Cathepsin L is a lysosomal cysteine protease expressed in most eukaryotic cells. Cathepsin L is known to hydrolyze a number of proteins, including the proform of urokinase-type plasminogen activator, which is activated by Cathepsin L cleavage (1). Cathepsin L has also been shown to proteolytically inactivate alpha 1-antitrypsin and secretory leucoprotease inhibitor, two major protease inhibitors of the respiratory tract (2). These observations, combined with the demonstration of increased Cathepsin L activity in the epithelial lining fluid of the lungs of emphysema patients, have led to the suggestion that the enzyme may be involved in the progression of this disease. Cathepsin L has also been identified as a major excreted protein of transformed fibroblasts, indicating the enzyme could be involved in malignant tumor growth (3). Human Cathepsin L activity is greatest under mildly acidic conditions, from pH 4.5‑6.5. The stability of the enzyme decreases at higher pH values.
Human Cathepsin L Antibody
R&D Systems | Catalog # AF952


Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Avian - Chicken, Xenograft
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Immunoprecipitation
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Cathepsin L
Glu113-Val333
Accession # P07711
Glu113-Val333
Accession # P07711
Specificity
Detects human Cathepsin L in ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Cathepsin L Antibody
Detection of Recombinant Human Cathepsin L by Western Blot.
Western blot shows 10 ng of Recombinant Human Cathepsin L (952-CY), Recombinant Mouse Cathepsin L (1515-CY), Recombinant Human Cathepsin V (1080-CY), Recombinant Human Cathepsin K, Recombinant Human Cathepsin S (1183-CY), Recombinant Human Cathepsin H (7516-CY), and Recombinant Human Cathepsin F. PVDF Membrane was probed with 0.1 µg/mL of Goat Anti-Human Cathepsin L Antigen Affinity-purified Polyclonal Antibody (Catalog # AF952) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF109). A specific band was detected for Cathepsin L at approximately 35 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 3.Cathepsin L in Human Kidney.
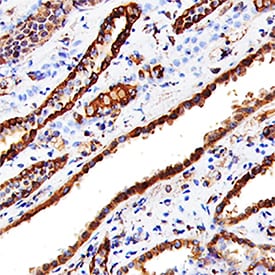
Cathepsin L was detected in immersion fixed paraffin-embedded sections of human kidney using Goat Anti-Human Cathepsin L Antigen Affinity-purified Polyclonal Antibody (Catalog # AF952) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to convoluted tubules. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Cathepsin L by Western Blot
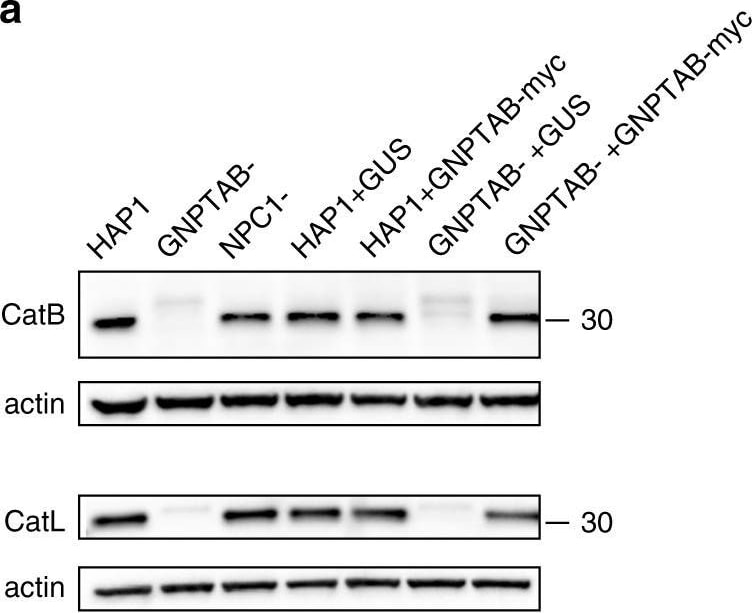
Cathepsins are reduced in GNPTAB- knockout cells, but restored upon GNPTAB reconstitution. a Immunoblotting of lysates from parental, knockout and reconstituted cells for CatB and CatL. The migration of a 30 kDa molecular mass marker is shown to the right. b CatB activity in lysates from parental HAP1, or knockout GNPTAB- or NPC1-cells. Cells were treated with cathepsin B inhibitor (Bi), cathepsin L inhibitor (Li), at 10 or 1 µM, or DMSO vehicle control for 1 h at 37 °C, before lysis and incubation with fluorescent CatB peptide substrate. After 1 h incubation at room temperature, fluorescence was measured. Data represent the mean ± s.d. of three technical replicates. A representative of three independent experiments is shown. c CatB activity in lysates from reconstituted cells. Lysates from transduced HAP1 or GNPTAB− cells were generated and tested for CatB activity, as for panel b. Data represent the mean ± s.d. of three technical replicates. A representative of two independent experiments is shown. For both panels b and c, statistical analysis was performed with a two-tailed Student’s t-test with significance shown as **** P ≤ 0.0001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/30655525), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Cathepsin L by Western Blot
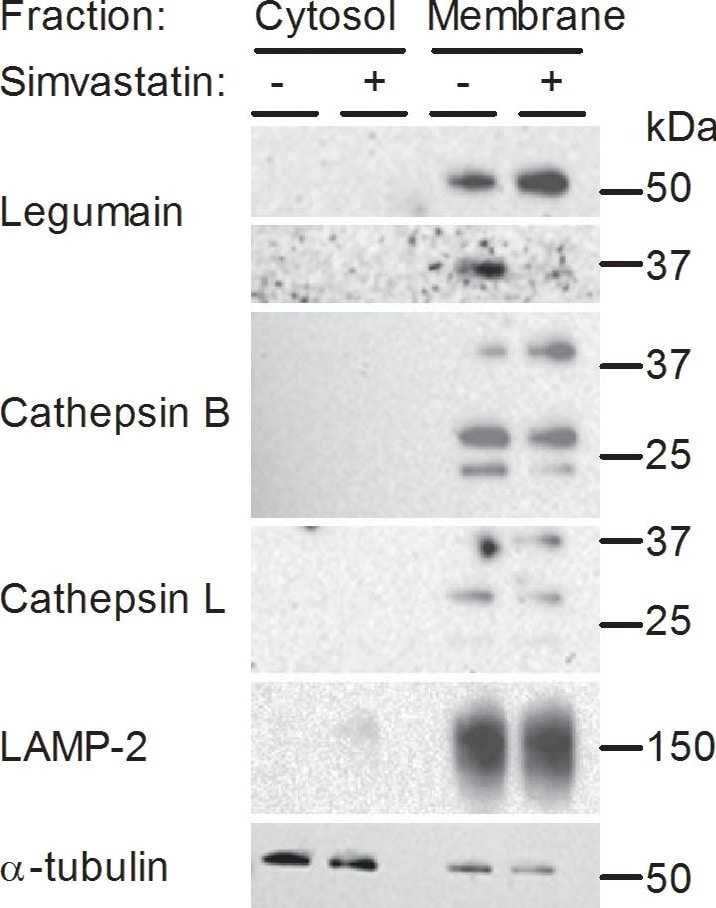
Legumain, cathepsin B and L expressions in cytosolic and membrane fractions of myotubes.Myotubes were treated with (+) or without (−) 30 µM simvastatin for 48 h before subcellular fractionation was performed. One representative immunoblot of legumain, cathepsin B and L in the cytosolic and membrane fractions is shown. All lanes were loaded with equal amount of total proteins and probed with antibodies as indicated. LAMP-2 and alpha -tubulin are shown as cell compartment controls (n = 3). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24416446), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Cathepsin L Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human kidney
Sample: Immersion fixed paraffin-embedded sections of human kidney
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Recombinant Human Cathepsin L (Catalog # 952-CY), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Recombinant Human Cathepsin L (Catalog # 952-CY), see our available Western blot detection antibodies
Western Blot
0.1 µg/mL
Sample: Recombinant Human Cathepsin L (Catalog # 952-CY)
Sample: Recombinant Human Cathepsin L (Catalog # 952-CY)
Human Cathepsin L Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cathepsin L
References
- Goretzki, L. et al. (1992) FEBS Lett. 297:112.
- Taggart, C.C. et al. (2001) J. Biol. Chem. 276:33345.
- Gottesman, M.M. and F. Cabral (1981) Biochemistry 20:1659.
Alternate Names
CTSL, CTSL1
Gene Symbol
CTSL
UniProt
Additional Cathepsin L Products
Product Documents for Human Cathepsin L Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Cathepsin L Antibody
For research use only
Related Research Areas
Citations for Human Cathepsin L Antibody
Customer Reviews for Human Cathepsin L Antibody
There are currently no reviews for this product. Be the first to review Human Cathepsin L Antibody and earn rewards!
Have you used Human Cathepsin L Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...