Human Relaxin-1, also called H1 Relaxin or RLN1, is one of three human relaxins in the structurally related insulin/relaxin superfamily (1, 2). Relaxin-1 is thought to be the result of duplication of the Relaxin-2 gene in higher primates only. In species below higher primates, Relaxin-1 is the equivalent of human Relaxin-2. Relaxin-1 is found in some but not all tissues expressing Relaxin-2. It is prominent in the prostate, but also present in decidua, placenta, endometrium and at low levels in the myocardium (2, 3). As with other insulin/relaxin superfamily members, human Relaxin-1 is synthesized as a preprohormone (4). Processing of the 21 kDa preprorelaxin-1 includes removal of the signal sequence, formation of two disulfide bonds between A and B chains and removal of the intervening C-chain by a prohormone convertase. The resulting mature protein is an unglycosylated, 6 kDa dimer of disulfide-linked A and B chains. Human Relaxin-1 shares 76% amino acid (aa) identity with human Relaxin-2, and 43%, 50%, and 43% aa identity with mouse, rat, and canine Relaxin-1, respectively. An alternate splice form of unknown significance has a 47 aa substitution which does not have typical C-chain cleavage motifs (5). Relaxins confer activity by binding to leucine-rich G-protein coupled receptors LGR7 and LGR8 (2, 6). Prostatic relaxins are anti-apoptotic and contribute to development and maintenance of male fertility. It is not clear whether human Relaxins -1 and -2 have distinct functions. Both use the same receptor and have the same critical amino acids for folding and for receptor interaction. While receptor affinity is similar, activity is lower for Relaxin-1 as compared to Relaxin-2 (7). Progesterone increases expression of only Relaxin-2, while glucocorticoids increase expression of both (8).


Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Lys26-Cys185
Accession # P04808
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Pro‑Relaxin‑1 Antibody
Detection of Human Relaxin‑1 by Western Blot.
Western blot shows lysates of LNCaP human prostate cancer cell line. PVDF Membrane was probed with 1 µg/mL of Goat Anti-Human Pro-Relaxin-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3257) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). A specific band was detected for Relaxin-1 at approximately 7 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Human Relaxin‑1 ELISA Standard Curve.
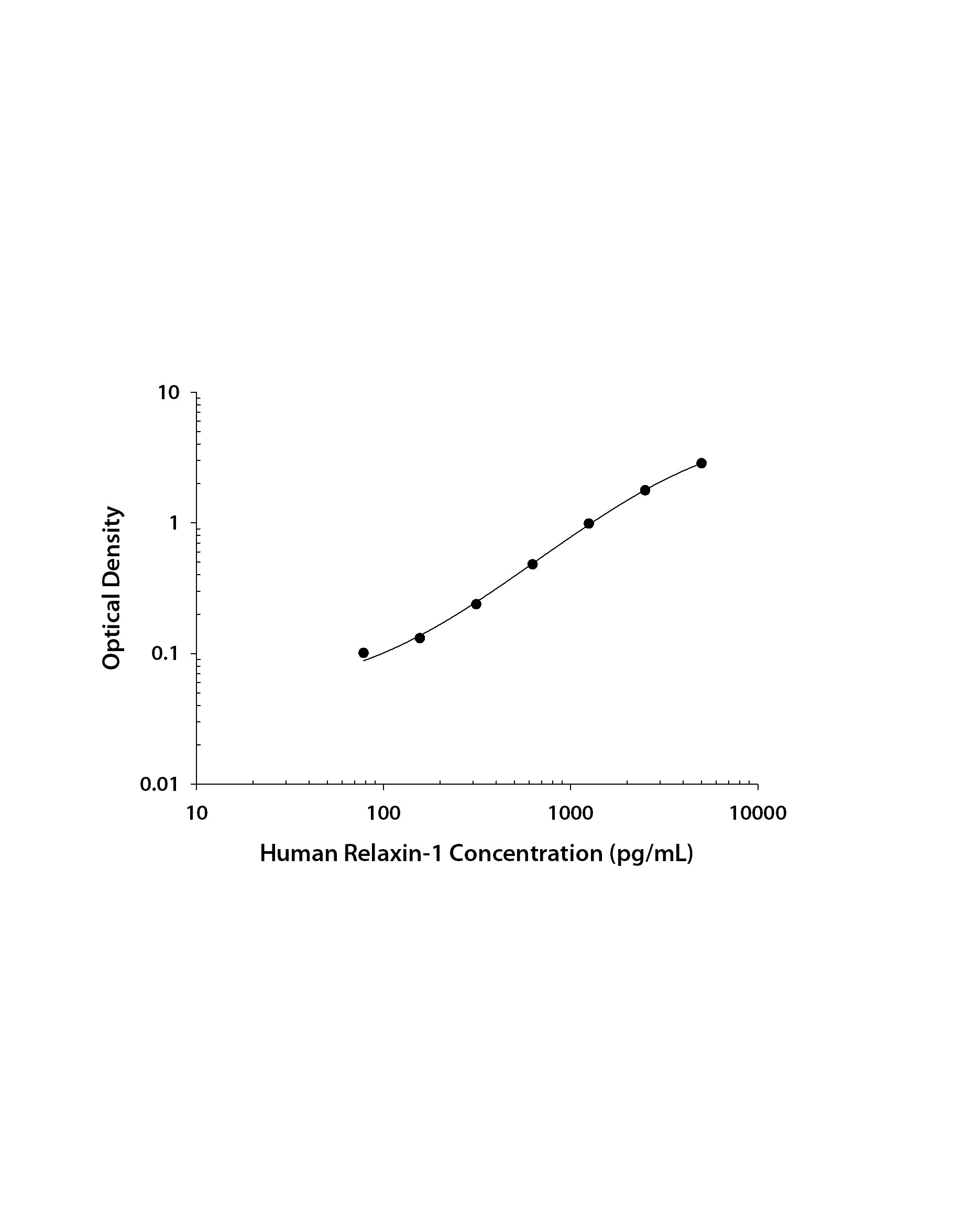
Recombinant Human Relaxin‑1 protein was serially diluted 2-fold and captured by Rat Anti-Human Relaxin‑1 Monoclonal Antibody (MAB32571) coated on a Clear Polystyrene Microplate (DY990). Goat Anti-Human Pro Relaxin‑1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3257) was biotinylated and incubated with the protein captured on the plate. Detection of the standard curve was achieved by incubating Streptavidin-HRP (DY998) followed by Substrate Solution (DY999) and stopping the enzymatic reaction with Stop Solution (DY994).Applications for Human Pro‑Relaxin‑1 Antibody
ELISA
This antibody functions as an ELISA detection antibody when paired with Rat Anti-Human Relaxin‑1 Monoclonal Antibody (Catalog # MAB32571).
This product is intended for assay development on various assay platforms requiring antibody pairs. We recommend the Human Relaxin-1 DuoSet ELISA Kit (Catalog # DY3257) for convenient development of a sandwich ELISA.
Western Blot
Sample: LNCaP human prostate cancer cell line
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Relaxin-1
References
- Hayes, E.S. (2004) Reprod. Biol. Endocrinol. 2:36.
- Sherwood, O.D. (2004) Endocr. Rev. 25:205.
- Wilkinson, T.N. et al. (2005) BMC Evol. Biol. 5:14.
- Hudson, P. et al. (1984) EMBO J. 3:2333.
- Gunnersen, J.M. et al. (1996) Mol. Cell. Endocrinol. 118:85.
- Hsu, S.Y. et al. (2002) Science 295:671.
- Schwabe, C. and E.E. Bullesbach (1994) FASEB J. 8:1152.
- Garibay-Tupas, J.L. et al. (2004) Mol. Cell. Endocrinol. 219:115.
Alternate Names
Gene Symbol
UniProt
Additional Relaxin-1 Products
Product Documents for Human Pro‑Relaxin‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Pro‑Relaxin‑1 Antibody
For research use only
Related Research Areas
Customer Reviews for Human Pro‑Relaxin‑1 Antibody
There are currently no reviews for this product. Be the first to review Human Pro‑Relaxin‑1 Antibody and earn rewards!
Have you used Human Pro‑Relaxin‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars