Human CXCR2/IL-8RB Antibody
R&D Systems | Catalog # MAB331
Clone 48311 was used by HLDA to establish CD designation

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Guinea Pig, Rabbit
Applications
Validated:
Immunohistochemistry, Neutralization, Flow Cytometry, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, Bioassay, ELISA Development, Functional Assay
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2A Clone # 48311
Loading...
Product Specifications
Immunogen
NS0 mouse myeloma cell line transfected with human CXCR2/IL-8 RB
Met1-Leu355
Accession # AAB25880
Met1-Leu355
Accession # AAB25880
Specificity
Detects human CXCR2/IL-8 RB transfected NS0 cells but not the parental cell line. It does not cross-react with CXCR1 (IL-8 RA).
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2A
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human CXCR2/IL-8RB Antibody
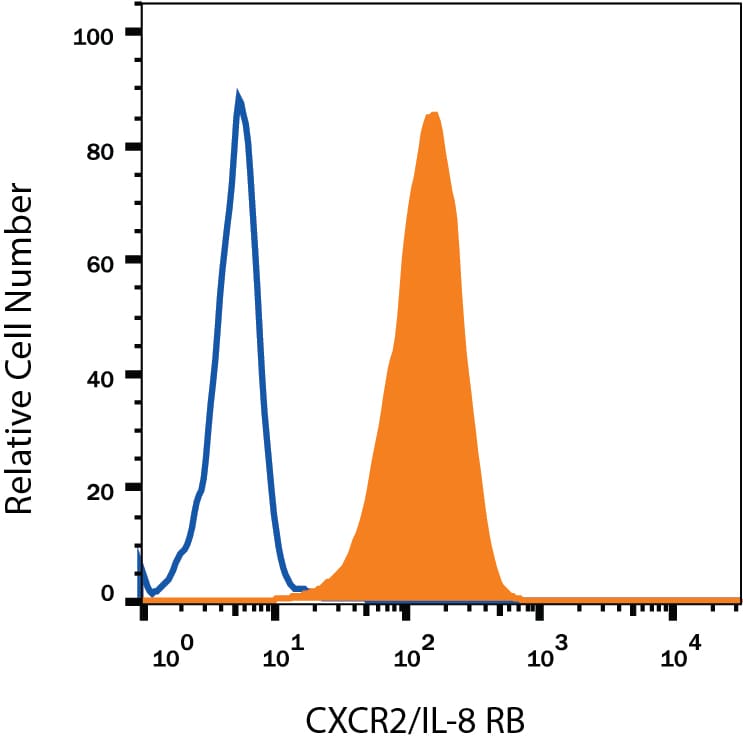
Detection of CXCR2/IL‑8 RB in Human Blood Granulocytes by Flow Cytometry.
Human peripheral blood granulocytes were stained with Mouse Anti-Human CXCR2/IL-8 RB Monoclonal Antibody (Catalog # MAB331, filled histogram) or isotype control antibody (Catalog # MAB003, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0102B).CXCR2/IL‑8 RB in Human Lymph Node.
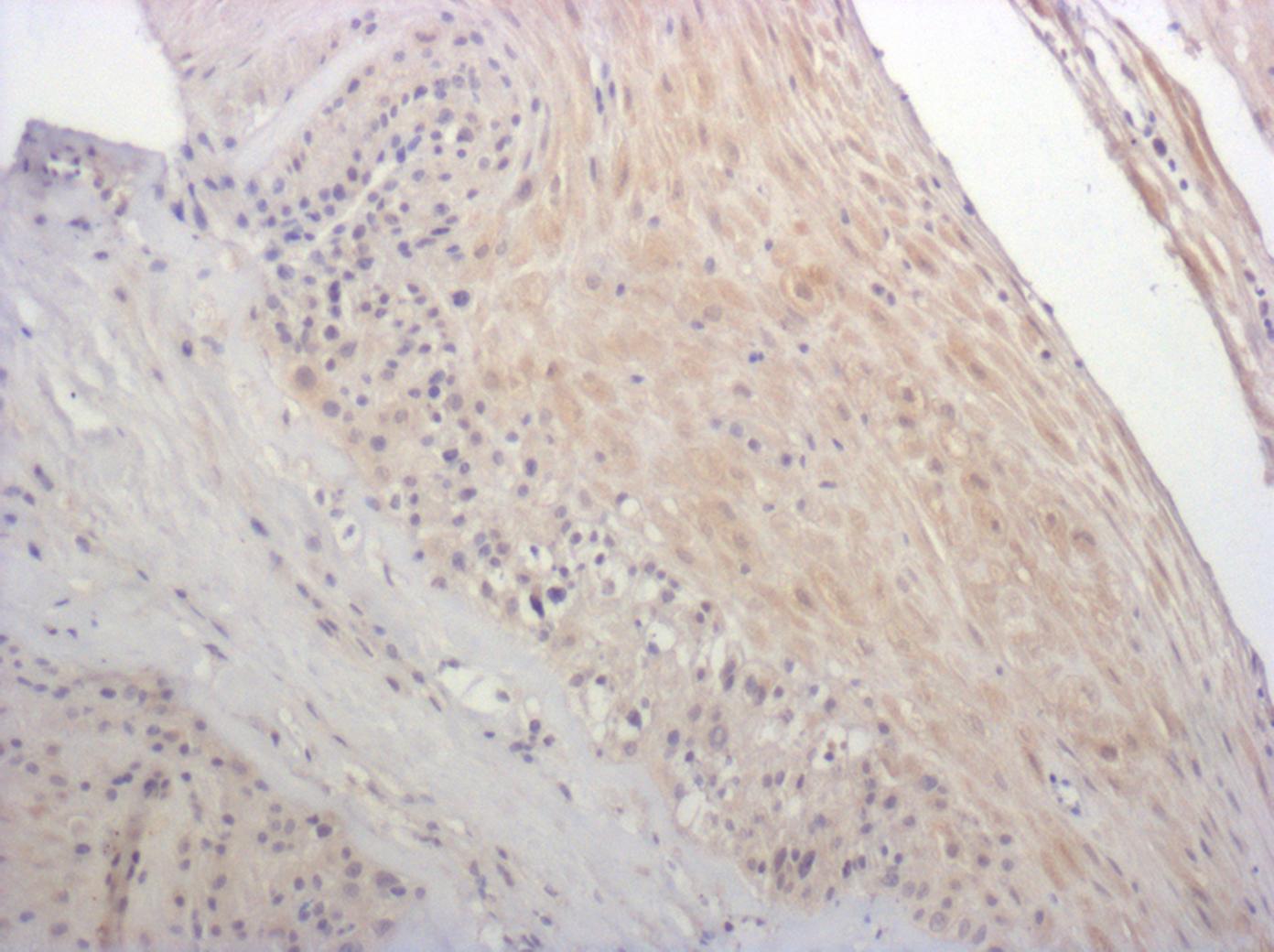
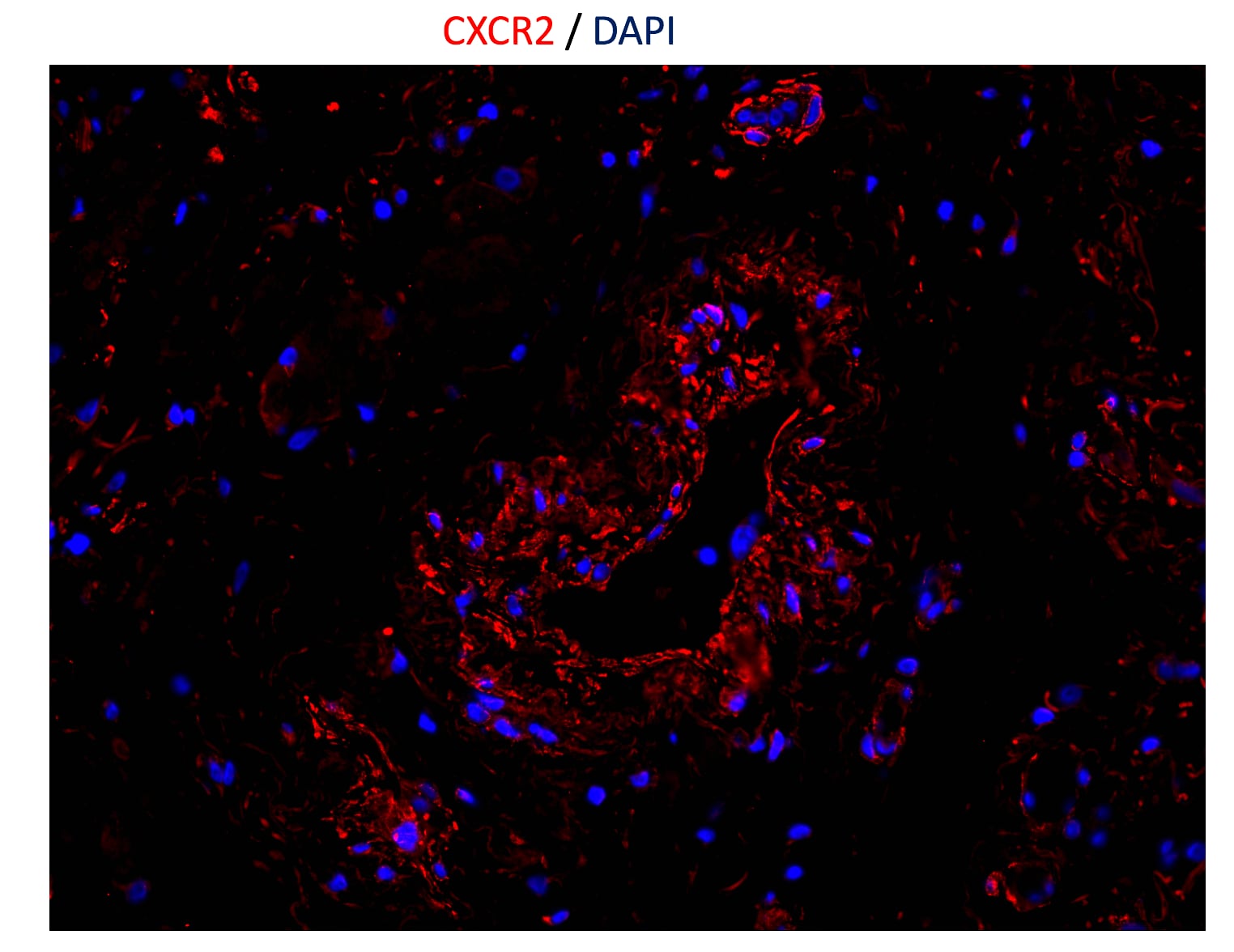
CXCR2/IL-8 RB was detected in immersion fixed paraffin-embedded sections of human lymph node using 15 µg/mL Mouse Anti-Human CXCR2/IL-8 RB Monoclonal Antibody (Catalog # MAB331) overnight at 4 °C. Tissue was stained with the Anti-Mouse HRP-AEC Cell & Tissue Staining Kit (red; Catalog # CTS003) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Chemotaxis Induced by CXCL1/GRO alpha and Neutralization by Human CXCR2/IL‑8 RB Antibody.
Recombinant Human CXCL1/GROa (Catalog # 275-GR) chemoattracts the BaF3 mouse pro-B cell line transfected with human CXCR2 in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin (Catalog # AR002). Chemotaxis elicited by Recombinant Human CXCL1/GROa (5 ng/mL) is neutralized (green line) by increasing concentrations of Mouse Anti-Human CXCR2/IL-8 RB Monoclonal Antibody (Catalog # MAB331). The ND50 is typically 1-5 µg/mL.Detection of CXCR2/IL-8RB by Western Blot
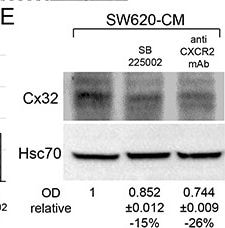
SW620 cell-secreted factors require CXCR2 signaling pathway to induce the endothelial Cx32 expression and tube formationA. IL-8 secretion in conditioned media from SW480 and SW620 cells was examined through ELISA. CRC cells were exposed or not to the HMEC-CM. All cell media were collected after 6 h (mean ± SD, *P-values < 0.01 Mann-Whitney U test and Kruskal-Wallis test; n = 4). B. SW620-CM increase the endothelial expression of the CXCR2 receptor. A slight but significant increase in optical density (OD; relative to control) of bands was detected in response to SW620-CM compared with unstimulated HMEC (P-values < 0.01 Mann-Whitney U test and Kruskal-Wallis test; n = 4). No inhibitory effect was observed by pre-treating HMEC with SB225002 (200 nM), the CXCR2 antagonist. Representative of 4 experiments (Hsc70 as loading control; 100 μg/lane). C–D. Endothelial CXCR2 conveys angiogenic effects of SW620-CM. HMEC were pretreated or not with neutralizing anti-CXCR2 antibody (anti-CXCR2 mAb; 10 μg/ml) or SB225002. Cells were exposed to SW620-CM or human recombinant rhIL-8 (1 ng/ml) for 6 h. C. Representative Images of tube formation (Bar 80 μm). The dotted areas are enlarged in the inserts on the right. Arrows indicated branch points. D. Number of branch points per field of view was quantified (mean ± SD, n = 4; *P < 0.01 vs control). E. Blocking CXCR2 significantly diminished SW620-CM-induced expression of Cx32 in HMEC (P-values < 0.01 vs SW620-CM Mann-Whitney U test and Kruskal-Wallis test; n = 3). HMEC were exposed to cell-conditioned media for 6 h. In some cases, HMEC were pretreated with anti-CXCR2 mAb or SB225002, as indicated. This is a representative of three experiments with similar results (Hsc70 as loading control; 100 μg/lane). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/26320187), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCR2/IL-8RB by Flow Cytometry
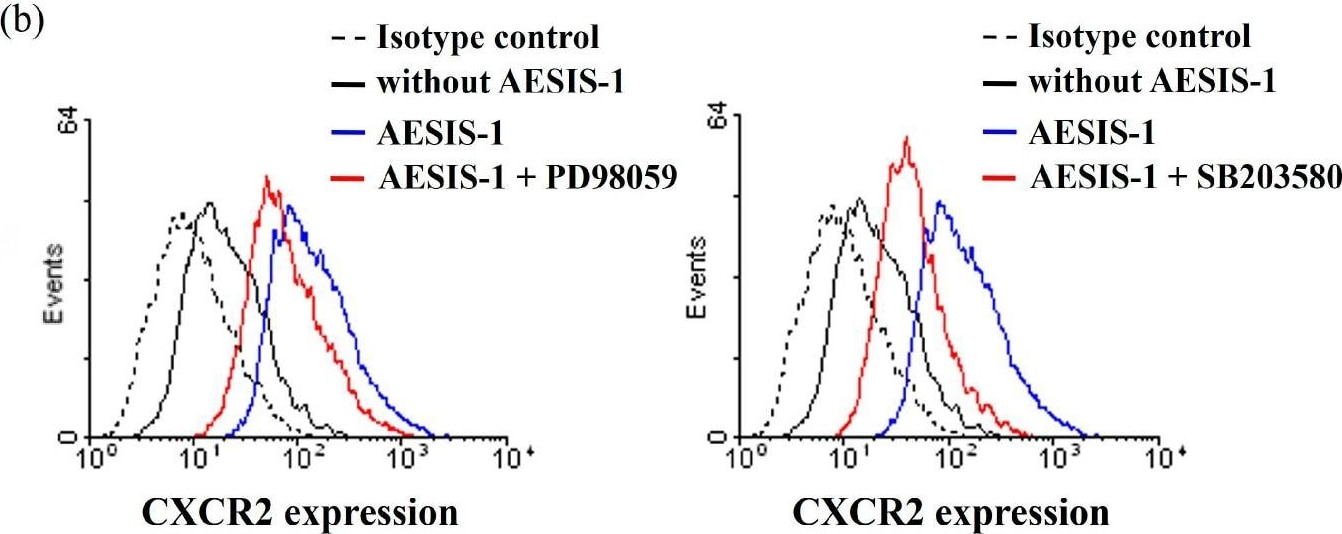
AESIS-1 enhances migration activities and CXCR2 expression through ERK and p38 MAPK signaling in HDF cells. Cells were treated with 20 ng/mL AESIS-1 for 24 h. MAPK inhibitors (ERK-PD98059, 10 μM; p38-SB203580, 10 μM; and JNK-SP600215, 20 μM) were added to cells 1 h before peptide treatment. (a) Transwell migration assay was performed using MAPK inhibitors and AESIS-1-treated HDF cells for 12 incubation hours with or without CXCL8 in lower chamber. Error bars, mean ± SD. One-way ANOVA with Turkey’s multiple comparisons test was used for statistical analysis. * p < 0.05, *** p < 0.001 (versus AESIS-1-treated group, with CXCL8-treated control); §§§ p < 0.001 (versus without AESIS-1-treated group, with CXCL8-treated group); ### p < 0.001 (versus without AESIS-1-, CXCL8-treated groups). (b) Surface protein expression of CXCR2 was measured with flow cytometric analysis after HDF cells were treated with AESIS-1 for 24 h with or without MAPK inhibitor pre-treatment. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/7/3937), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human CXCR2/IL-8RB Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: Human whole blood granulocytes
Sample: Human whole blood granulocytes
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lymph node
Sample: Immersion fixed paraffin-embedded sections of human lymph node
Neutralization
Measured by its ability to neutralize CXCL1/GRO alpha /KC/CINC‑1-induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human CXCR2. The Neutralization Dose (ND50) is typically 1-5 µg/mL in the presence of 5 ng/mL Recombinant Human CXCL1/GRO alpha /KC/CINC‑1.
Reviewed Applications
Read 6 reviews rated 4.3 using MAB331 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from ascites
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CXCR2/IL-8RB
Long Name
Interleukin 8 Receptor B
Alternate Names
CD182, CDw128b, CMKAR2, CXCR-2, IL-8 RB, IL8RB
Gene Symbol
CXCR2
UniProt
Additional CXCR2/IL-8RB Products
Product Documents for Human CXCR2/IL-8RB Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CXCR2/IL-8RB Antibody
For research use only
Citations for Human CXCR2/IL-8RB Antibody
Customer Reviews for Human CXCR2/IL-8RB Antibody (6)
4.3 out of 5
6 Customer Ratings
Have you used Human CXCR2/IL-8RB Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
6 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: MyofibroblastsSpecies: HumanVerified Customer | Posted 08/05/2021
-
Application: Block/NeutralizeSample Tested: Peripheral blood neutrophilsSpecies: HumanVerified Customer | Posted 04/26/2017
-
Application: ImmunohistochemistrySample Tested: fetal membraneSpecies: MouseVerified Customer | Posted 01/23/2017sodium citrate antigen retrieval
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Adult lungSpecies: HumanVerified Customer | Posted 11/04/2016
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 22763101Species: HumanVerified Customer | Posted 02/20/2015
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 22626766Species: HumanVerified Customer | Posted 02/20/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways