Vascular adhesion protein-1 (VAP-1), also called AOC3 (amine oxidase copper-containing 3) or SSAO (semicarbazide-sensitive amine oxidase), is a copper amine oxidase with a topaquinone cofactor. VAP-1 is a Type II integral membrane protein, but a soluble form of the enzyme is present in human serum, and its level increases in diabetes and some inflammatory liver diseases (1, 2). Human and mouse VAP-1 share 83% amino acid sequence identity. VAP-1 catalyzes the oxidative deamination of small primary amines such as methylamine, benzylamine, and aminoacetone in a reaction that produces an aldehyde, ammonia, and H2O2 (3). The enzyme is sensitive to inhibition by semicarbazide. VAP-1 expression is highest in the endothelium of lung, heart, and intestine, but low in tissues such as brain, spleen, kidney, and liver (4). VAP-1 vascular expression is regulated at sites of inflammation through its release from intracellular granules in which the protein is stored (5). The adhesive function of VAP-1 has been demonstrated in studies showing that the protein is important for the adherence of certain lymphocyte subtypes to inflamed endothelial tissues (6). VAP-1 mediated adhesion is involved in the process of leukocyte extravasation, an important feature of inflammatory responses. The role of VAP-1 amine oxidase activity in this process is not fully defined, but it appears to be carbohydrate-dependent (7). VAP-1 is considered to be a therapeutic target for diabetes, oxidative stress, and inflammatory diseases (8).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 393112
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant human VAP‑1/AOC3
Gly27-Asn763
Accession # Q16853
Gly27-Asn763
Accession # Q16853
Specificity
Detects human VAP‑1/AOC3 in direct ELISAs and Western blots. In Western blots, 50% cross-reactivity with recombinant mouseVAP‑1/AOC3 is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human VAP‑1/AOC3 Antibody
Detection of VAP-1/AOC3 by Western Blot
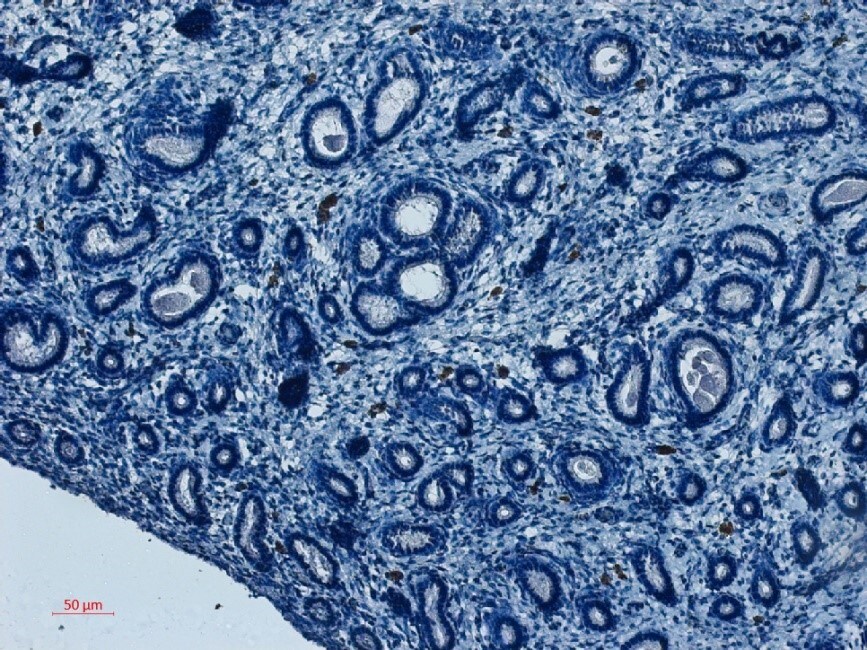
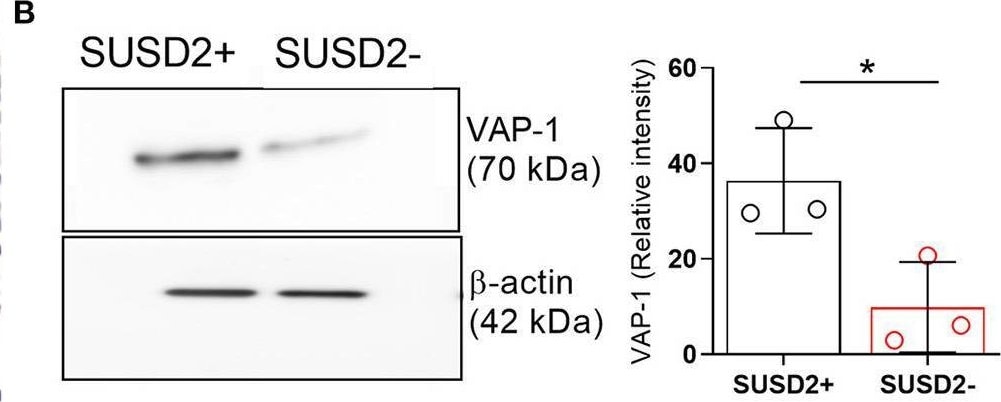
Biophysical and functional characterization of endometrial pericytes and EnSCs. (A) Immunohistochemistry showing VAP-1 expression in luteal-phase human endometrium, hematoxylin was used to stain the nuclei. Staining indicates VAP-1 reactivity in the vasculature and spiral arteries, arrow pointing to spiral artery. Scale bar: 200 μm. (B) VAP-1 expression was determined at protein level in freshly isolated SUSD2+ pericytes and SUSD2− EnSCs from three independent endometrial biopsies by Western blot analysis and normalized to beta -actin and relative intensity determined by densitometry; Student's t-test; *P < 0.05. (C) Expression of ELN (left panel), CNN1 (middle panel) and MYH11 (right panel) transcripts was determined by RT-qPCR in freshly isolated SUSD2+ pericytes and SUSD2− EnSCs from four independent endometrial biopsies; Student's t-test; *P < 0.05; **P < 0.01. (D) Representative graphs showing proliferation and migration SUSD2+ pericytes and SUSD2− EnSCs monitored in real-time using the xCELLigence system for the indicated time-points. (E) freshly isolated SUSD2+ pericytes and SUSD2− EnSCs were embedded into collagen at a density of 1 × 106 cells per gel. Single cell contraction force was measured using the depth-sensing nanoindentation system. Data represent mean ± SEM of four biological repeat experiments. Student's t-test; **P < 0.01; ****P < 0.0001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33537312), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human VAP‑1/AOC3 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Recombinant Human VAP‑1/AOC3 (Catalog # 3957-AO) under reducing conditions only
Sample: Recombinant Human VAP‑1/AOC3 (Catalog # 3957-AO) under reducing conditions only
Reviewed Applications
Read 1 review rated 3 using MAB3957 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: VAP-1/AOC3
References
- Kurkijärvi, R. et al. (1998) J. Immunol. 161:1549.
- Gearing, A.J.H. and W. Newman (1993) Immunol. Today 14:506.
- Lizcano, J.M. et al. (1998) Biochem. J. 331:69.
- Smith, D.J. et al. (1998) J. Exp. Med. 188:17.
- Jaakkala K. et al. (2000) Am. J. Pathol. 157:463.
- Salmi, M. and J. Jalkanen (2001) Trends Immunol. 22:211.
- Salmi, M. and J. Jalkanen (1996) J. Exp. Med. 183:569.
- Dunkel, P. et al. (2008) Curr. Med. Chem. 15:1827.
Long Name
Vascular Adhesion Protein-1
Alternate Names
AOC3, HPAO, SSAO, VAP1
Gene Symbol
AOC3
UniProt
Additional VAP-1/AOC3 Products
Product Documents for Human VAP‑1/AOC3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human VAP‑1/AOC3 Antibody
For research use only
Related Research Areas
Citations for Human VAP‑1/AOC3 Antibody
Customer Reviews for Human VAP‑1/AOC3 Antibody (1)
3 out of 5
1 Customer Rating
Have you used Human VAP‑1/AOC3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: EndometriumSpecies: EquineVerified Customer | Posted 02/20/2020The Antibody worked great in a Positive control (Human Lung) but no reactivity to the equine protein was possible.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...