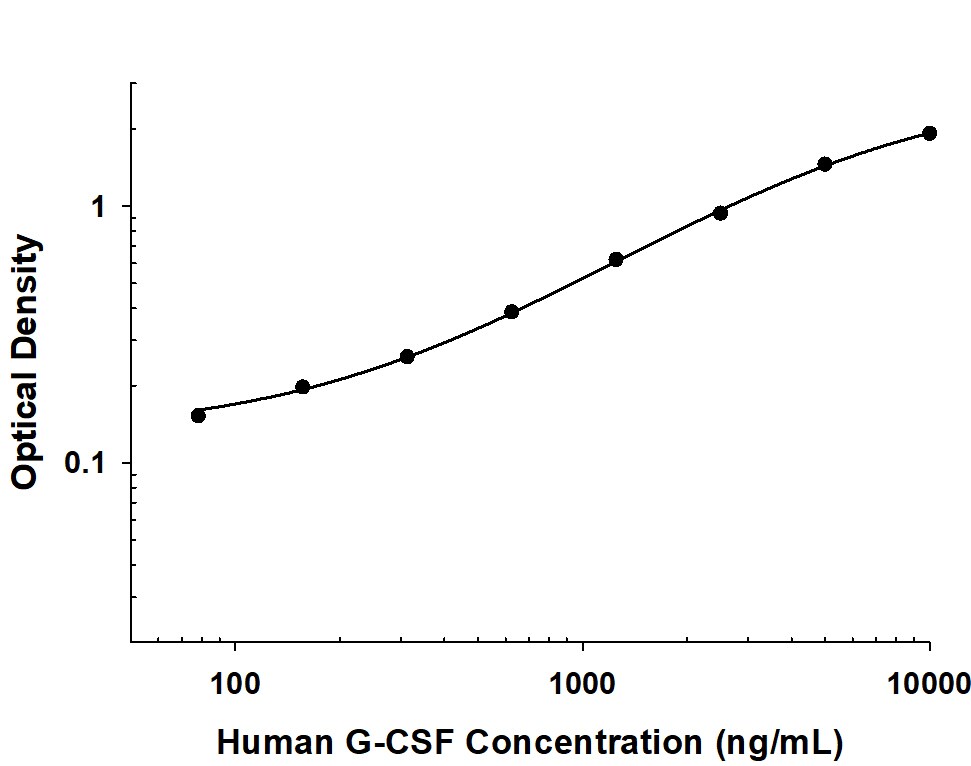
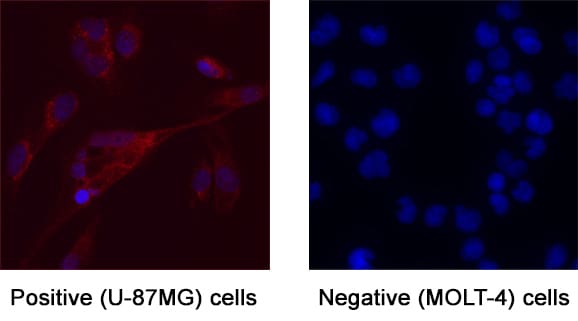
G-CSF
is a pleiotropic cytokine best known for its specific effects on the
proliferation, differentiation, and activation of hematopoietic cells of
the neutrophilic granulocyte lineage. It is produced mainly by
monocytes and macrophages upon activation by endotoxin, TNF-alpha and IFN-gamma.
Other cell types including fibroblasts, endothelial cells, astrocytes
and bone marrow stromal cells can also secrete G-CSF after LPS, IL-1 or
TNF-alpha activation. In addition, various carcinoma cell lines and
myeloblastic leukemia cells can express G-CSF constitutively.
In
humans, two distinct cDNA clones for G-CSF, encoding 207 and 204 amino
acid precursor proteins, have been isolated. Both proteins have a 30
amino acid signal peptide and have identical amino acid sequences except
for a three amino acid insertion (deletion) at the 35th amino acid
residue from the N-terminus of the mature protein. Human G-CSF is 73%
identical at the amino acid level to murine G-CSF and the two proteins
show species cross-reactivity.
In vitro, G-CSF stimulates
growth, differentiation and functions of cells from the neutrophil
lineage. It also has blast cell growth factor activity and can synergize
with IL-3 to shorten the Go period of early hematopoietic progenitors. Consistent with its in vitro
functions, G-CSF has been found to play important roles in defense
against infection, in inflammation and repair, and in the maintenance of
steady state hematopoiesis.