a member of the NGL family of synaptic LRR adhesion molecules (1, 2). Human LRRC4 cDNA encodes 653 amino acids (aa) that include a 38 aa signal sequence, a 489 aa extracellular domain (ECD), a 21 aa transmembrane domain, and a 105 aa cytoplasmic domain. The ECD contains nine LRRs (aa 74‑288), a C2 type Ig like domain (aa 354‑440), and a Thr-rich segment (aa 455‑526). Within the ECD, human LRRC4 shares 98% aa identity with mouse and rat, 99% aa identity with canine and bovine, and 99.6% aa identity with equine LRRC4. It also shares 54‑55% aa identity with family members LRRC4C/NGL-1 and LRRC4B/NGL-3, but each recognizes different ligands (1). LRRC4 is predominantly expressed in the brain on neurons and astrocytes as a ligand for netrin-G2 on the dendritic surface of synaptic neurons (2-4). It is proposed to regulate the formation of excitatory synapses via recruitment of PSD-95 to the cytoplasmic domain after aggregation of LRRC4 at the surface (3, 5). It suppresses proliferation by downregulating cell signaling pathways, resulting in altered expression of cell cycle regulating proteins and delay at the late G1 phase (1, 2, 6-8). It is thus considered a tumor suppressor protein and is often downregulated in brain tumors, particularly gliomas (1, 2, 6). Forced expression of LRRC4 in tumor cells slows proliferation and promotes differentiation (1, 4, 9). Addition of soluable LRRC4 to cultured neurons reduces excitatory synapse formation (3).

Key Product Details
Species Reactivity
Human, Mouse
Applications
Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 701424
Loading...
Product Specifications
Immunogen
Chinese hamster ovary cell line CHO-derived recombinant human LRRC4
Ala39-Lys527
Accession # Q9HBW1
Ala39-Lys527
Accession # Q9HBW1
Specificity
Detects human and mouse LRRC4 in direct ELISAs and Western blots. In direct ELISAs and Western blots, no
cross-reactivity with recombinant human (rh) LRRC3, rhLRRC4B, or rhLRRC32 is
observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human/Mouse LRRC4 Antibody
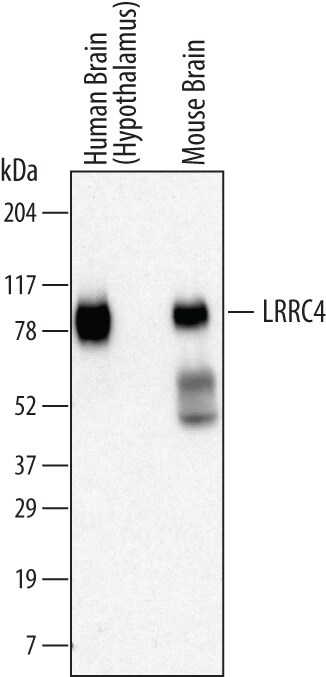
Detection of Human and Mouse LRRC4 by Western Blot.
Western blot shows lysates of human brain (hypothalamus) tissue and mouse brain tissue. PVDF membrane was probed with 1 µg/mL of Mouse Anti-Human LRRC4 Monoclonal Antibody (Catalog # MAB4995) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF007). A specific band was detected for LRRC4 at approximately 80 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Human/Mouse LRRC4 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Human brain (hypothalamus) tissue and mouse brain tissue
Sample: Human brain (hypothalamus) tissue and mouse brain tissue
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: LRRC4
References
- Woo, J. et al. (2009) Mol. Cell. Neurosci. 42:1.
- Zhang, Q. et al. (2005) FEBS Lett. 579:3674.
- Kim, S. et al. (2006) Nat. Neurosci. 9:1294.
- Wu, M. et al. (2007) Acta Biochim Biophys Sin (Shanghai) 39:731.
- Nishimura-Akiyoshi, S. et al. (2007) Proc. Natl. Acad. Sci. USA 104:14801.
- Wu, M. et al. (2006) Mol. Biol. Cell 17:3534.
- Wu, M. et al. (2008) J. Cell. Biochem. 103:245.
- Wu, M. et al. (2008) J. Cell. Physiol. 214:65.
- Zhang, W. et al. (2008) Genes Brain Behav. 7:385.
Long Name
Leucine-rich Repeat Containing 4
Alternate Names
BAG, NAG14
Gene Symbol
LRRC4
UniProt
Additional LRRC4 Products
Product Documents for Human/Mouse LRRC4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse LRRC4 Antibody
For research use only
Related Research Areas
Customer Reviews for Human/Mouse LRRC4 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse LRRC4 Antibody and earn rewards!
Have you used Human/Mouse LRRC4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
