TNF receptor 1 (TNF RI; also called TNF R‑p55/p60, TNFRSF1A and CD120a) is a type I transmembrane protein that belongs to the TNF receptor superfamily (1, 2). TNF RI is widely expressed and is present on the cell surface as a trimer of 55 kDa subunits. It serves as a receptor for both TNF‑ alpha and TNF‑ beta /lymphotoxin. Each subunit contains four TNF‑ alpha trimer‑binding cysteine‑rich domains (CRD) in its extracellular domain (ECD) (1‑6). TNF‑ alpha binding to TNF R1 induces the sequestration of TNFRI in lipid rafts, where it activates NF kappa B and is cleaved by ADAM‑17/TACE (7, 8). Release of the 28‑34 kDa TNF RI ECD occurs constitutively, and in response to products of pathogens such as LPS, CpG DNA or S. aureus protein A (1, 7‑12). Full‑length TNF RI may also be released in exosome‑like vesicles (12). Such release helps to resolve inflammatory reactions as it down‑regulates cell surface TNF RI and provides soluble TNF RI to bind TNF‑ alpha (6, 13, 14). Exclusion from lipid rafts causes endocytosis of TNF RI complexes and induces apoptosis (7, 15). Although there is a second receptor for TNF‑ alpha (TNF R2), TNF RI is thought to mediate most of the cellular effects of TNF‑ alpha (3). TNF R1 is essential for proper development of lymph node germinal centers and Peyer’s patches, and for combating intracellular pathogens such as Listeria monocytogenes (1‑3). Mouse TNF RI is a 454 amino acid (aa) protein that contains a 21 aa signal sequence and a 191 aa ECD with a PLAD domain (6). This mediates constitutive trimer formation. The PLAD domain is followed by four CRDs, a 23 aa transmembrane domain, and a 219 aa cytoplasmic sequence that contains a neutral sphingomyelinase activation domain and a death domain (16). The ECD of mouse TNF RI shows 67%, 70%, 64%, 70% and 88% aa identity with canine, feline, procine, human and rat TNF RI, respectively; and it shows 23% aa identity with the ECD of TNF RII.
Mouse TNF RI/TNFRSF1A Antibody
R&D Systems | Catalog # AF-425-PB

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ile22-Ala212
Accession # P25118
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Mouse TNF RI/TNFRSF1A Antibody
Detection of Mouse TNF RI/TNFRSF1A by Western Blot.
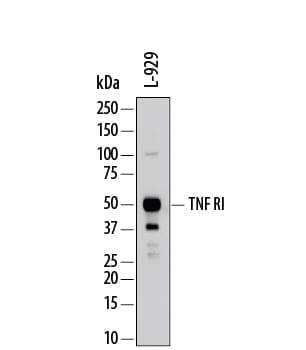
Western blot shows lysates of L-929 mouse fibroblast cell line. PVDF membrane was probed with 0.2 µg/mL of Goat Anti-Mouse TNF RI/TNFRSF1A Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-425-PB) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). A specific band was detected for TNF RI/TNFRSF1A at approximately 50-55 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of TNF RI/TNFRSF1A in L‑929 Mouse Cell Line by Flow Cytometry.
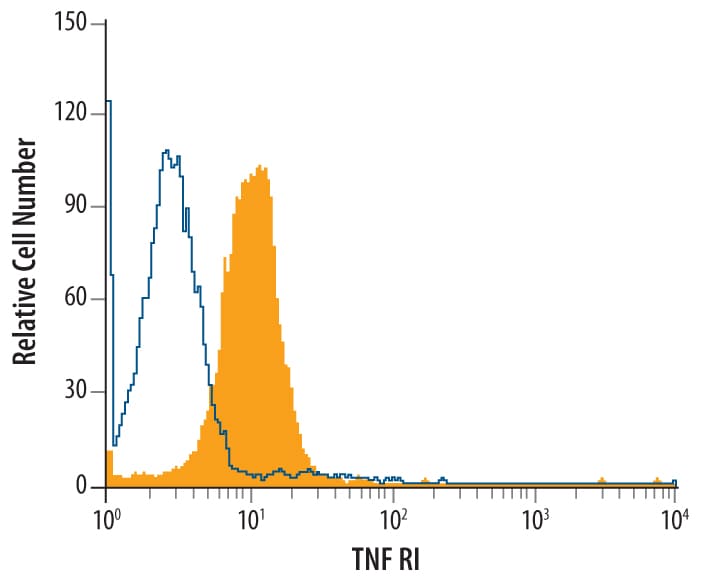
L-929 mouse fibroblast cell line was stained with Goat Anti-Mouse TNF RI/TNFRSF1A Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-425-PB, filled histo-gram) or control antibody (Catalog # AB-108-C, open histo-gram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107).Detection of Mouse TNF RI/TNFRSF1A by Western Blot
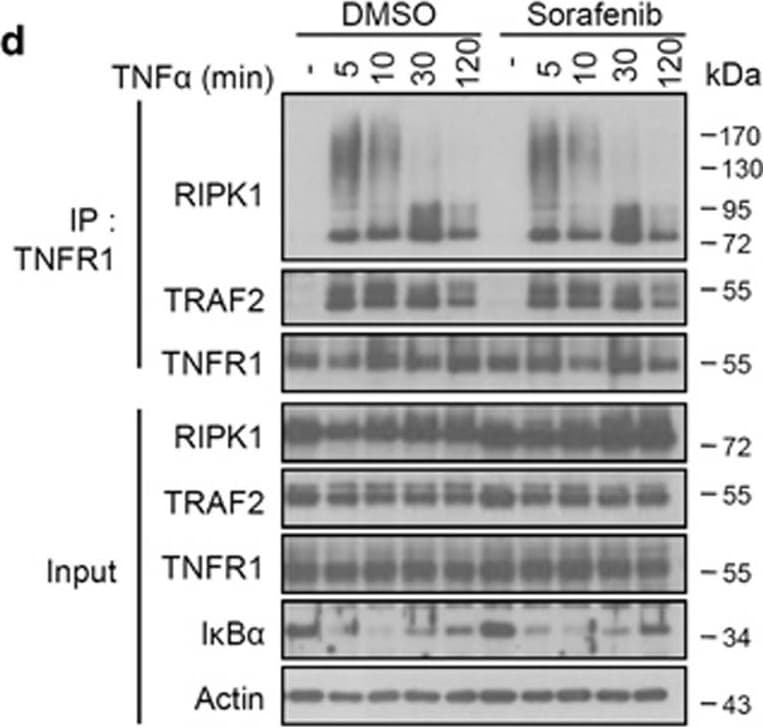
Sorafenib does not influence complex I formation, but protects against necroptosis by targeting the necrosome complex. (a) L929 cells were pretreated with 10 μM Nec-1s, Sorafenib or DMSO for 1 h and stimulated with mTNF (10 ng/ml) for the indicated time. Cells were lysed and immunoblotted with the indicated antibodies. *Phosphorylated RIPK1. (b,c) L929 cells were pretreated with 10 μM DMSO, Nec-1 or Sorafenib for 1 h and stimulated with mTNF (10 ng/ml) for 2 h. The relative mRNA levels of mA20 and mI kappa B-alpha were analyzed by qRT-PCR. All bars represent mean±S.D.; n=3, ns=non-significant. (d) L929 cells were pretreated with 10 μM Sorafenib or DMSO and stimulated with mTNF (10 ng/ml) for the indicated time. Cell lysates were immunoprecipitated with anti-mTNFR1 antibody. (e,f) L929 (e) and HT-29 (f) cells were pretreated with 10 μM Nec-1, Sorafenib (Sora) or DMSO and stimulated with zVAD (10 μM)+mTNF (5 ng/ml) (L929) or zVAD (30 μM)+BV6 (1 μM)+hTNF (30 ng/ml) (HT-29) for the time indicated. Cell lysates were immunoprecipitated with the indicated antibodies and both immunoprecipitates (IP) and total lysates (input) were immunoblotted with the indicated antibodies (WB) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/28661484), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse TNF RI/TNFRSF1A Antibody
Agonist Activity
The ED50 for this effect is typically 0.05-0.15 μg/mL.
CyTOF-ready
Flow Cytometry
Sample: L‑929 mouse fibroblast cell line
Immunohistochemistry
Sample: Perfusion fixed frozen sections of mouse intestine
Western Blot
Sample: L‑929 mouse fibroblast cell line
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: TNF RI/TNFRSF1A
References
- Pfeffer, K. (2003) Cytokine Growth Factor Rev. 14:185.
- Hehlgans, T. and K. Pfeffer (2005) Immunology 115:1.
- Peschon, J.J. et al. (1998) J. Immunol. 160:943.
- Banner, D.W et al. (1993) Cell 73:431.
- Medvedev, A.E. et al. (1996) J. Biol. Chem. 271:9778.
- Chan, F.K. et al. (2000) Science 288:2351.
- Legler, D.F. et al. (2003) Immunity 18:655.
- Tellier, E. et al. (2006) Exp. Cell Res. 312:3969.
- Xanthoulea, S. et al. (2004) J. Exp. Med. 200:367.
- Jin, L. et al. (2000) J. Immunol. 165:5153.
- Gomez, M.I. et al. (2006) J. Biol. Chem. 281:20190.
- Islam, A. et al. (2006) J. Biol. Chem. 281:6860.
- Garton, K.J. et al. (2006) J. Leukoc. Biol. 79:1105.
- McDermott, M.F. et al. (1999) Cell 97:133.
- Schneider-Brachert, W. et al. (2004) Immunity 21:415.
- Lewis, M. et al. (1991) Proc. Natl. Acad. Sci. USA 88:2830.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional TNF RI/TNFRSF1A Products
Product Documents for Mouse TNF RI/TNFRSF1A Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse TNF RI/TNFRSF1A Antibody
For research use only
Citations for Mouse TNF RI/TNFRSF1A Antibody
Customer Reviews for Mouse TNF RI/TNFRSF1A Antibody
There are currently no reviews for this product. Be the first to review Mouse TNF RI/TNFRSF1A Antibody and earn rewards!
Have you used Mouse TNF RI/TNFRSF1A Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions
TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions