Epithelial Cellular Adhesion Molecule (EpCAM), also known as KS1/4, gp40, GA733-2, 17-1A, and TROP-1, is a 40 kDa transmembrane glycoprotein composed of a 242 amino acid (aa) extracellular domain with two epidermal-growth-factor-like (EGF-like) repeats within the cysteine-rich N-terminal region, a 23 aa transmembrane domain, and a 26 aa cytoplasmic domain. Human and mouse EpCAM share 82% aa sequence identity. In human, EpCAM also shares 49% aa sequence homology with TROP-2/EGP-1. During embryonic development, EpCAM is detected in fetal lung, kidney, liver, pancreas, skin, and germ cells. In adults, human EpCAM is detected in basolateral cell membranes of all simple, pseudo-stratified, and transitional epithelia, but is not detected in normal squamous stratified epithelia, mesenchymal tissue, muscular tissue, neuro-endocrine tissue, or lymphoid tissue (1). EpCAM expression has been found to increase in actively proliferating epithelia tissues and during adult liver regeneration (1, 2). EpCAM expression is also found to increase in human malignant neoplasias, with most carcinoma expressing EpCAM including those of arising from squamousal epithelia (1). EpCAM has been shown function as a homophilic Ca2+ independent adhesion molecule (3). Homophilic adhesion via EpCAM requires the interaction of both EGF-like repeats, with the first EGF-like repeat mediating reciprocal interaction between EpCAM molecules on opposing cells, while the second repeat is involved in lateral interaction of EpCAM. Lateral interaction of EpCAM lead to the formation of dimers and tetramers (4). During homophilic adhesion the cytoplasmic tail of EpCAM interacts with the actin cytoskeleton via a direct association alpha -Actinin (5).
Human EpCAM/TROP‑1 Antibody
R&D Systems | Catalog # MAB9601

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Extracellular domain
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human EpCAM/TROP‑1 Antibody
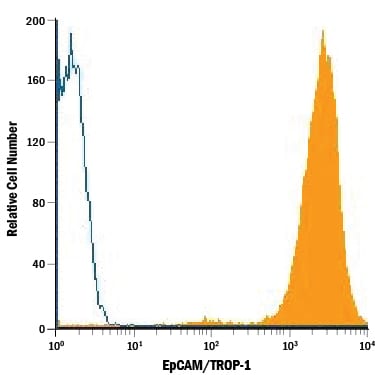
Detection of EpCAM/TROP‑1 in HT‑29 Human Cell Line by Flow Cytometry.
HT-29 human colon adenocarcinoma cell line was stained with Mouse Anti-Human EpCAM/TROP-1 Monoclonal Antibody (Catalog # MAB9601, filled histogram) or isotype control antibody (MAB0041), open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (F0102B).Applications for Human EpCAM/TROP‑1 Antibody
CyTOF-ready
Flow Cytometry
Sample: HT‑29 human colon adenocarcinoma cell line
Human EpCAM/TROP-1 Sandwich Immunoassay
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: EpCAM/TROP1
References
- Balzar, M. et al. (1999) J. Mol. Med. 77:699.
- Boer, C.J, et al. (1999) J. Pathol. 188:201.
- Litvinow, S.V. et al. (1994) J. Cell Biol. 125:437.
- Balzar, M. et al. (2001) Mol. Cell. Biol. 21:2570.
- Balzar, M. et al. (1998) Mol. Cell. Biol. 18:4388.
Long Name
Alternate Names
Gene Symbol
Additional EpCAM/TROP1 Products
Product Documents for Human EpCAM/TROP‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human EpCAM/TROP‑1 Antibody
For research use only
Citations for Human EpCAM/TROP‑1 Antibody
Customer Reviews for Human EpCAM/TROP‑1 Antibody
There are currently no reviews for this product. Be the first to review Human EpCAM/TROP‑1 Antibody and earn rewards!
Have you used Human EpCAM/TROP‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars



