Interleukin 8 (IL-8), also named monocyte-derived neutrophil chemotactic factor (MDNCF), neutrophil-activating protein 1 (NAP-1), neutrophil-activating factor (NAF) and granulocyte chemotactic peptide (GCP), belongs to the Glu-Leu-Arg motif containing (ELR+) CXC chemokine family and has been designated CXCL8. IL-8 is a potent neutrophil chemoattractant that recruits neutrophils to sites of inflammation. IL-8 also activates neutrophil functions and through a poorly understood mechanism, promotes angiogenesis. The biological activites of IL-8 is mediated by two types of G protein-coupled chemokine receptors, CXCR1 and CXCR2. In normal tissues, IL-8 expression and secretion is barely detectable. Upon stimulation by a wide range of pro-inflammatory signals including exposure to IL-1, TNF, bacterial or viral products, IL-8 production is rapidly induced in many different cell types. Secreted IL-8 is not glycosylated but has N-terminal sequence heterogenecity due to proteolytic processing. In human, two major forms, the 72 amino acid (aa) monocyte-derived IL-8 and the 77 aa endothelial IL-8 have been identified. Whereas the 72 aa isoform is a more potent chemoattractant, only the 77 aa isoform can induce apoptosis in leukemic cells. The N-terminal pentapeptide in the 77 aa isoform has been identified as the active site for the IL-8 apoptotic activity. Canine IL-8 encodes a 101 aa precursor protein with a putative 22 aa signal peptide. It shares 77% and 87% aa sequence identity with human and porcine IL-8, respectively. Similar to human IL-8, recombinant canine IL-8 also undergoes
N‑terminal processing. Two major peptides (the 79 aa and 74 aa variants that differ by an analogous N-terminal pentapeptide) are present in the recombinant canine IL-8 preparations.

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Canine
Cited:
Canine
Applications
Validated:
Western Blot, Neutralization, Immunocytochemistry
Cited:
Western Blot, Bioassay
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 258911
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant canine CXCL8/IL-8
Ala23-Pro101
Accession # P41324
Ala23-Pro101
Accession # P41324
Specificity
Detects canine CXCL8/IL-8 in direct ELISAs and Western blots. Does not cross-react with recombinant
human CXCL1, 2, 3, 5, 6, 7, 8, 9, 10, 11, CXCL12/SDF-1 alpha, 12/SDF-1 beta, 13, 16,
recombinant mouse CXCL1, 6, 9, 10, CXCL12/SDF-1 alpha, 13, recombinant rat (rr) CXCL1, CXCL3/CINC-2 alpha,
rrCXCL3/CINC-2 beta, or recombinant porcine CXCL8.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Canine IL-8/CXCL8 Antibody
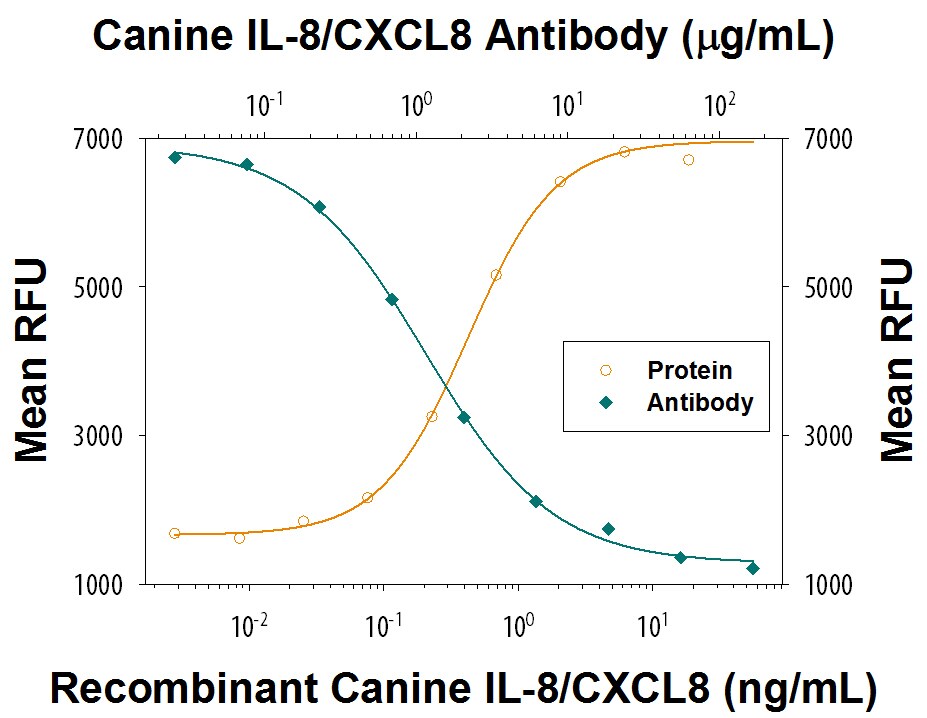
Chemotaxis Induced by IL-8/CXCL8 and Neutralization by Canine IL-8/CXCL8 Antibody.
Recombinant Canine IL-8/CXCL8 (Catalog # 1608-CL) chemoattracts the BaF3 mouse pro-B cell line transfected with human CXCR2 in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin (Catalog # AR002). Chemotaxis elicited by Recombinant Canine IL-8/CXCL8 (10 ng/mL) is neutralized (green line) by increasing concentrations of Mouse Anti-Canine IL-8/CXCL8 Monoclonal Antibody (Catalog # MAB1608). The ND50 is typically 0.3-1.2 µg/mL.IL-8/CXCL8 in Canine PBMCs.
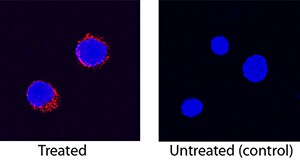
IL-8/CXCL8 was detected in immersion fixed canine peripheral blood mononuclear cells (PBMCs) treated with calcium ionomycin and PMA using Mouse Anti-Canine IL-8/CXCL8 Monoclonal Antibody (Catalog # MAB1608) at 25 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Canine IL-8/CXCL8 Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed canine peripheral blood mononuclear cells (PBMCs) treated with calcium ionomycin and PMA
Sample: Immersion fixed canine peripheral blood mononuclear cells (PBMCs) treated with calcium ionomycin and PMA
Western Blot
1 µg/mL
Sample: Recombinant Canine IL-8/CXCL8 (Catalog # 1608-CL)
Sample: Recombinant Canine IL-8/CXCL8 (Catalog # 1608-CL)
Neutralization
Measured by its ability to neutralize IL-8/CXCL8-induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human CXCR2. The Neutralization Dose (ND50) is typically 0.3-1.2 µg/mL in the presence of 10 ng/mL Recombinant Canine IL-8/CXCL8.
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-8/CXCL8
References
- Van Damme, J. et al. (1998) in The Cytokine Handbook, A.W. Thomson, ed., Academic Press, New York., p. 271.
- Terui, Y. et al. (1998) Blood 92:2672.
- Terui, Y. et al. (1999) Cancer Research 59:5651.
Long Name
Interleukin 8
Alternate Names
CXCL8, GCP1, IL8, LAI, LECT, LUCT, LYNAP, MDNCF, MONAP, NAF, NAP1, NCF, TCF, TSG1
Gene Symbol
CXCL8
UniProt
Additional IL-8/CXCL8 Products
Product Documents for Canine IL-8/CXCL8 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Canine IL-8/CXCL8 Antibody
For research use only
Customer Reviews for Canine IL-8/CXCL8 Antibody
There are currently no reviews for this product. Be the first to review Canine IL-8/CXCL8 Antibody and earn rewards!
Have you used Canine IL-8/CXCL8 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars