Myeloperoxidase (MPO) is a heme-containing enzyme belonging to the XPO subfamily of peroxidases. It is an abundant neutrophil and monocyte glycoprotein that catalyzes the hydrogen peroxide-dependent conversion of chloride, bromide, and iodide to multiple reactive species (1). Post-translational processing of MPO involves the insertion of a heme moiety and the proteolytic removal of both a propeptide and a 6 aa internal peptide (2). This results in a disulfide-linked dimer composed of a 60 kDa heavy and 12 kDa light chain that associate into a 150 kDa enzymatically active tetramer. The tetramer contains two heme groups and one disulfide bond between the heavy chains (2). Alternate splicing generates two additional isoforms of MPO, one with a 32 aa insertion in the light chain, and another with a deletion of the signal sequence and part of the propeptide (3). Human and mouse MPO share 87% aa sequence identity. MPO activity results in protein nitrosylation and the formation of 3-chlorotyrosine and dityrosine crosslinks (4‑6). Modification of ApoB100, as well as the lipid and cholesterol components of LDL and HDL, promotes the development of atherosclerosis (5, 7‑9). MPO is also associated with a variety of other diseases (1), and inhibits vasodilation in inflammation by depleting the levels of NO (10). Serum albumin functions as a carrier protein during MPO movement to the basolateral side of epithelial cells (11). MPO is stored in neutrophil azurophilic granules. Upon cellular activation, it is deposited into pathogen-containing phagosomes (2). While mice lacking MPO are impaired in clearing select microbial infections, MPO deficiency in humans does not necessarily result in heightened susceptibility to infections (12, 13).
Human Myeloperoxidase/MPO Antibody
R&D Systems | Catalog # AF3174

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Mouse
Applications
Validated:
Western Blot, Immunocytochemistry, Simple Western
Cited:
Immunohistochemistry, ELISA Development
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Human polymorphonuclear leukocytes Myeloperoxidase and mouse myeloma cell line NS0-derived recombinant human Myeloperoxidase/MPO
Ala49-Ser745
Accession # P05164
Ala49-Ser745
Accession # P05164
Specificity
Detects human Myeloperoxidase/MPO in direct ELISAs and Western blots. In direct ELISAs, less than 30% cross-reactivity with recombinant mouse MPO is observed and less than 10% cross-reactivity with recombinant human Eosinophil Peroxidase (EPPO) is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Myeloperoxidase/MPO Antibody
Detection of Human Myeloperoxidase/MPO by Western Blot.
Western blot shows lysates of HL-60 human acute promyelocytic leukemia cell line and human neutrophil. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Human Myeloperoxidase/MPO Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3174) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for Myeloperoxidase/MPO at approximately 60-65 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 2.Myeloperoxidase/MPO in HL‑60 Human Cell Line.
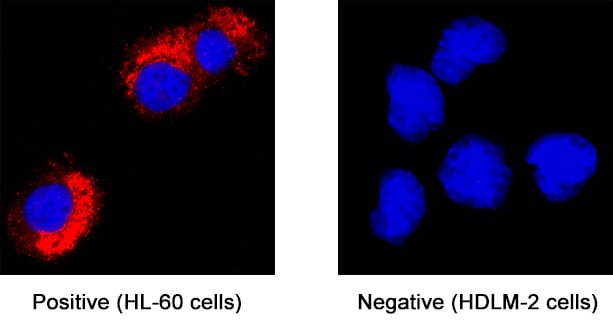
Myeloperoxidase/MPO was detected in immersion fixed HL‑60 human acute promyelocytic leukemia cell line (positive staining) and HDLM‑2 human Hodgkin’s lymphoma cell line (negative staining) using Goat Anti-Human Myeloperoxidase/MPO Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3174) at 1.7 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. Staining was performed using our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of Human Myeloperoxidase/MPO by Simple WesternTM.
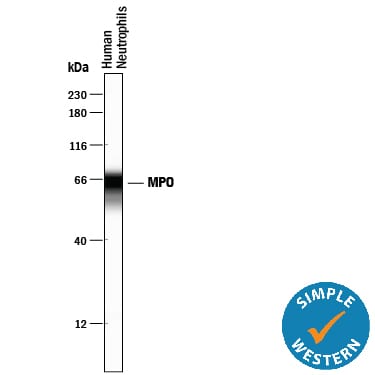
Simple Western lane view shows lysates of human neutrophils, loaded at 0.2 mg/mL. A specific band was detected for Myeloperoxidase/MPO at approximately 65 kDa (as indicated) using 5 µg/mL of Goat Anti-Human Myeloperoxidase/MPO Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3174) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Mouse Myeloperoxidase/MPO by Immunocytochemistry/Immunofluorescence
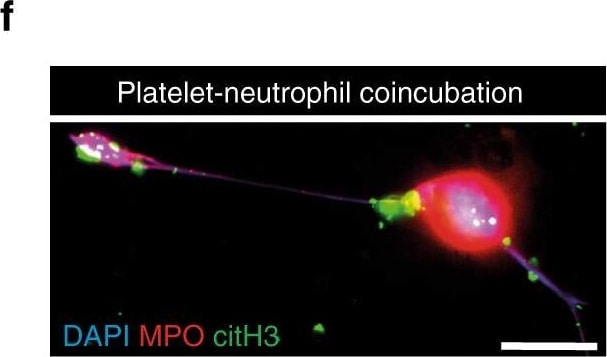
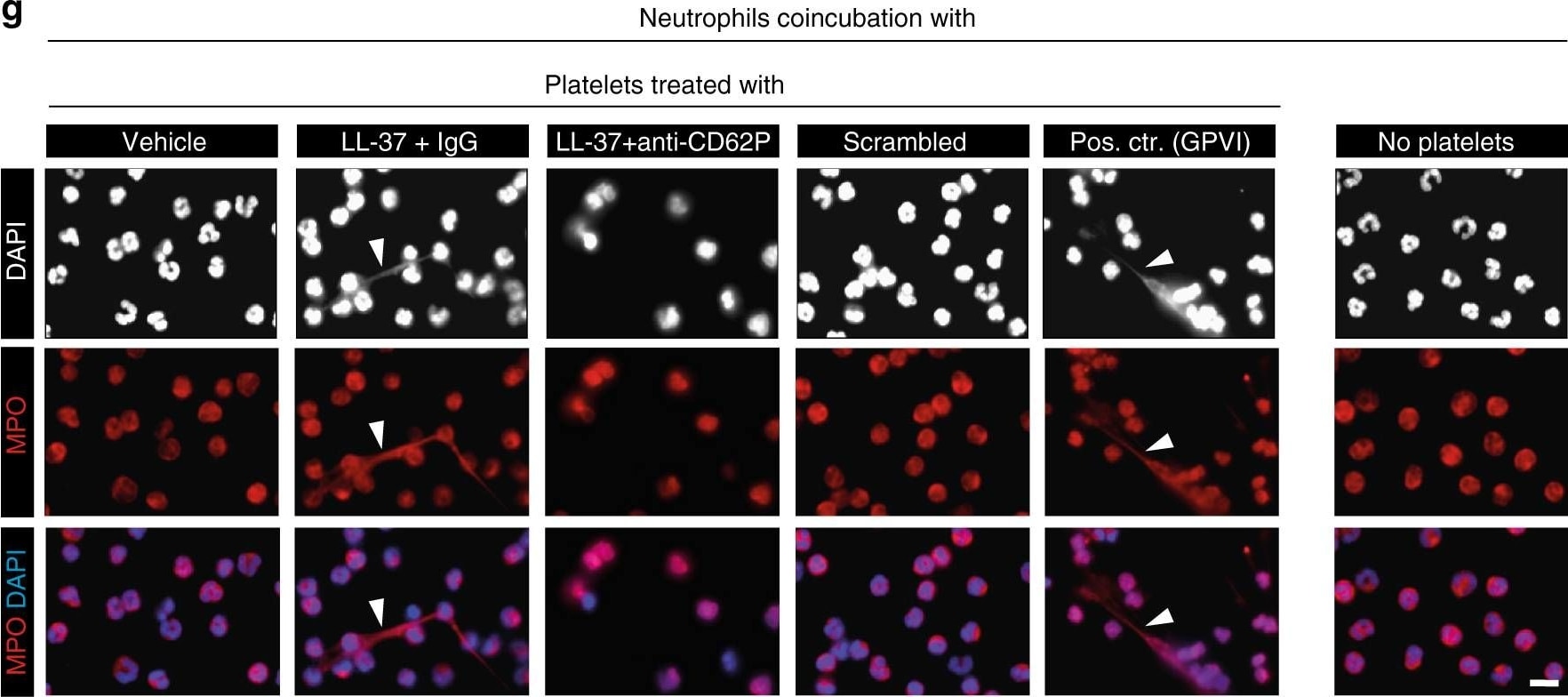
Cathelicidins induce platelet–neutrophil interactions. a–h Co-incubation experiments. Human platelets were pretreated with LL-37 or scrambled control peptide (Scra) and platelet–neutrophil interactions were analyzed. a–e Flow cytometry analysis of a platelet–neutrophil aggregates formation (n = 9), b platelet–neutrophil aggregates in the presence of a blocking antibody against P-selectin and respective isotype control (n = 5), c CD11b expression on neutrophils, d neutrophil intracellular formation of reactive oxygen species (ROS), e shedding of neutrophil L-selectin (n = 4). TNF alpha (50 ng/mL) served as positive control. f–h Neutrophil extracellular trap (NET) formation assay. f Representative epifluorescence image of a NET. DAPI (nuclear stain, blue), myeloperoxidase (MPO, red), and citrullinated histone H3 (citH3, green). Bar, 10 µm. g NET formation was induced by platelets that were pretreated with LL-37 or a GPVI-activating antibody (HGP4C9). Upper row (DAPI nuclear stain, white), middle row (MPO, red), and bottom row (merged image of DAPI in blue, and MPO in red). Arrowheads indicate NET. Bar, 10 µm. h Quantitative analysis of NET formation (n = 4). i, j Interactions of mouse cells. i Platelet–neutrophil aggregates formation of mouse neutrophils with platelets isolated from wild type (WT) or P-selectin deficient mice (n = 7). j Platelet–neutrophil aggregates formation after co-incubation of isolated WT platelets with PMA (50 µmol/L) activated neutrophils of WT or CRAMP−/− mice (n = 4). Graphs show mean and SEM. P-values were determined by one-way repeated measures ANOVA with Bonferroni correction (a–c), paired t-test (d, e), ANOVA on Ranks/Dunn’s method (h) or Mann–Whitney U-test (i, j) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29670076), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Myeloperoxidase/MPO by Immunocytochemistry/Immunofluorescence
Cathelicidins induce platelet–neutrophil interactions. a–h Co-incubation experiments. Human platelets were pretreated with LL-37 or scrambled control peptide (Scra) and platelet–neutrophil interactions were analyzed. a–e Flow cytometry analysis of a platelet–neutrophil aggregates formation (n = 9), b platelet–neutrophil aggregates in the presence of a blocking antibody against P-selectin and respective isotype control (n = 5), c CD11b expression on neutrophils, d neutrophil intracellular formation of reactive oxygen species (ROS), e shedding of neutrophil L-selectin (n = 4). TNF alpha (50 ng/mL) served as positive control. f–h Neutrophil extracellular trap (NET) formation assay. f Representative epifluorescence image of a NET. DAPI (nuclear stain, blue), myeloperoxidase (MPO, red), and citrullinated histone H3 (citH3, green). Bar, 10 µm. g NET formation was induced by platelets that were pretreated with LL-37 or a GPVI-activating antibody (HGP4C9). Upper row (DAPI nuclear stain, white), middle row (MPO, red), and bottom row (merged image of DAPI in blue, and MPO in red). Arrowheads indicate NET. Bar, 10 µm. h Quantitative analysis of NET formation (n = 4). i, j Interactions of mouse cells. i Platelet–neutrophil aggregates formation of mouse neutrophils with platelets isolated from wild type (WT) or P-selectin deficient mice (n = 7). j Platelet–neutrophil aggregates formation after co-incubation of isolated WT platelets with PMA (50 µmol/L) activated neutrophils of WT or CRAMP−/− mice (n = 4). Graphs show mean and SEM. P-values were determined by one-way repeated measures ANOVA with Bonferroni correction (a–c), paired t-test (d, e), ANOVA on Ranks/Dunn’s method (h) or Mann–Whitney U-test (i, j) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29670076), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Myeloperoxidase/MPO by Immunocytochemistry/Immunofluorescence
Cathelicidins are present in human and mouse arterial thrombi. a, b Representative images of coronary artery thrombi isolated from five patients with acute myocardial infarction. a Immunohistochemistry for LL-37 indicated enrichment within leukocytes (arrowhead), but also stained leukocyte-free areas (asterisk). Bars, 200 µm (overview) and 10 µm (magnification). b Immunofluorescence analysis of LL-37 (red), myeloperoxidase (MPO, yellow), CD41 (platelets, green), and DAPI (nuclei, blue). Bar, 10 µm. c, d Representative images of murine carotid artery thrombi generated by ferric chloride injury. c Immunohistochemistry for cathelicidin-related antimicrobial peptide (CRAMP, mouse homologue for LL-37) indicated enrichment within leukocytes (arrowhead), but also stained leukocyte-free areas (asterisk). Bars, 10 µm. d Immunofluorescence analysis of CRAMP (red), CD41 (platelets, green), and DAPI (nuclei, blue). Bar, 10 µm. e Analysis of CRAMP binding in arterial thrombosis in vivo. 5-FAM-labeled CRAMP or scrambled control was injected into wild type mice before induction of ferric chloride injury. Platelets were labeled in vivo using a DyLight649-labeled non-blocking GPIb beta antibody. Left: 5-FAM-labeled CRAMP (green) associated with platelets (GPIb, red in merged image) in the forming thrombus. Right: Image for 5-FAM-labeled control peptide and platelets (GPIb, red in merged image). Bar, 500 µm. See also Supplemental Movies 1, 2. f Flow cytometry analysis of LL-37 binding to isolated human, platelets in vitro. 5-FAM-labeled LL-37 (red), scrambled 5-FAM-labeled control peptide (blue), or vehicle (gray). Graph shows mean and SEM. P-value was determined by unpaired t-test Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29670076), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Myeloperoxidase/MPO Antibody
Application
Recommended Usage
Immunocytochemistry
1.7-15 µg/mL
Sample: Immersion fixed HL‑60 human acute promyelocytic leukemia cell line
Sample: Immersion fixed HL‑60 human acute promyelocytic leukemia cell line
Simple Western
5 µg/mL
Sample: Human neutrophils
Sample: Human neutrophils
Western Blot
0.5 µg/mL
Sample: HL‑60 human acute promyelocytic leukemia cell line and human neutrophils
Sample: HL‑60 human acute promyelocytic leukemia cell line and human neutrophils
Reviewed Applications
Read 1 review rated 3 using AF3174 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Myeloperoxidase/MPO
References
- Klebanoff, S.J. (2005) J. Leukoc. Biol. 77:598.
- Hansson, M. et al. (2006) Arch. Biochem. Biophys. 445:214.
- Hashinaka, K. et al. (1988) Biochemistry 27:5906.
- van Dalen, C.J. et al. (2000) J. Biol. Chem. 275:11638.
- Hazen, S.L. and J.W. Heinecke (1997) J. Clin. Invest. 99:2075.
- Heinecke, J.W. et al. (1993) J. Clin. Invest. 91:2866.
- Podrez, E.A. et al. (1999) J. Clin. Invest. 103:1547.
- Bergt, C. et al. (2004) Proc. Natl. Acad. Sci. 101:13032.
- Hazen, S.L. et al. (1996) J. Biol. Chem. 271:23080.
- Eiserich, J.P. et al. (2002) Science 296:2391.
- Tiruppathi, C. et al. (2004) Proc. Natl. Acad. Sci. 101:7699.
- Aratani Y. et al. (2000) J. Infect. Dis. 182:1276.
- Kutter, D. (1998) J. Mol. Med. 76:669.
Alternate Names
MPO
Gene Symbol
MPO
UniProt
Additional Myeloperoxidase/MPO Products
Product Documents for Human Myeloperoxidase/MPO Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Myeloperoxidase/MPO Antibody
For research use only
Related Research Areas
Citations for Human Myeloperoxidase/MPO Antibody
Customer Reviews for Human Myeloperoxidase/MPO Antibody (1)
3 out of 5
1 Customer Rating
Have you used Human Myeloperoxidase/MPO Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ELISASample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 12/20/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways