Siglecs(1) (sialic acid binding Ig-like lectins) are I-type (Ig-type) lectins belonging to the Ig superfamily. They are characterized by an N-terminal Ig-like V-type domain which mediates sialic acid binding, followed by varying numbers of Ig-like C2-type domains (1, 2). Eleven human Siglecs have been cloned and characterized. They are sialoadhesin/CD169/Siglec-1, CD22/Siglec-2, CD33/Siglec-3, Myelin-Associated Glycoprotein (MAG/Siglec-4a) and Siglec-5 to -11 (1‑4). To date, no Siglec has been shown to recognized any cell surface ligand other than sialic acids, suggesting that interactions with glycans containing this carbohydrate are important in mediating the biological functions of Siglecs. Siglecs 5 to 11 share a high degree of sequence similarity with CD33/Siglec-3 both in their extracellular and intracellular regions. They are collectively referred to as CD33-related Siglecs. One remarkable feature of the CD33-related Siglecs is their differential expression pattern within the hematopoietic system (2, 3). This fact, together with the presence of two conserved immunoreceptor tyrosine-based inhibition motifs (ITIMs) in their cytoplasmic tails, suggests that CD33-related Siglecs are involved in the regulation of cellular activation within the immune system.
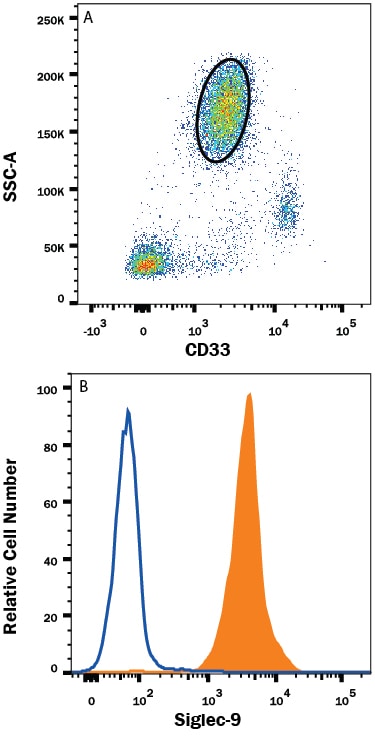
The cDNA of human Siglec-9 encodes a 463 amino acid (aa) polypeptide with a hydrophobic signal peptide, an N-terminal Ig-likeV-type domain, two Ig-like C2-type domains, a transmembrane region and a cytoplasmic tail (5, 6). In peripheral blood leukocytes, Siglec-9 is expressed on neutrophils, monocytes, a fraction of NK cells, B cells, and a minor subset of CD8+ T cells (5). It binds equally well to both 2,3- and 2,6-linked sialic acid (5, 6). Siglec-9 is closely related to Siglec-7, and they share ~80% amino acid sequence identity. The gene encoding siglec-9 was mapped to chromosome 19q13.4.