100 results for "SLC6A3/DAT1" in Products
SLC6A3/DAT1 Products
The dopamine transporter (DAT) is a 70 - 80 kDa member of the Na+-neurotransmitter symporter family of transmembrane (TM) proteins. DAT plays a crucial role in the synaptic clearance of dopamine (DA). It mediates the uptake of dopamine by the presynaptic terminal, thus limiting the strength of the dopamingeric response. One molecule of DA is accompanied by two Na+ and one Cl- ion. Molecules such as amphetamine both competitively inhibit DA uptake, and induce DA release through the DAT, ...
| Reactivity: | Mouse, Rat, Human (Negative) |
| Details: | Mouse IgG1 Monoclonal Clone #mAb16 |
| Applications: | IHC, WB, ELISA, ICC/IF, IP |
| Reactivity: | Mouse, Rat, Human (Negative) |
| Details: | Mouse IgG1 Monoclonal Clone #mAb16 |
| Applications: | IHC, WB, ELISA, ICC/IF, IP |
| Reactivity: | Human, Mouse, Rat, Golden Syrian Hamster, Hamster, +1 More |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
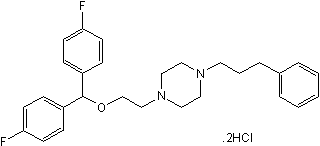
Selective DA uptake inhibitor; also σ ligand
| Chemical Name: | 1-[2-[Bis-(4-fluorophenyl)methoxy]ethyl]-4-(3-phenylpropyl)piperazine dihydrochloride |
| Purity: | ≥98% |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #CL3123 |
| Applications: | IHC |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #CL3123 |
| Applications: | IHC |
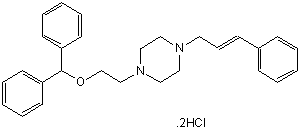
Potent and selective DA uptake inhibitor
| Chemical Name: | 1-(2-Diphenylmethoxyethyl)-4-(3-phenyl-2-propenyl)-piperazine dihydrochloride |
| Purity: | ≥98% |
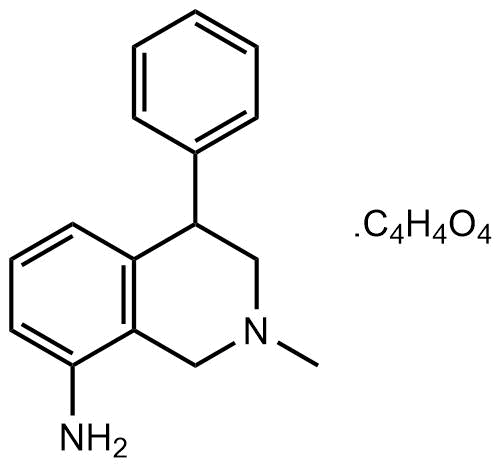
Potent noradrenalin and dopamine uptake inhibitor. Antidepressant
| Chemical Name: | 1,2,3,4-Tetrahydro-2-methyl-4-phenyl-8-isoquinolinamine maleate |
| Purity: | ≥98% |
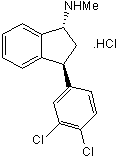
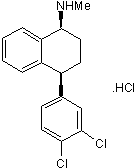
Potent 5-HT uptake inhibitor; also inhibits dopamine and noradrenalin uptake
| Alternate Names: | Lu 19-005 |
| Chemical Name: | (1R,3S)-rel-3-(3,4-Dichlorophenyl)-2,3-dihydro-N-methyl-1H-inden-1-amine hydrochloride |
| Purity: | ≥98% |
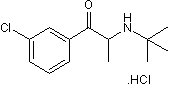
Non-selective inhibitor of dopamine and noradrenalin transporters
| Chemical Name: | 1-(3-Chlorophenyl)-2-[(1,1-dimethylethyl)amino]-1-propanone hydrochloride |
| Purity: | ≥98% |
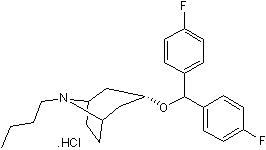
High affinity dopamine uptake inhibitor
| Chemical Name: | (3-endo)-3-[Bis(4-fluorophenyl)methoxy]-8-butyl-8-azabicyclo[3.2.1]octane hydrochloride |
| Purity: | ≥98% |
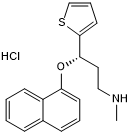
Potent 5-HT and NA reuptake inhibitor (SNRI); also blocks dopamine reuptake
| Chemical Name: | (+)-(S)-N-Methyl-3-(1-naphthyloxy)-3-(2-thienyl)propanamine hydrochloride |
| Purity: | ≥98% |
| Reactivity: | Human |
| Reactivity: | Human |
| Reactivity: | Human, Primate |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
5-HT reuptake inhibitor
| Chemical Name: | (1S,4S)-4-(3,4-Dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine hydrochloride |
| Purity: | ≥98% |
| Reactivity: | Mouse |
| Reactivity: | Human |
| Source: | Wheat germ |
| Applications: | WB, ELISA, AP, MA, PAGE |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #SR1877 |
| Applications: | WB, Flow, IP |
| Reactivity: | Human |
| Sensitivity: | 0.10 ng/mL |
| Assay Range: | 0.16 - 10 ng/mL |
| Applications: | ELISA |
| Reactivity: | Mouse |
| Sensitivity: | 4.69 pg/mL |
| Assay Range: | 7.81 - 500 pg/mL |
| Applications: | ELISA |
| Reactivity: | Rat |
| Sensitivity: | 0.19 ng/mL |
| Assay Range: | 0.31 - 20 ng/mL |
| Applications: | ELISA |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
| Reactivity: | Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |
| Reactivity: | Human |
| Applications: | WB |


![Western Blot: SLC6A3/DAT1 Antibody (mAb16) [NBP2-22164] Western Blot: SLC6A3/DAT1 Antibody (mAb16) [NBP2-22164]](https://resources.rndsystems.com/images/products/SLC6A3-DAT1-Antibody-mAb16-Western-Blot-NBP2-22164-img0005.jpg)
![Western Blot: SLC6A3/DAT1 Antibody (mAb16)Azide and BSA Free [NBP2-80954] Western Blot: SLC6A3/DAT1 Antibody (mAb16)Azide and BSA Free [NBP2-80954]](https://resources.rndsystems.com/images/products/SLC6A3-DAT1-Antibody-mAb16-Azide-and-BSA-Free-Western-Blot-NBP2-80954-img0002.jpg)
![Western Blot: SLC6A3/DAT1 Antibody [NB300-254] Western Blot: SLC6A3/DAT1 Antibody [NB300-254]](https://resources.rndsystems.com/images/products/SLC6A3-DAT1-Antibody-Western-Blot-NB300-254-img0005.jpg)

![Immunohistochemistry-Paraffin: SLC6A3/DAT1 Antibody (CL3123) [NBP2-46649] Immunohistochemistry-Paraffin: SLC6A3/DAT1 Antibody (CL3123) [NBP2-46649]](https://resources.rndsystems.com/images/products/SLC6A3-DAT1-Antibody-3123-Immunohistochemistry-NBP2-46649-img0007.jpg)
![Immunohistochemistry-Paraffin: SLC6A3/DAT1 Antibody [NBP3-43803] SLC6A3/DAT1 Antibody (CL3123) - Azide and BSA Free Immunohistochemistry-Paraffin: SLC6A3/DAT1 Antibody [NBP3-43803]](https://resources.rndsystems.com/images/products/nbp3-43803_mouse-slc6a3-dat1-mab-cl3123-azide-and-bsa-free-282202512145338.jpg)


![Western Blot: SLC6A3/DAT1 Antibody [NB300-255] Western Blot: SLC6A3/DAT1 Antibody [NB300-255]](https://resources.rndsystems.com/images/products/DAT1-Antibody-Western-Blot-NB300-255-img0001.jpg)
![SDS-PAGE: Recombinant Human SLC6A3/DAT1 GST (N-Term) Protein [H00006531-Q01] SDS-PAGE: Recombinant Human SLC6A3/DAT1 GST (N-Term) Protein [H00006531-Q01]](https://resources.rndsystems.com/images/products/qc_test-H00006531-Q01-1.jpg)
![Western Blot: SLC6A3/DAT1 Antibody (SR1877) [NBP3-21721] -](https://resources.rndsystems.com/images/products/nbp3-21721_rabbit-slc6a3-dat1-mab-sr1877-5720231754825.jpg)
![ELISA: Human SLC6A3/DAT1 ELISA Kit (Colorimetric) [NBP2-67271] - Human SLC6A3/DAT1 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-67271_human-slc6a3-dat1-elisa-kit-colorimetric-132202416225253.jpg)
![ELISA: Mouse SLC6A3/DAT1 ELISA Kit (Colorimetric) [NBP2-67272] - Mouse SLC6A3/DAT1 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-67272_mouse-slc6a3-dat1-elisa-kit-colorimetric-13220241626119.jpg)
![ELISA: Rat SLC6A3/DAT1 ELISA Kit (Colorimetric) [NBP2-67273] - Rat SLC6A3/DAT1 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-67273_rat-slc6a3-dat1-elisa-kit-colorimetric-132202416184888.jpg)
![Immunohistochemistry-Paraffin: SLC6A3/DAT1 Antibody [NBP2-68583] Immunohistochemistry-Paraffin: SLC6A3/DAT1 Antibody [NBP2-68583]](https://resources.rndsystems.com/images/products/SLC6A3-DAT1-Antibody-Immunohistochemistry-NBP2-68583-img0001.jpg)
![Western Blot: SLC6A3/DAT1 [p Thr53] Antibody [NBP2-29533] Western Blot: SLC6A3/DAT1 [p Thr53] Antibody [NBP2-29533]](https://resources.rndsystems.com/images/products/DAT1-[p-Thr53]-Antibody-Western-Blot-NBP2-29533-img0001.jpg)