PDGF is a major serum mitogen that can exist as a homo- or heterodimeric protein consisting of disulfide-linked PDGF-A and PDGF-B chains. The PDGF‑AA, PDGF‑BB, and PDGF‑AB isoforms have been shown to bind to two distinct cell surface PDGF receptors with different affinities. Whereas PDGF R alpha binds all three PDGF isoforms with high affinity, PDGF R beta binds PDGF-BB and -AB, but not PDGF-AA. Both PDGF R alpha and PDGF R beta are members of the class III subfamily of receptor tyrosine kinases (RTK) that also includes the receptors for M-CSF, SCF, and Flt-3 ligand. All class III RTKs are characterized by the presence of five immunoglobulin-like domains in their extracellular region and a split kinase domain in their intracellular region. PDGF binding induces receptor homo-and heterodimerization and signal transduction. The expression of the alpha and beta receptors is independently regulated in various cell types. Only PDGF R alpha is expressed in oligodendrocyte progenitor cells, mesothelial cell, and liver endothelial cells. Soluble PDGF R alpha has been detected in cell conditioned medium and human plasma. Recombinant soluble PDGF R alpha binds PDGF with high affinity and is a potent PDGF antagonist (1).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Porcine, Primate - Callithrix jacchus (Common Marmoset), Rabbit
Applications
Validated:
Immunohistochemistry, Western Blot, Neutralization
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, Immunodepletion
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant human PDGF R alpha
Gln24-Glu524
Accession # P16234
Gln24-Glu524
Accession # P16234
Specificity
Detects human PDGF R alpha in direct ELISAs and Western blots. In direct ELISAs, less than 2% cross-reactivity with recombinant mouse PDGF R alpha, recombinant human (rh) PDGF R beta, rhFGF R2, and rhFGF R3 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human PDGF R alpha Antibody
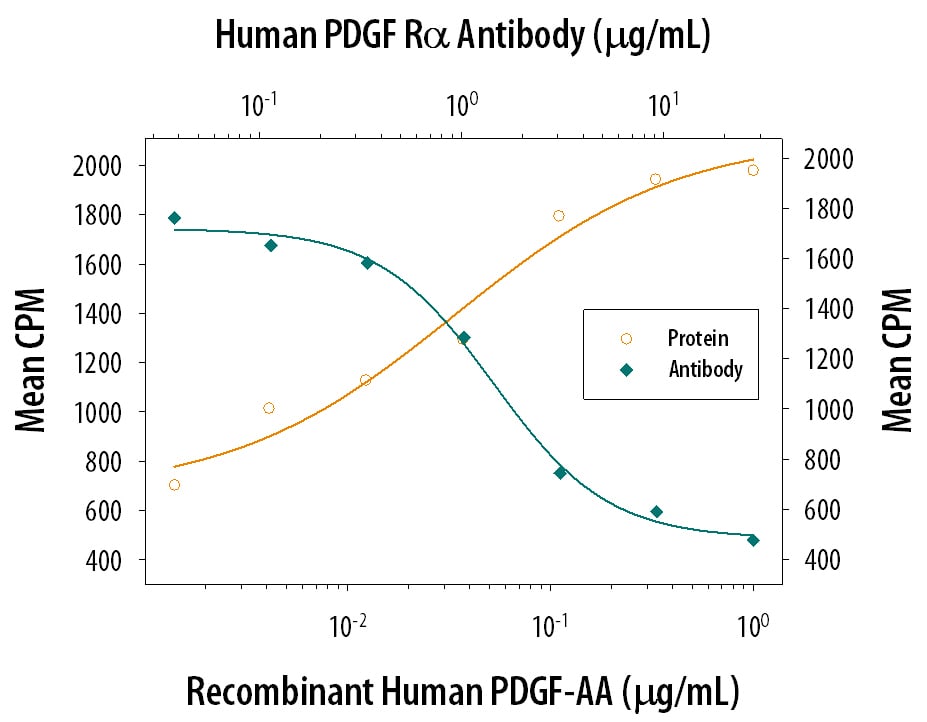
Cell Proliferation Induced by PDGF‑AA and Neutralization by Human PDGF R alpha Antibody.
Recombinant Human PDGF-AA (Catalog # 221-AA) stimulates proliferation in the WS-1 human fetal skin fibroblast cell line in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Human PDGF-AA (10 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Human PDGF Ra Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-307-NA). The ND50 is typically 1-6 µg/mL.PDGF R alpha in Human Breast Cancer Tissue.
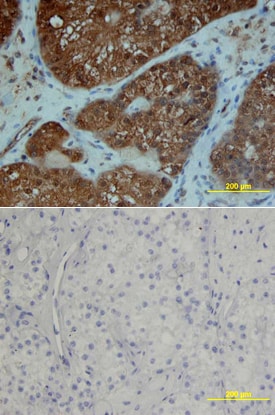
PDGF Ra was detected in immersion fixed paraffin-embedded sections of human breast cancer tissue using Goat Anti-Human PDGF Ra Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-307-NA) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Lower panel shows a lack of labeling if primary antibodies are omitted and tissue is stained only with secondary antibody followed by incubation with detection reagents. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.PDGF R alpha in Human Ovary.
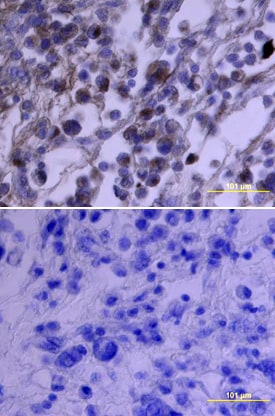
PDGF Ra was detected in immersion fixed paraffin-embedded sections of human ovarian array using Goat Anti-Human PDGF Ra Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-307-NA) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.PDGF R alpha in Human Osteosarcoma.
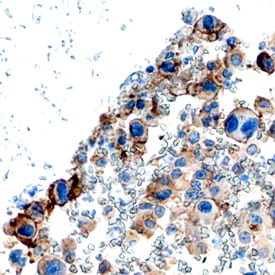
PDGF Ra was detected in immersion fixed paraffin-embedded sections of human osteosarcoma using Goat Anti-Human PDGF Ra Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-307-NA) at 3 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to plasma membranes. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
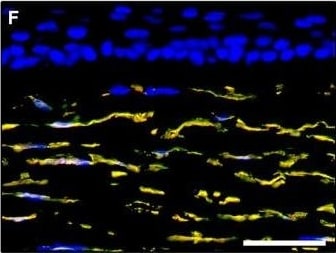
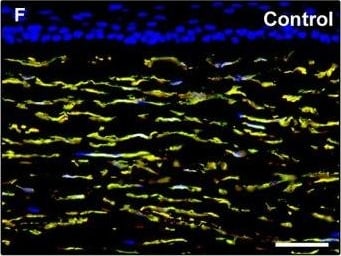
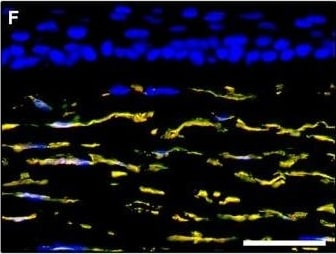
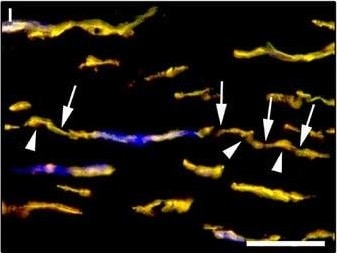
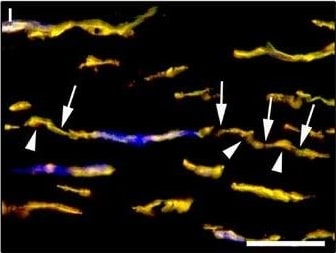
Representative light and fluorescence microscopy photomicrographs of normal human corneal sections. (A–C) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) CD34‐positive stromal cells are orderly arranged and parallel to the corneal surface. (B and C) At higher magnification, the CD34‐positive stromal cells appear as spindle‐shaped cells with a small oval body and typically two long and thin moniliform cell processes characterized by the alternation of slender segments (arrows) and knobs/dilations (arrowheads) along their length. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. Colocalization of CD34 and PDGFR alpha in stromal cells gives rise to yellow staining either in the anterior corneal stroma (D–F) or in the deeper corneal stromal layer (G–I). CD34+/PDGFR alpha + stromal cells display cell morphologies very evocative for telocytes: a small cell body with very long prolongations (telopodes) characterized by a moniliform silhouette with the alternation of podoms (arrowheads) and podomers (arrows). (J–L) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. Either in the subepithelial corneal stroma or in the deeper stromal layer, numerous CD34‐positive stromal cells coexpress c‐kit. Inset: Higher magnification view of CD34+/c‐kit+ corneal stromal cells. Scale bar: 50 μm (A, D–F and J–L), 25 μm (B, C and G–I). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
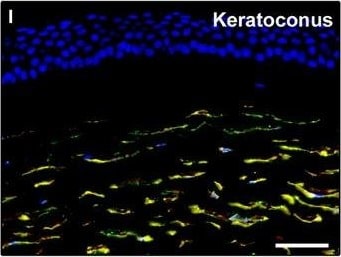
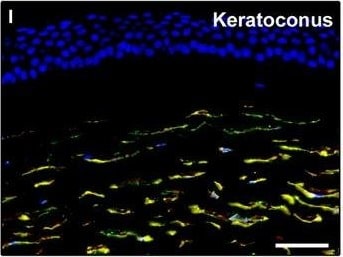
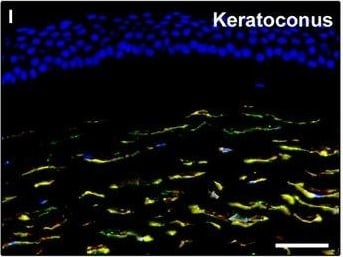
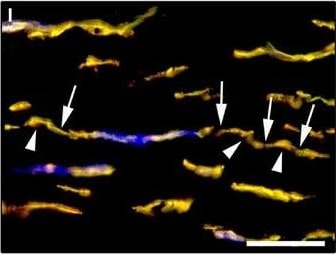
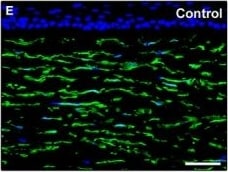
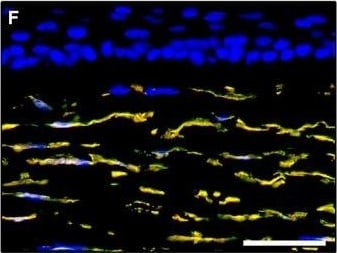
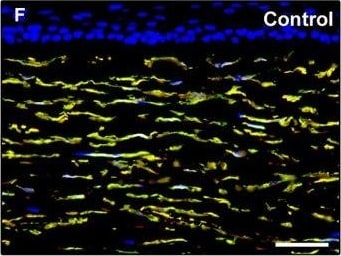
Representative light and fluorescence microscopy photomicrographs of normal and keratoconic human corneal sections. (A and B) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) In control normal corneas, CD34‐positive stromal cells displaying morphological features of telocytes are orderly arranged and parallel to the corneal surface. (B) In keratoconus, a patchy loss of CD34‐positive stromal cells is mostly evident in the anterior corneal stroma. Insets: Higher magnification views of CD34‐positive corneal stromal cells. (C) Results of quantitative analysis of CD34‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. (D–F) In control normal corneas, CD34+/PDGFR alpha + stromal cells (telocytes) are orderly distributed throughout the stromal compartment. (G–I) In keratoconic corneas, a patchy loss of CD34+/PDGFR alpha + stromal cells (telocytes) is mainly evident in the subepithelial stroma. (J and K) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. (J) In control normal corneas, numerous CD34+/c‐kit+ stromal cells (telocytes) are present throughout the stromal layer. (K) In keratoconic corneas, the CD34+/c‐kit+ stromal cell subpopulation is almost completely lost. (L) Results of quantitative analysis of c‐kit‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. TC: telocytes; hpf: high‐power field. Scale bar: 100 μm (A and B), 50 μm (D–K). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal and keratoconic human corneal sections. (A and B) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) In control normal corneas, CD34‐positive stromal cells displaying morphological features of telocytes are orderly arranged and parallel to the corneal surface. (B) In keratoconus, a patchy loss of CD34‐positive stromal cells is mostly evident in the anterior corneal stroma. Insets: Higher magnification views of CD34‐positive corneal stromal cells. (C) Results of quantitative analysis of CD34‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. (D–F) In control normal corneas, CD34+/PDGFR alpha + stromal cells (telocytes) are orderly distributed throughout the stromal compartment. (G–I) In keratoconic corneas, a patchy loss of CD34+/PDGFR alpha + stromal cells (telocytes) is mainly evident in the subepithelial stroma. (J and K) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. (J) In control normal corneas, numerous CD34+/c‐kit+ stromal cells (telocytes) are present throughout the stromal layer. (K) In keratoconic corneas, the CD34+/c‐kit+ stromal cell subpopulation is almost completely lost. (L) Results of quantitative analysis of c‐kit‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. TC: telocytes; hpf: high‐power field. Scale bar: 100 μm (A and B), 50 μm (D–K). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal and keratoconic human corneal sections. (A and B) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) In control normal corneas, CD34‐positive stromal cells displaying morphological features of telocytes are orderly arranged and parallel to the corneal surface. (B) In keratoconus, a patchy loss of CD34‐positive stromal cells is mostly evident in the anterior corneal stroma. Insets: Higher magnification views of CD34‐positive corneal stromal cells. (C) Results of quantitative analysis of CD34‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. (D–F) In control normal corneas, CD34+/PDGFR alpha + stromal cells (telocytes) are orderly distributed throughout the stromal compartment. (G–I) In keratoconic corneas, a patchy loss of CD34+/PDGFR alpha + stromal cells (telocytes) is mainly evident in the subepithelial stroma. (J and K) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. (J) In control normal corneas, numerous CD34+/c‐kit+ stromal cells (telocytes) are present throughout the stromal layer. (K) In keratoconic corneas, the CD34+/c‐kit+ stromal cell subpopulation is almost completely lost. (L) Results of quantitative analysis of c‐kit‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. TC: telocytes; hpf: high‐power field. Scale bar: 100 μm (A and B), 50 μm (D–K). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal human corneal sections. (A–C) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) CD34‐positive stromal cells are orderly arranged and parallel to the corneal surface. (B and C) At higher magnification, the CD34‐positive stromal cells appear as spindle‐shaped cells with a small oval body and typically two long and thin moniliform cell processes characterized by the alternation of slender segments (arrows) and knobs/dilations (arrowheads) along their length. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. Colocalization of CD34 and PDGFR alpha in stromal cells gives rise to yellow staining either in the anterior corneal stroma (D–F) or in the deeper corneal stromal layer (G–I). CD34+/PDGFR alpha + stromal cells display cell morphologies very evocative for telocytes: a small cell body with very long prolongations (telopodes) characterized by a moniliform silhouette with the alternation of podoms (arrowheads) and podomers (arrows). (J–L) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. Either in the subepithelial corneal stroma or in the deeper stromal layer, numerous CD34‐positive stromal cells coexpress c‐kit. Inset: Higher magnification view of CD34+/c‐kit+ corneal stromal cells. Scale bar: 50 μm (A, D–F and J–L), 25 μm (B, C and G–I). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal and keratoconic human corneal sections. (A and B) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) In control normal corneas, CD34‐positive stromal cells displaying morphological features of telocytes are orderly arranged and parallel to the corneal surface. (B) In keratoconus, a patchy loss of CD34‐positive stromal cells is mostly evident in the anterior corneal stroma. Insets: Higher magnification views of CD34‐positive corneal stromal cells. (C) Results of quantitative analysis of CD34‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. (D–F) In control normal corneas, CD34+/PDGFR alpha + stromal cells (telocytes) are orderly distributed throughout the stromal compartment. (G–I) In keratoconic corneas, a patchy loss of CD34+/PDGFR alpha + stromal cells (telocytes) is mainly evident in the subepithelial stroma. (J and K) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. (J) In control normal corneas, numerous CD34+/c‐kit+ stromal cells (telocytes) are present throughout the stromal layer. (K) In keratoconic corneas, the CD34+/c‐kit+ stromal cell subpopulation is almost completely lost. (L) Results of quantitative analysis of c‐kit‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. TC: telocytes; hpf: high‐power field. Scale bar: 100 μm (A and B), 50 μm (D–K). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal and keratoconic human corneal sections. (A and B) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) In control normal corneas, CD34‐positive stromal cells displaying morphological features of telocytes are orderly arranged and parallel to the corneal surface. (B) In keratoconus, a patchy loss of CD34‐positive stromal cells is mostly evident in the anterior corneal stroma. Insets: Higher magnification views of CD34‐positive corneal stromal cells. (C) Results of quantitative analysis of CD34‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. (D–F) In control normal corneas, CD34+/PDGFR alpha + stromal cells (telocytes) are orderly distributed throughout the stromal compartment. (G–I) In keratoconic corneas, a patchy loss of CD34+/PDGFR alpha + stromal cells (telocytes) is mainly evident in the subepithelial stroma. (J and K) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. (J) In control normal corneas, numerous CD34+/c‐kit+ stromal cells (telocytes) are present throughout the stromal layer. (K) In keratoconic corneas, the CD34+/c‐kit+ stromal cell subpopulation is almost completely lost. (L) Results of quantitative analysis of c‐kit‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. TC: telocytes; hpf: high‐power field. Scale bar: 100 μm (A and B), 50 μm (D–K). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal human corneal sections. (A–C) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) CD34‐positive stromal cells are orderly arranged and parallel to the corneal surface. (B and C) At higher magnification, the CD34‐positive stromal cells appear as spindle‐shaped cells with a small oval body and typically two long and thin moniliform cell processes characterized by the alternation of slender segments (arrows) and knobs/dilations (arrowheads) along their length. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. Colocalization of CD34 and PDGFR alpha in stromal cells gives rise to yellow staining either in the anterior corneal stroma (D–F) or in the deeper corneal stromal layer (G–I). CD34+/PDGFR alpha + stromal cells display cell morphologies very evocative for telocytes: a small cell body with very long prolongations (telopodes) characterized by a moniliform silhouette with the alternation of podoms (arrowheads) and podomers (arrows). (J–L) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. Either in the subepithelial corneal stroma or in the deeper stromal layer, numerous CD34‐positive stromal cells coexpress c‐kit. Inset: Higher magnification view of CD34+/c‐kit+ corneal stromal cells. Scale bar: 50 μm (A, D–F and J–L), 25 μm (B, C and G–I). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal human corneal sections. (A–C) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) CD34‐positive stromal cells are orderly arranged and parallel to the corneal surface. (B and C) At higher magnification, the CD34‐positive stromal cells appear as spindle‐shaped cells with a small oval body and typically two long and thin moniliform cell processes characterized by the alternation of slender segments (arrows) and knobs/dilations (arrowheads) along their length. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. Colocalization of CD34 and PDGFR alpha in stromal cells gives rise to yellow staining either in the anterior corneal stroma (D–F) or in the deeper corneal stromal layer (G–I). CD34+/PDGFR alpha + stromal cells display cell morphologies very evocative for telocytes: a small cell body with very long prolongations (telopodes) characterized by a moniliform silhouette with the alternation of podoms (arrowheads) and podomers (arrows). (J–L) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. Either in the subepithelial corneal stroma or in the deeper stromal layer, numerous CD34‐positive stromal cells coexpress c‐kit. Inset: Higher magnification view of CD34+/c‐kit+ corneal stromal cells. Scale bar: 50 μm (A, D–F and J–L), 25 μm (B, C and G–I). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal human corneal sections. (A–C) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) CD34‐positive stromal cells are orderly arranged and parallel to the corneal surface. (B and C) At higher magnification, the CD34‐positive stromal cells appear as spindle‐shaped cells with a small oval body and typically two long and thin moniliform cell processes characterized by the alternation of slender segments (arrows) and knobs/dilations (arrowheads) along their length. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. Colocalization of CD34 and PDGFR alpha in stromal cells gives rise to yellow staining either in the anterior corneal stroma (D–F) or in the deeper corneal stromal layer (G–I). CD34+/PDGFR alpha + stromal cells display cell morphologies very evocative for telocytes: a small cell body with very long prolongations (telopodes) characterized by a moniliform silhouette with the alternation of podoms (arrowheads) and podomers (arrows). (J–L) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. Either in the subepithelial corneal stroma or in the deeper stromal layer, numerous CD34‐positive stromal cells coexpress c‐kit. Inset: Higher magnification view of CD34+/c‐kit+ corneal stromal cells. Scale bar: 50 μm (A, D–F and J–L), 25 μm (B, C and G–I). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal human corneal sections. (A–C) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) CD34‐positive stromal cells are orderly arranged and parallel to the corneal surface. (B and C) At higher magnification, the CD34‐positive stromal cells appear as spindle‐shaped cells with a small oval body and typically two long and thin moniliform cell processes characterized by the alternation of slender segments (arrows) and knobs/dilations (arrowheads) along their length. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. Colocalization of CD34 and PDGFR alpha in stromal cells gives rise to yellow staining either in the anterior corneal stroma (D–F) or in the deeper corneal stromal layer (G–I). CD34+/PDGFR alpha + stromal cells display cell morphologies very evocative for telocytes: a small cell body with very long prolongations (telopodes) characterized by a moniliform silhouette with the alternation of podoms (arrowheads) and podomers (arrows). (J–L) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. Either in the subepithelial corneal stroma or in the deeper stromal layer, numerous CD34‐positive stromal cells coexpress c‐kit. Inset: Higher magnification view of CD34+/c‐kit+ corneal stromal cells. Scale bar: 50 μm (A, D–F and J–L), 25 μm (B, C and G–I). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PDGFR alpha by Immunocytochemistry/Immunofluorescence
Representative light and fluorescence microscopy photomicrographs of normal and keratoconic human corneal sections. (A and B) CD34 immunoperoxidase‐based immunohistochemistry with haematoxylin counterstain. (A) In control normal corneas, CD34‐positive stromal cells displaying morphological features of telocytes are orderly arranged and parallel to the corneal surface. (B) In keratoconus, a patchy loss of CD34‐positive stromal cells is mostly evident in the anterior corneal stroma. Insets: Higher magnification views of CD34‐positive corneal stromal cells. (C) Results of quantitative analysis of CD34‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. (D–I) Double immunofluorescence labelling for CD34 (red) and PDGFR alpha (green) with DAPI (blue) counterstain for nuclei. (D–F) In control normal corneas, CD34+/PDGFR alpha + stromal cells (telocytes) are orderly distributed throughout the stromal compartment. (G–I) In keratoconic corneas, a patchy loss of CD34+/PDGFR alpha + stromal cells (telocytes) is mainly evident in the subepithelial stroma. (J and K) Double immunofluorescence labelling for CD34 (red) and c‐kit (green) with DAPI (blue) counterstain for nuclei. (J) In control normal corneas, numerous CD34+/c‐kit+ stromal cells (telocytes) are present throughout the stromal layer. (K) In keratoconic corneas, the CD34+/c‐kit+ stromal cell subpopulation is almost completely lost. (L) Results of quantitative analysis of c‐kit‐positive telocyte counts per high‐power field in the corneal stroma of healthy controls (n = 6) and patients with keratoconus (n = 6). Data are mean ± S.D. *P < 0.001 versus control. TC: telocytes; hpf: high‐power field. Scale bar: 100 μm (A and B), 50 μm (D–K). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28714595), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human PDGF R alpha Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human breast cancer tissue, human ovarian array, and human osteosarcoma
Sample: Immersion fixed paraffin-embedded sections of human breast cancer tissue, human ovarian array, and human osteosarcoma
Western Blot
0.1 µg/mL
Sample: Recombinant Human PDGF R alpha (Catalog # 322-PR)
Sample: Recombinant Human PDGF R alpha (Catalog # 322-PR)
Neutralization
Measured by its ability to neutralize PDGF‑AA-induced proliferation in the WS-1 human fetal skin fibroblast cell line. The Neutralization Dose (ND50) is typically 1-6 µg/mL in the presence of 10 ng/mL Recombinant Human PDGF‑AA.
Reviewed Applications
Read 3 reviews rated 4.3 using AF-307-NA in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: PDGF R alpha
References
- Heldin, C.H. and L. Claesson-Welsh (1994) Guidebook to Cytokines and Their Receptors, Nicola, N.A. (ed) Oxford University Press, New York, NY p. 202.
Long Name
Platelet-derived Growth Factor Receptor alpha
Alternate Names
CD140a, PDGFR alpha, PDGFRA
Gene Symbol
PDGFRA
UniProt
Additional PDGF R alpha Products
Product Documents for Human PDGF R alpha Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human PDGF R alpha Antibody
For research use only
Citations for Human PDGF R alpha Antibody
Customer Reviews for Human PDGF R alpha Antibody (3)
4.3 out of 5
3 Customer Ratings
Have you used Human PDGF R alpha Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
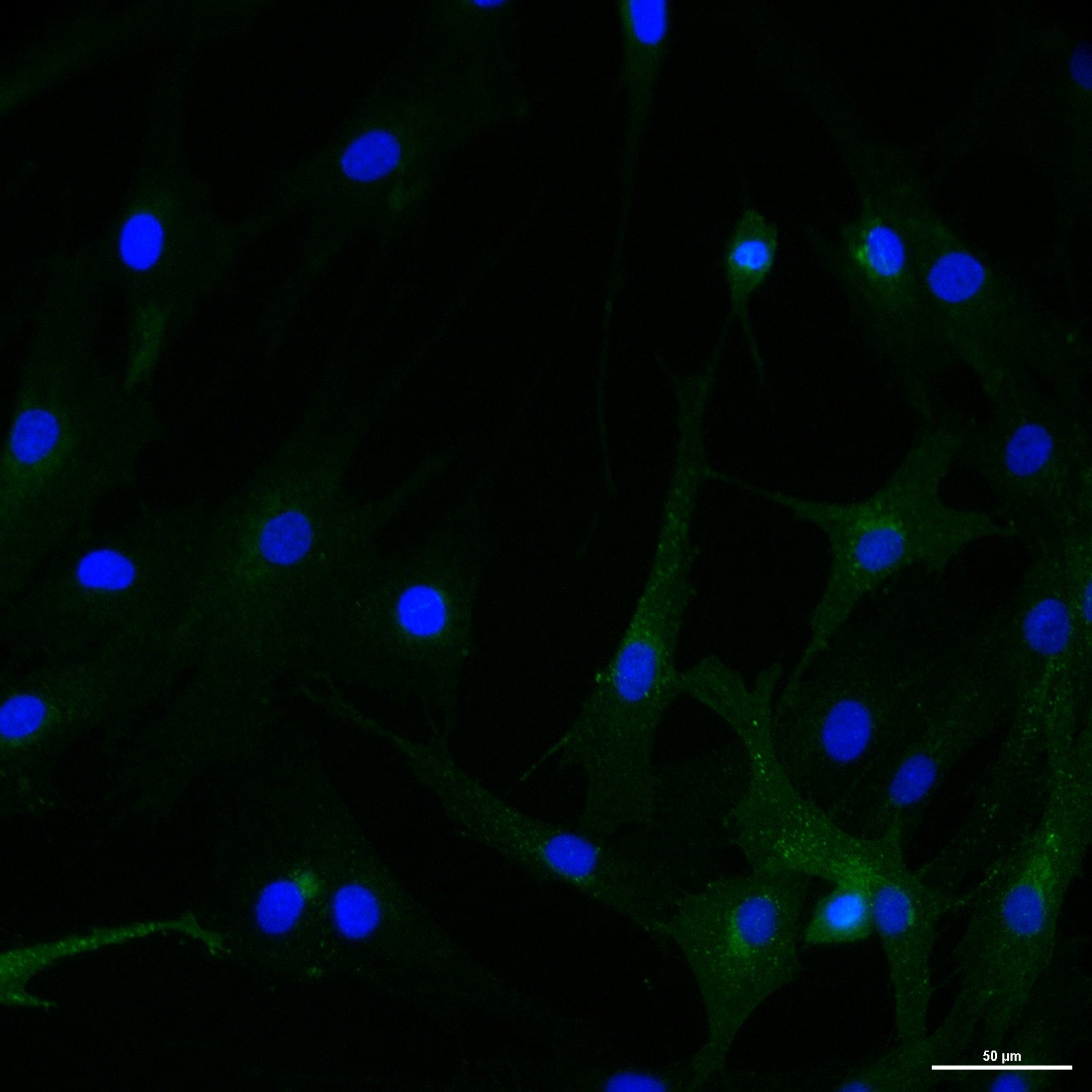
Application: Immunohistochemistry-FrozenSample Tested: human dermal fibroblastsSpecies: HumanVerified Customer | Posted 02/22/2019
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Spinal cord and Brain tissueSpecies: MouseVerified Customer | Posted 06/13/2017
-
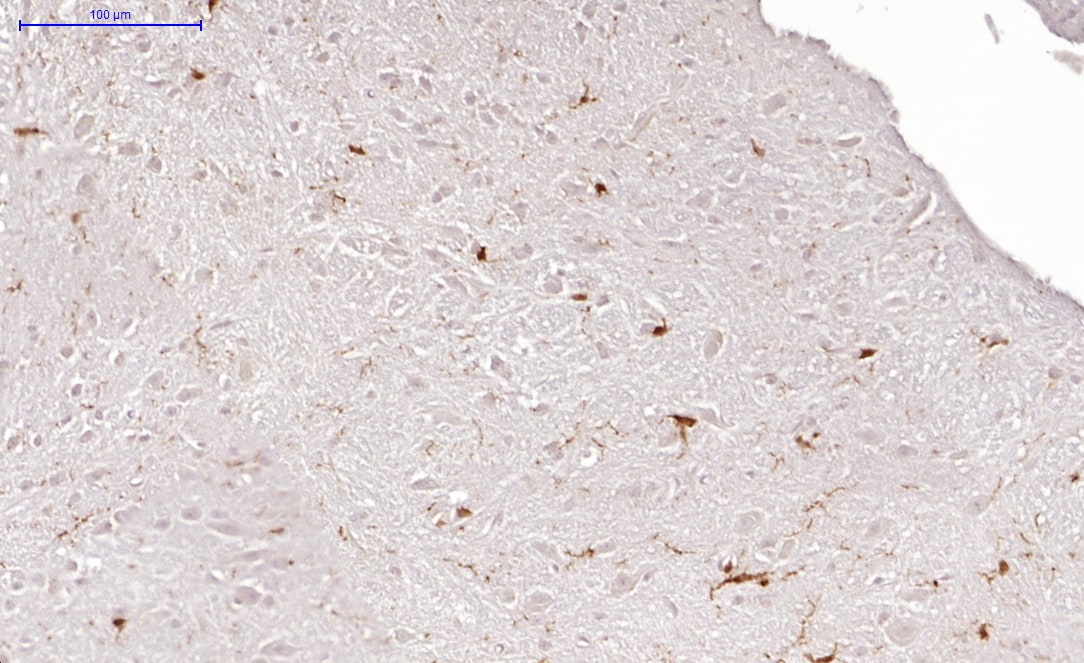
Application: ImmunohistochemistrySample Tested: brain and spinal cordSpecies: MouseVerified Customer | Posted 05/17/2017Mouse spinal cordworked on mouse spinal cord
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars