Tissue inhibitors of metalloproteinases (TIMPs) are a family of proteins that regulate the activation and proteolytic activity of the zinc enzymes known as matrix metalloproteinases (MMPs). There are four members of the family: TIMP-1, TIMP-2, TIMP-3, and TIMP-4. TIMP-1 is a glycoprotein with a molecular mass of 32‑34 kDa produced by a wide range of cell types. TIMP-1 inhibits active MMP-mediated proteolysis by forming an N-terminal, non-covalent binary complex with the MMP active site. TIMP-1 also associates C-terminally with pro-MMP-9 in a complex which may play a role in regulating activation. Independent of MMPs, TIMP-1 has been shown to have a role in tissue homeostasis.

Key Product Details
Species Reactivity
Validated:
Rat
Cited:
Mouse, Rat
Applications
Validated:
Immunohistochemistry, Western Blot, Immunoprecipitation
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant rat TIMP-1
Cys24-Ala217
Accession # P30120
Cys24-Ala217
Accession # P30120
Specificity
Detects rat TIMP-1 in direct ELISAs and Western blots. In direct ELISAs, approximately 40% cross-reactivity with recombinant human (rh) TIMP‑1 and recombinant mouse TIMP-1 is observed and less than 1% cross-reactivity with rhTIMP-2, rhTIMP-3, and rhTIMP-4 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Rat TIMP‑1 Antibody
Detection of Rat TIMP-1 by Western Blot
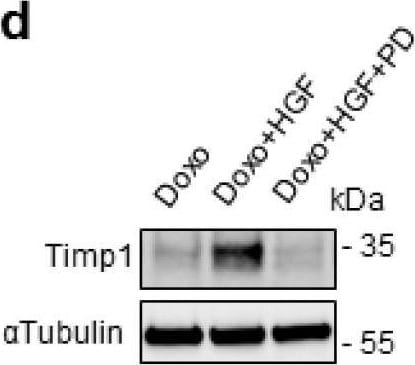
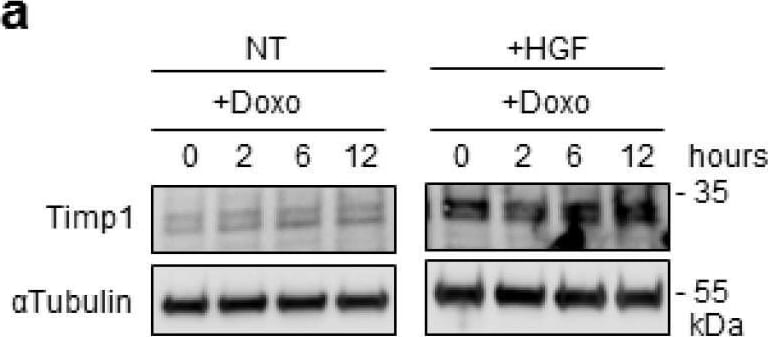
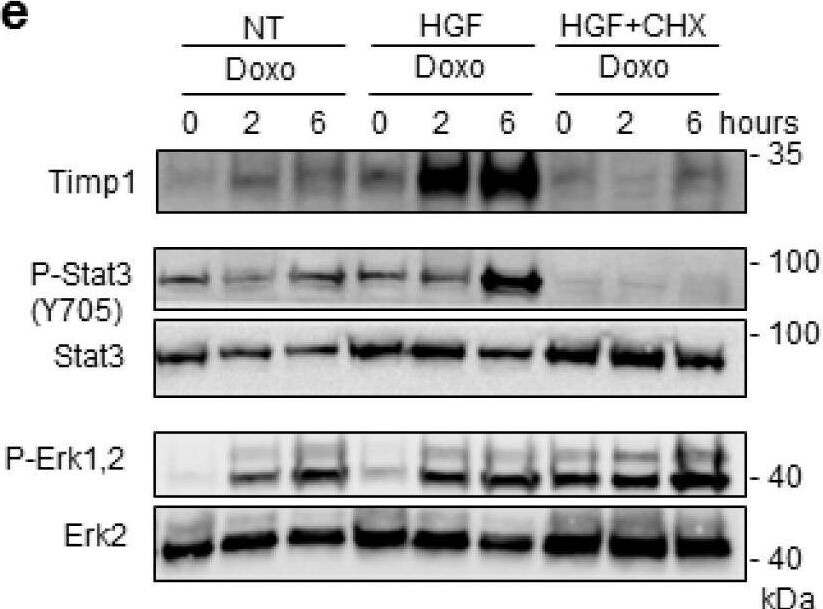
Preconditioning with HGF induces Timp1 (tissue inhibitor of metalloproteinases-1) protein synthesis, which is followed by Stat3 phosphorylation and protection against Doxo-induced apoptosis. (a) H9c2 cardiomyoblasts were treated with Doxo (25 μM) or Doxo+HGF (0.5 nM). For cell treatments, see Figure 1a. Timp1 protein levels were evaluated by Western blotting at different points of recovery time (a). Cells were also treated with (b) 500 nM JNJ38877605 Met inhibitor (Doxo+HGF+JNJ) or (c,d) 1 µM PD98059 Erk1,2 inhibitor (Doxo+HGF+PD). Protein (b,d) and mRNA (c) levels of Timp1 were analyzed after 24 h of recovery time. Polr2a was used as reference gene for the expression data normalization. *** p < 0.005 significantly different from Doxo-treated cells. (e,f) H9c2 cells were pretreated with HGF alone, or with HGF+Cycloheximide (CHX, 10 μM) for 4 h. Then, Doxo (25 μM) was added in the last 1 h. The CHX treatment was performed 30 min before adding HGF and was maintained during all the treatment protocol. Protein levels of Timp1, P-Stat3 (Y705), Stat3, P-Erk1,2, Erk2 (e) and total and cleaved caspase 3 (f) were detected at different time points of recovery time (0, 2 and 6 h). (g,h) H9c2 cells were treated with Doxo (25 μM), Doxo+HGF (0.5 nM) or Doxo+HGF+Timp1 siRNA. Protein levels of Timp1, P-Stat3 (Y705), Stat3, P-Erk1,2 and Erk2 (g) and gamma H2AX, H2AX and cleaved/total caspase 3 ratios (h) were measured after 24 h of recovery time. The ratios calculated between cleaved and total caspase 3 are shown. alpha tubulin was used as the loading control in all Western blots. Data are representative results of three independent experimental replicates. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32722178), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of TIMP‑1 in Rat Brain.
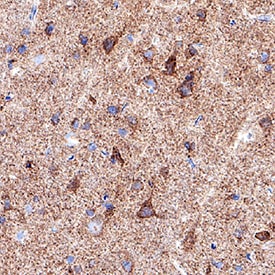
TIMP‑1 was detected in perfusion fixed paraffin-embedded sections of Rat Brain using Goat Anti-Rat TIMP‑1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF580) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using VisUCyte Antigen Retrieval Reagent-Basic (Catalog # VCTS021). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to neuronal cell bodies. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Rat TIMP-1 by Western Blot
Preconditioning with HGF induces Timp1 (tissue inhibitor of metalloproteinases-1) protein synthesis, which is followed by Stat3 phosphorylation and protection against Doxo-induced apoptosis. (a) H9c2 cardiomyoblasts were treated with Doxo (25 μM) or Doxo+HGF (0.5 nM). For cell treatments, see Figure 1a. Timp1 protein levels were evaluated by Western blotting at different points of recovery time (a). Cells were also treated with (b) 500 nM JNJ38877605 Met inhibitor (Doxo+HGF+JNJ) or (c,d) 1 µM PD98059 Erk1,2 inhibitor (Doxo+HGF+PD). Protein (b,d) and mRNA (c) levels of Timp1 were analyzed after 24 h of recovery time. Polr2a was used as reference gene for the expression data normalization. *** p < 0.005 significantly different from Doxo-treated cells. (e,f) H9c2 cells were pretreated with HGF alone, or with HGF+Cycloheximide (CHX, 10 μM) for 4 h. Then, Doxo (25 μM) was added in the last 1 h. The CHX treatment was performed 30 min before adding HGF and was maintained during all the treatment protocol. Protein levels of Timp1, P-Stat3 (Y705), Stat3, P-Erk1,2, Erk2 (e) and total and cleaved caspase 3 (f) were detected at different time points of recovery time (0, 2 and 6 h). (g,h) H9c2 cells were treated with Doxo (25 μM), Doxo+HGF (0.5 nM) or Doxo+HGF+Timp1 siRNA. Protein levels of Timp1, P-Stat3 (Y705), Stat3, P-Erk1,2 and Erk2 (g) and gamma H2AX, H2AX and cleaved/total caspase 3 ratios (h) were measured after 24 h of recovery time. The ratios calculated between cleaved and total caspase 3 are shown. alpha tubulin was used as the loading control in all Western blots. Data are representative results of three independent experimental replicates. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32722178), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Rat TIMP-1 by Western Blot
Preconditioning with HGF induces Timp1 (tissue inhibitor of metalloproteinases-1) protein synthesis, which is followed by Stat3 phosphorylation and protection against Doxo-induced apoptosis. (a) H9c2 cardiomyoblasts were treated with Doxo (25 μM) or Doxo+HGF (0.5 nM). For cell treatments, see Figure 1a. Timp1 protein levels were evaluated by Western blotting at different points of recovery time (a). Cells were also treated with (b) 500 nM JNJ38877605 Met inhibitor (Doxo+HGF+JNJ) or (c,d) 1 µM PD98059 Erk1,2 inhibitor (Doxo+HGF+PD). Protein (b,d) and mRNA (c) levels of Timp1 were analyzed after 24 h of recovery time. Polr2a was used as reference gene for the expression data normalization. *** p < 0.005 significantly different from Doxo-treated cells. (e,f) H9c2 cells were pretreated with HGF alone, or with HGF+Cycloheximide (CHX, 10 μM) for 4 h. Then, Doxo (25 μM) was added in the last 1 h. The CHX treatment was performed 30 min before adding HGF and was maintained during all the treatment protocol. Protein levels of Timp1, P-Stat3 (Y705), Stat3, P-Erk1,2, Erk2 (e) and total and cleaved caspase 3 (f) were detected at different time points of recovery time (0, 2 and 6 h). (g,h) H9c2 cells were treated with Doxo (25 μM), Doxo+HGF (0.5 nM) or Doxo+HGF+Timp1 siRNA. Protein levels of Timp1, P-Stat3 (Y705), Stat3, P-Erk1,2 and Erk2 (g) and gamma H2AX, H2AX and cleaved/total caspase 3 ratios (h) were measured after 24 h of recovery time. The ratios calculated between cleaved and total caspase 3 are shown. alpha tubulin was used as the loading control in all Western blots. Data are representative results of three independent experimental replicates. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32722178), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Rat TIMP‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed paraffin-embedded sections of Rat Brain.
Sample: Perfusion fixed paraffin-embedded sections of Rat Brain.
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Recombinant Rat TIMP‑1 (Catalog # 580-RT), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Recombinant Rat TIMP‑1 (Catalog # 580-RT), see our available Western blot detection antibodies
Western Blot
0.1 µg/mL
Sample: Recombinant Rat TIMP‑1 (Catalog # 580-RT)
Sample: Recombinant Rat TIMP‑1 (Catalog # 580-RT)
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: TIMP-1
Long Name
Tissue Inhibitors of Metalloproteinases 1
Alternate Names
TIMP1
Gene Symbol
TIMP1
UniProt
Additional TIMP-1 Products
Product Documents for Rat TIMP‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Rat TIMP‑1 Antibody
For research use only
Related Research Areas
Citations for Rat TIMP‑1 Antibody
Customer Reviews for Rat TIMP‑1 Antibody
There are currently no reviews for this product. Be the first to review Rat TIMP‑1 Antibody and earn rewards!
Have you used Rat TIMP‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...