HGF R, also known as Met (from N-methyl-N’-nitro-N-nitrosoguanidine induced), is a glycosylated receptor tyrosine kinase that plays a central role in epithelial morphogenesis and cancer development. HGF R is synthesized as a single chain precursor which undergoes cotranslational proteolytic cleavage. This generates a mature HGF R that is a disulfide-linked dimer composed of a 50 kDa extracellular alpha chain and a 145 kDa transmembrane beta chain (1, 2). The extracellular domain (ECD) contains a seven bladed beta -propeller sema domain, a cysteine-rich PSI/MRS, and four Ig-like E-set domains, while the cytoplasmic region includes the tyrosine kinase domain (3, 4). Proteolysis and alternate splicing generate additional forms of human HGF R which either lack of the kinase domain, consist of secreted extracellular domains, or are deficient in proteolytic separation of the alpha and beta chains (5 - 7). The sema domain, which is formed by both the alpha and beta chains of HGF R, mediates both ligand binding and receptor dimerization (3, 8). Ligand-induced tyrosine phosphorylation in the cytoplasmic region activates the kinase domain and provides docking sites for multiple SH2-containing molecules (9, 10). HGF stimulation induces HGF R downregulation via internalization and proteasome-dependent degradation (11). In the absence of ligand, HGF R forms noncovalent complexes with a variety of membrane proteins including CD44v6, CD151, EGF R, Fas, Integrin alpha 6/ beta 4, Plexins B1, 2, 3, and MSP R/Ron (12 - 19). Ligation of one complex component triggers activation of the other, followed by cooperative signaling effects (12 - 19). Formation of some of these heteromeric complexes is a requirement for epithelial cell morphogenesis and tumor cell invasion (12, 16, 17). Paracrine induction of epithelial cell scattering and branching tubulogenesis results from the stimulation of HGF R on undifferentiated epithelium by HGF released from neighboring mesenchymal cells (20). Genetic polymorphisms, chromosomal translocation, overexpression, and additional splicing and proteolytic cleavage of HGF R have been described in a wide range of cancers (1). Within the ECD, human HGF R shares 86% - 88% aa sequence identity with canine, mouse, and rat HGF R.
Human Phospho-HGFR/c-MET (Y1003) Antibody
R&D Systems | Catalog # AF4059


Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Western Blot, Array Development, Protein Array
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Phosphopeptide containing human HGF R Y1003 site
Specificity
Detects human and mouse HGF R/c-MET when phosphorylated at Y1003.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Human Phospho-HGFR/c-MET (Y1003) Antibody
Detection of Human Phospho-HGF R/c-MET (Y1003) by Western Blot.
Western blot shows lysates of A431 human epithelial carcinoma cell line untreated (-) or treated (+) with 100 µM pervanadate (PV) for 10 minutes. PVDF membrane was probed with 1 µg/mL of Rabbit Anti-Human Phospho-HGF R/c-MET (Y1003) Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4059), followed by HRP-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # HAF008). A specific band was detected for Phospho-HGF R/ c-MET (Y1003) at approximately 140 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Phospho-HGF R/c-MET (Y1003) in A431 Human Cell Line.
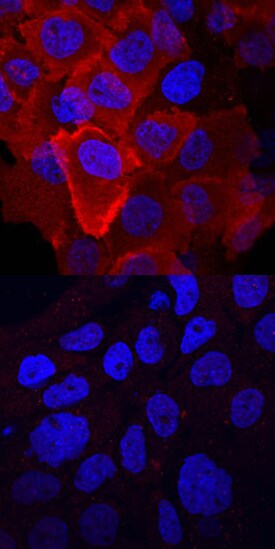
HGF R/c-MET phosphorylated at Y1003 was detected in immersion fixed A431 human epithelial carcinoma cell line untreated (lower panel) or treated (upper panel) with pervanadate using Rabbit Anti-Human Phospho-HGF R/c-MET (Y1003) Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4059) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rabbit IgG Secondary Antibody (red; Catalog # NL004) and counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human Phospho-HGFR/c-MET (Y1003) Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed A431 human epithelial carcinoma cell line treated with pervanadate
Sample: Immersion fixed A431 human epithelial carcinoma cell line treated with pervanadate
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human breast cancer tissue
Sample: Immersion fixed paraffin-embedded sections of human breast cancer tissue
Western Blot
1 µg/mL
Sample: Pervanadate-treated A431 human epithelial carcinoma cell line
Sample: Pervanadate-treated A431 human epithelial carcinoma cell line
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: HGFR/c-MET
References
- Birchmeier, C. et al. (2003) Nat. Rev. Mol. Cell Biol. 4:915.
- Corso, S. et al. (2005) Trends Mol. Med. 11:284.
- Gherardi, E. et al. (2003) Proc. Natl. Acad. Sci. 100:12039.
- Park, M. et al. (1987) Proc. Natl. Acad. Sci. 84:6379.
- Crepaldi, T. et al. (1994) J. Biol. Chem. 269:1750.
- Prat, M. et al. (1991) Mol. Cell. Biol. 12:5954.
- Rodrigues, G.A. et al. (1991) Mol. Cell. Biol. 11:2962.
- Kong-Beltran, M. et al. (2004) Cancer Cell 6:75.
- Naldini, L. et al. (1991) Mol. Cell. Biol. 11:1793.
- Ponzetto, C. et al. (1994) Cell 77:261.
- Jeffers, M. et al. (1997) Mol. Cell. Biol. 17:799.
- Orian-Rousseau, V. et al. (2002) Genes Dev. 16:3074.
- Klosek, S.K. et al. (2005) Biochem. Biophys. Res. Commun. 336:408.
- Jo, M. et al. (2000) J. Biol. Chem. 275:8806.
- Wang, X. et al. (2002) Mol. Cell 9:411.
- Trusolino, L. et al. (2001) Cell 107:643.
- Giordano, S. et al. (2002) Nat. Cell Biol. 4:720.
- Conrotto, P. et al. (2004) Oncogene 23:5131.
- Follenzi, A. et al. (2000) Oncogene 19:3041.
- Sonnenberg, E. et al. (1993) J. Cell Biol. 123:223.
Long Name
Hepatocyte Growth Factor Receptor
Alternate Names
c-MET, cMET, HGF R, MET
Gene Symbol
MET
Additional HGFR/c-MET Products
Product Documents for Human Phospho-HGFR/c-MET (Y1003) Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Phospho-HGFR/c-MET (Y1003) Antibody
For research use only
Citations for Human Phospho-HGFR/c-MET (Y1003) Antibody
Customer Reviews for Human Phospho-HGFR/c-MET (Y1003) Antibody
There are currently no reviews for this product. Be the first to review Human Phospho-HGFR/c-MET (Y1003) Antibody and earn rewards!
Have you used Human Phospho-HGFR/c-MET (Y1003) Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars