Vascular endothelial (VE) - cadherin (VE-CAD), also called 7B4 and cadherin-5, is a member of the cadherin family of cell adhesion molecules. Cadherins are
calcium‑dependent transmembrane proteins which bind to one another in a homophilic manner. On their cytoplasmic side, they associate with the three catenins, alpha, beta, and gamma (plakoglobin). This association links the cadherin protein to the cytoskeleton. Without association with the catenins, the cadherins are non-adhesive. Cadherins play a role in development, specifically in tissue formation. They may also help to maintain tissue architecture in the adult. VE-Cadherin has been shown to play important roles in vasculogenesis and angiogenesis. VE-cadherin is a classical cadherin molecule. Classical cadherins consist of a large extracellular domain which contains DXD and DXNDN repeats responsible for mediating calcium‑dependent adhesion, a single-pass transmembrane domain, and a short carboxy-terminal cytoplasmic domain responsible for interacting with the catenins. Human VE-cadherin is a 784 amino acid (aa) residue protein with a 25 aa signal sequence and a 759 aa propeptide. The mature protein begins at amino acid 48 and has a 546 aa extracellular domain, a 27 aa transmembrane domain, and a 164 aa cytoplasmic domain. The human and mouse mature VE-CAD proteins share approximately 74% homology.
Best Seller
Human VE‑Cadherin Antibody
R&D Systems | Catalog # AF938

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Bovine, Fish - Danio rerio (Zebrafish), Zebrafish
Applications
Validated:
Western Blot, Flow Cytometry, Immunocytochemistry, Simple Western, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Immunocytochemistry/ Immunofluorescence, ELISA Capture, IF/ICC, RNAscope compatible, Westen Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human VE-Cadherin
Asp48-Gln593
Accession # P33151
Asp48-Gln593
Accession # P33151
Specificity
Detects human VE-Cadherin in direct ELISAs. In direct ELISAs, approximately 10% cross-reactivity with recombinant mouse VE‑Cadherin is observed and less than 5% cross-reactivity with recombinant human E-Cadherin is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human VE‑Cadherin Antibody
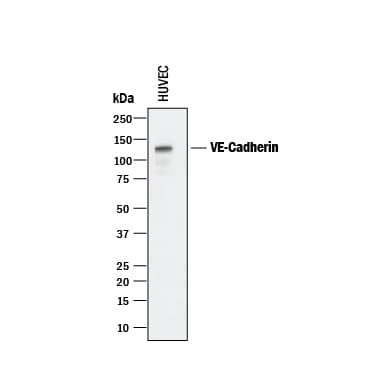
Detection of Human VE‑Cadherin by Western Blot.
Western blot shows lysate of HUVEC human umbilical vein endothelial cells. PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Human VE-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF938) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for VE-Cadherin at approximately 125 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.VE‑Cadherin in HUVEC Cells.
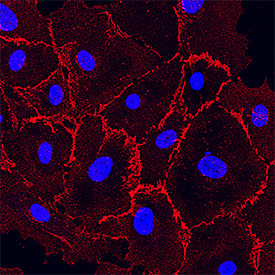
VE-Cadherin was detected in immersion fixed HUVEC human umbilical vein endothelial cells using Goat Anti-Human VE-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF938) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). Specific staining was localized to the plasma membrane. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human VE‑Cadherin by Simple WesternTM.
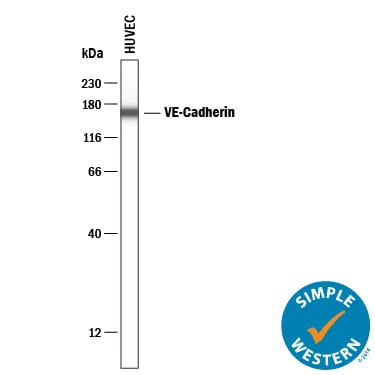
Simple Western lane view shows lysates of HUVEC human umbilical vein endothelial cells, loaded at 0.2 mg/mL. A specific band was detected for VE‑Cadherin at approximately 162 kDa (as indicated) using 12.5 µg/mL of Goat Anti-Human VE‑Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF938). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of VE-Cadherin by Western Blot
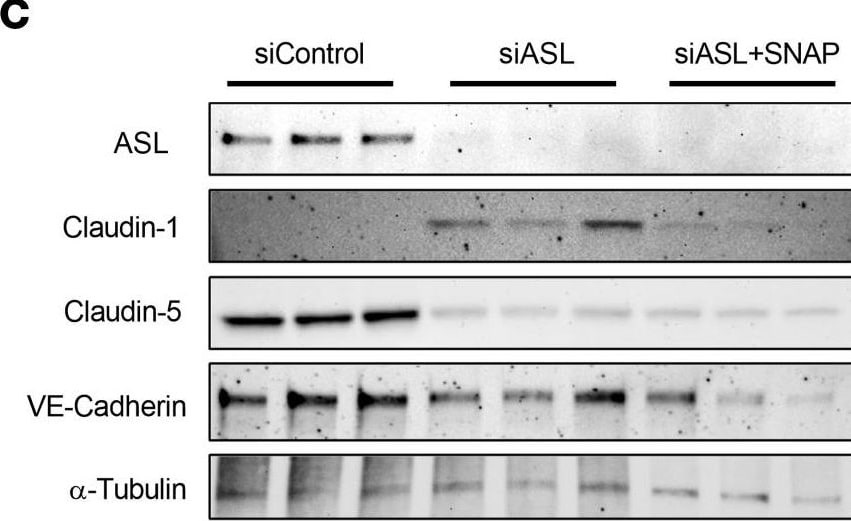
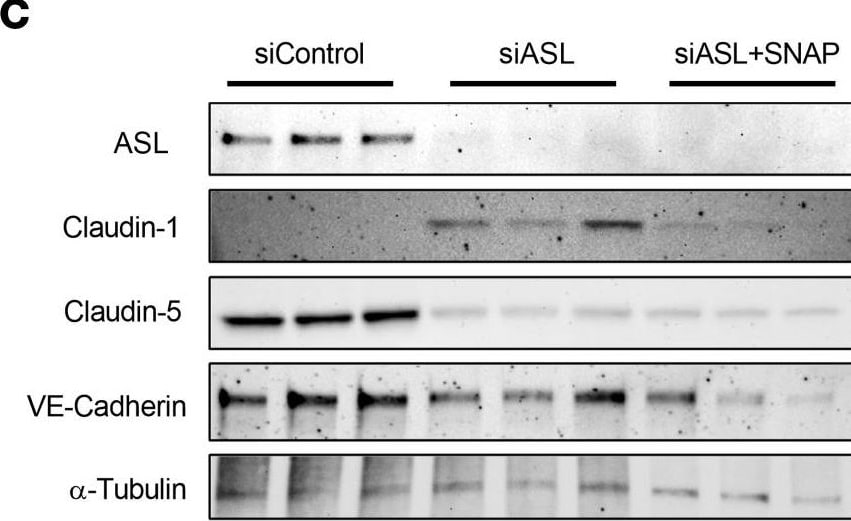
Treatment with an NO donor or Claudin-1 inhibition improved barrier integrity in ASL-deficient HBMECs.(A) TEER measurement in HBMECs treated with 0, 10, or 25 μM of S-nitroso-N-acetylpenicillamine (SNAP), an NO donor, 48 hours (hr) after subculture (n = 3). (B) Comparison of TEER levels in HBMECs treated with 25 μM of either N-acetylpenicillamine (NAP) or SNAP 48 hr after subculture. SNAP or NAP was added daily, starting 24 hr after subculture (n = 3). (C) Western blot analysis and (D) quantification investigating the effects of SNAP (25 μM) on expression of ASL, CLDN5, CLDN1, and VE-Cadherin in ASL-deficient HBMECs (n = 3). SNAP was added to the treatment group 24 hr prior to protein extraction. (E) Relative mRNA expression of CLDN1 in HBMECs 48 hr after transfection with siControl, siASL, or both siASL and siCLDN1 (n = 3). (F) Western blot analysis and (G) quantification to assess ASL, CLDN5, and CLDN1 protein level in HBMECs 48 hr after siRNA transfection (n = 3). Protein abundance was normalized to alpha -Tubulin level in D and G. (H) TEER measurement to investigate the effects of SNAP (25 μM) or siRNA-mediated inhibition of CLDN1 on paracellular barrier integrity of ASL-deficient HBMECs. Bar graphs represent mean values while error bars represent the standard deviation. *P < 0.05, **P < 0.01, and ***P < 0.001. One-way ANOVA, multiple comparisons. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37490345), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of VE-Cadherin by Western Blot
Treatment with an NO donor or Claudin-1 inhibition improved barrier integrity in ASL-deficient HBMECs.(A) TEER measurement in HBMECs treated with 0, 10, or 25 μM of S-nitroso-N-acetylpenicillamine (SNAP), an NO donor, 48 hours (hr) after subculture (n = 3). (B) Comparison of TEER levels in HBMECs treated with 25 μM of either N-acetylpenicillamine (NAP) or SNAP 48 hr after subculture. SNAP or NAP was added daily, starting 24 hr after subculture (n = 3). (C) Western blot analysis and (D) quantification investigating the effects of SNAP (25 μM) on expression of ASL, CLDN5, CLDN1, and VE-Cadherin in ASL-deficient HBMECs (n = 3). SNAP was added to the treatment group 24 hr prior to protein extraction. (E) Relative mRNA expression of CLDN1 in HBMECs 48 hr after transfection with siControl, siASL, or both siASL and siCLDN1 (n = 3). (F) Western blot analysis and (G) quantification to assess ASL, CLDN5, and CLDN1 protein level in HBMECs 48 hr after siRNA transfection (n = 3). Protein abundance was normalized to alpha -Tubulin level in D and G. (H) TEER measurement to investigate the effects of SNAP (25 μM) or siRNA-mediated inhibition of CLDN1 on paracellular barrier integrity of ASL-deficient HBMECs. Bar graphs represent mean values while error bars represent the standard deviation. *P < 0.05, **P < 0.01, and ***P < 0.001. One-way ANOVA, multiple comparisons. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37490345), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of VE-Cadherin by Western Blot
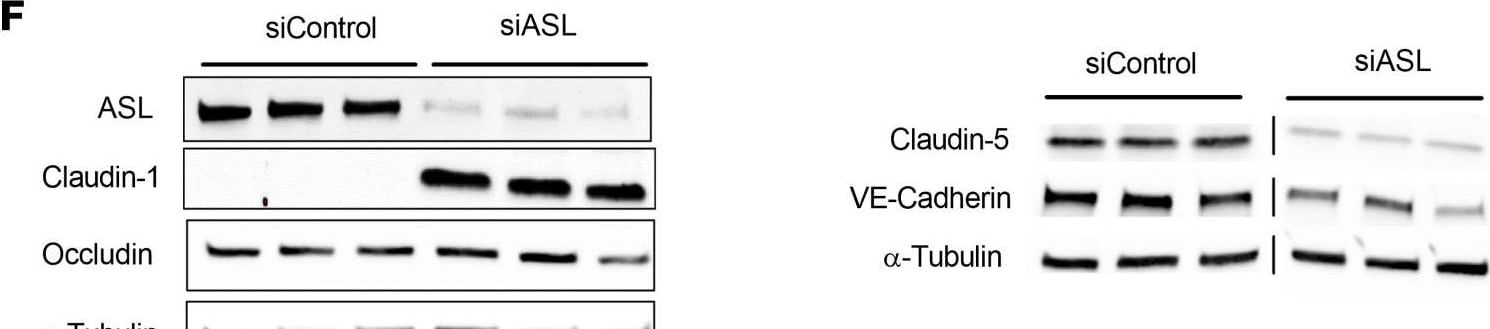
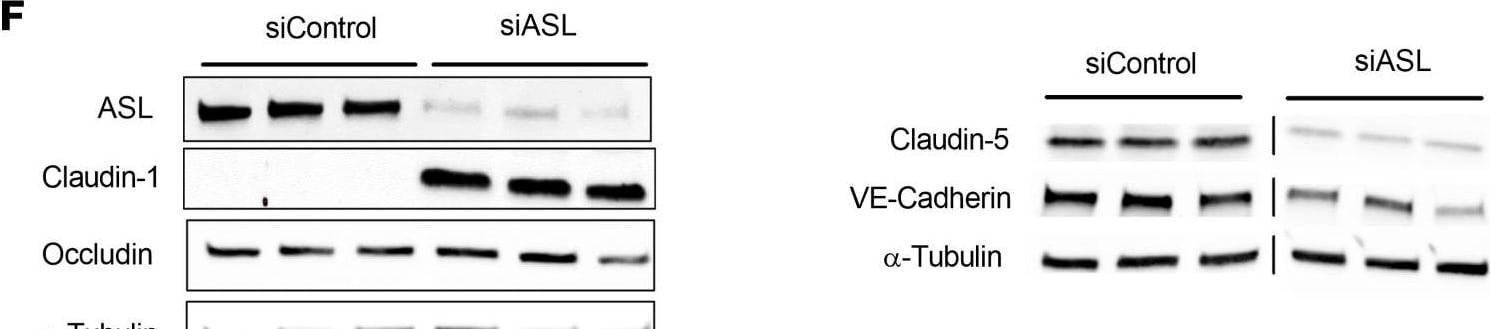
ASL knockdown in HBMECs leads to reduced NO signaling, decreased paracellular permeability, and dysregulated expression of BBB-associated genes.(A) Relative mRNA expression of ASL in HBMECs 2 days after transfection with control and ASL siRNAs (siControl and siASL) (n = 3). (B) Intracellular levels of NO in siControl and siASL-transfected HBMECs as measured by DAF-FM diacetate fluorescence assay (n = 9). (C) Intracellular cGMP levels in siControl and siASL-transfected HBMECs as measured by ELISA (n = 5). (D) Quantitative analysis of TEER level at 1–3 days after transfected HBMECs were seeded on Transwell inserts (n = 3). (E) Relative mRNA expression of CLDN5, CDH5, TJP1, OCLN, and CLDN1 in HBMECs 2 days after siRNA-mediated knockdown (n = 3). (F) Western blot analysis of ASL, CLDN1, OCLN, CLDN5, and VE-Cadherin protein levels in HBMECs 2 days after siControl and siASL transfection. Samples were run on 2 separate gels, and alpha -Tubulin was used as the internal control (n = 3). (G) Protein abundance was quantitatively measured after normalization to alpha -Tubulin level. Bar graphs represent mean values while error bars represent the standard deviation. *P < 0.05, **P < 0.01, and ***P < 0.001. Student’s t test. A.U., arbitrary units. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37490345), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of VE-Cadherin by Western Blot
ASL knockdown in HBMECs leads to reduced NO signaling, decreased paracellular permeability, and dysregulated expression of BBB-associated genes.(A) Relative mRNA expression of ASL in HBMECs 2 days after transfection with control and ASL siRNAs (siControl and siASL) (n = 3). (B) Intracellular levels of NO in siControl and siASL-transfected HBMECs as measured by DAF-FM diacetate fluorescence assay (n = 9). (C) Intracellular cGMP levels in siControl and siASL-transfected HBMECs as measured by ELISA (n = 5). (D) Quantitative analysis of TEER level at 1–3 days after transfected HBMECs were seeded on Transwell inserts (n = 3). (E) Relative mRNA expression of CLDN5, CDH5, TJP1, OCLN, and CLDN1 in HBMECs 2 days after siRNA-mediated knockdown (n = 3). (F) Western blot analysis of ASL, CLDN1, OCLN, CLDN5, and VE-Cadherin protein levels in HBMECs 2 days after siControl and siASL transfection. Samples were run on 2 separate gels, and alpha -Tubulin was used as the internal control (n = 3). (G) Protein abundance was quantitatively measured after normalization to alpha -Tubulin level. Bar graphs represent mean values while error bars represent the standard deviation. *P < 0.05, **P < 0.01, and ***P < 0.001. Student’s t test. A.U., arbitrary units. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37490345), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human VE‑Cadherin Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
2.5 µg/106 cells
Sample: HUVEC human umbilical vein endothelial cells
Sample: HUVEC human umbilical vein endothelial cells
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed HUVEC human umbilical vein endothelial cells
Sample: Immersion fixed HUVEC human umbilical vein endothelial cells
Simple Western
12.5 µg/mL
Sample: HUVEC human umbilical vein endothelial cells
Sample: HUVEC human umbilical vein endothelial cells
Western Blot
0.25 µg/mL
Sample: HUVEC human umbilical vein endothelial cells
Sample: HUVEC human umbilical vein endothelial cells
Reviewed Applications
Read 2 reviews rated 5 using AF938 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: VE-Cadherin
References
- Shimoyama, Y. et al. (1989) J. Cell Biol. 109:1787.
- Bussemakers, M.J.G. et al. (1993) Mol. Biol. Reports 17:123.
- Overduin, M. et al. (1995) Science 267:386.
- Takeichi, M. (1991) Science 251:1451.
- Nose, A. et al. (1987) EMBO J. 6:3655.
- Carmeliet, P. et al. (1999) Cell 98:147.
- Gory-Faure, S. et al. (1999) Development 126:2093.
Long Name
Vascular Endothelium Cadherin
Alternate Names
Cadherin-5, CD144, CDH5, VECadherin
Gene Symbol
CDH5
UniProt
Additional VE-Cadherin Products
Product Documents for Human VE‑Cadherin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human VE‑Cadherin Antibody
For research use only
Citations for Human VE‑Cadherin Antibody
Customer Reviews for Human VE‑Cadherin Antibody (2)
5 out of 5
2 Customer Ratings
Have you used Human VE‑Cadherin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: Simple WesternSample Tested: Lung tissueSpecies: HumanVerified Customer | Posted 11/19/2024VE-Cadherin expression in human lungs1:20 in milk-free antibody diluent. 1h of incubation in the primary antibodies.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: HMVEC human microvascular endothelial cellsSpecies: HumanVerified Customer | Posted 06/09/2021
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Blood-Brain Barrier and Immune Cell Transmigration: ICAM-1/CD54 Signaling Pathways
Blood-Brain Barrier and Immune Cell Transmigration: ICAM-1/CD54 Signaling Pathways
 Blood-Brain Barrier and Immune Cell Transmigration: Pathways Overview
Blood-Brain Barrier and Immune Cell Transmigration: Pathways Overview
 Blood-Brain Barrier and Immune Cell Transmigration: VCAM-1/CD106 Signaling Pathways
Blood-Brain Barrier and Immune Cell Transmigration: VCAM-1/CD106 Signaling Pathways
 Blood-Brain Barrier and Immune Cell Transmigration: VEGF Signaling Pathways
Blood-Brain Barrier and Immune Cell Transmigration: VEGF Signaling Pathways

Blood-Brain Barrier Pathway: Anatomy
 Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways

 Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways



_c8a7eef7-82f8-4d1e-aa32-1898ec2bbe25.png)