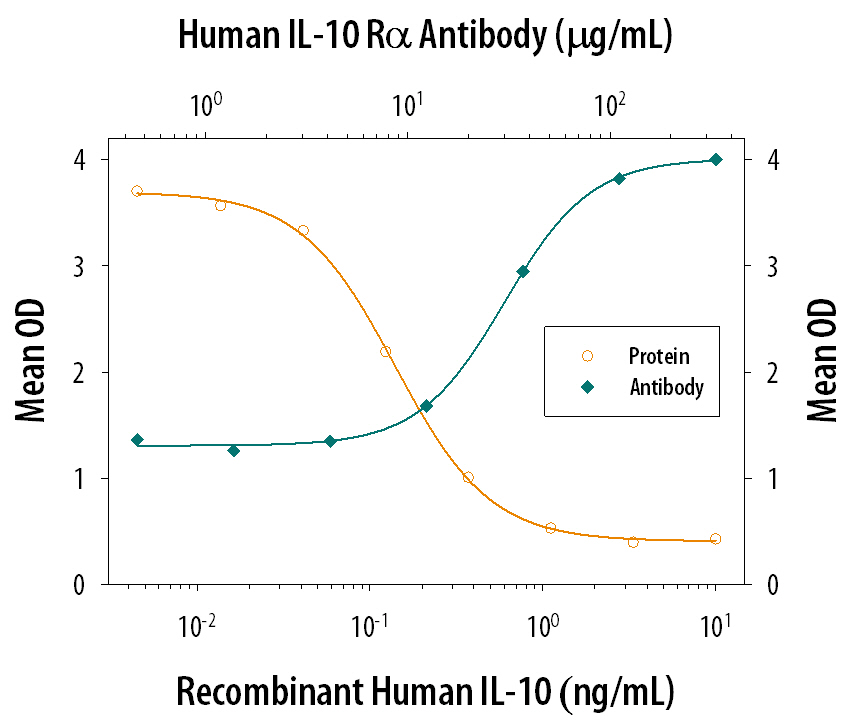
IL-10, initially designated cytokine synthesis inhibitory factor (CSIF), is a potent immunosuppressant of macrophage functions. IL-10 is also a pleiotropic cytokine with multiple immunostimulatory as well as immunosuppressive effects on a variety of other cell types. IL-10 binds specifically and with high affinity to cell-surface receptors. Mouse and human cDNA clones encoding the ligand-binding IL-10 receptor (IL-10 R) have been isolated. The IL-10 R mRNA has been detected in all cell types that are known to respond to IL-10.
Human and mouse IL-10 receptors are structurally related to the IFN-gamma receptor. These receptors are members of the class II subgroup of the cytokine receptor superfamily. The deduced amino acid sequence of human IL-10 R is approximately 60% identical to mouse IL-10 R. Although human IL-10 has cross-species activities and is active on mouse cells, mouse IL-10 is species-specific in its actions and does not bind to the human IL-10 receptor. The human IL-10 R gene has been mapped to chromosome 11q23.3. Recombinant IL-10 soluble receptor, consisting of the extracellular domain of IL-10 R, binds IL-10 with high affinity in solution and is a potent IL-10 antagonist.