Key Product Details
Validated by
Knockout/Knockdown
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Rat, Primate - Macaca fascicularis (Crab-eating Monkey or Cynomolgus Macaque), Primate - Macaca mulatta (Rhesus Macaque), Primate - Pongo (Orangutan), Rabbit, Xenograft
Applications
Validated:
Western Blot, Immunocytochemistry, Simple Western, Immunoprecipitation, Chromatin Immunoprecipitation (ChIP)
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Chromatin Immunoprecipitation (ChIP), Bioassay, Co-Immunoprecipitation, Differentiation
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Oct‑3/4
Met1-Asn265 (Met262Leu)
Accession # Q01860
Met1-Asn265 (Met262Leu)
Accession # Q01860
Specificity
Detects human Oct-3/4 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse Oct‑3/4 Antibody
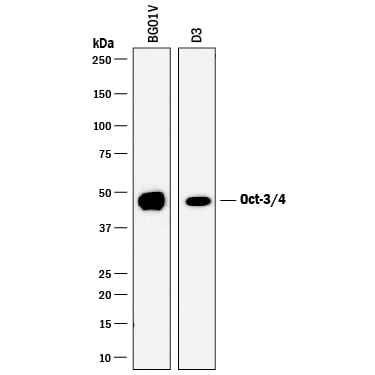
Detection of Human and Mouse Oct‑3/4 by Western Blot.
Western blot shows lysates of BG01V human embryonic stem cells and D3 mouse embryonic stem cell line. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Human/Mouse Oct-3/4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1759) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for Oct-3/4 at approximately 48 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Alkaline Phosphatase and Oct‑3/4 in BG01V Human Stem Cells.
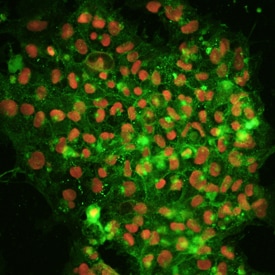
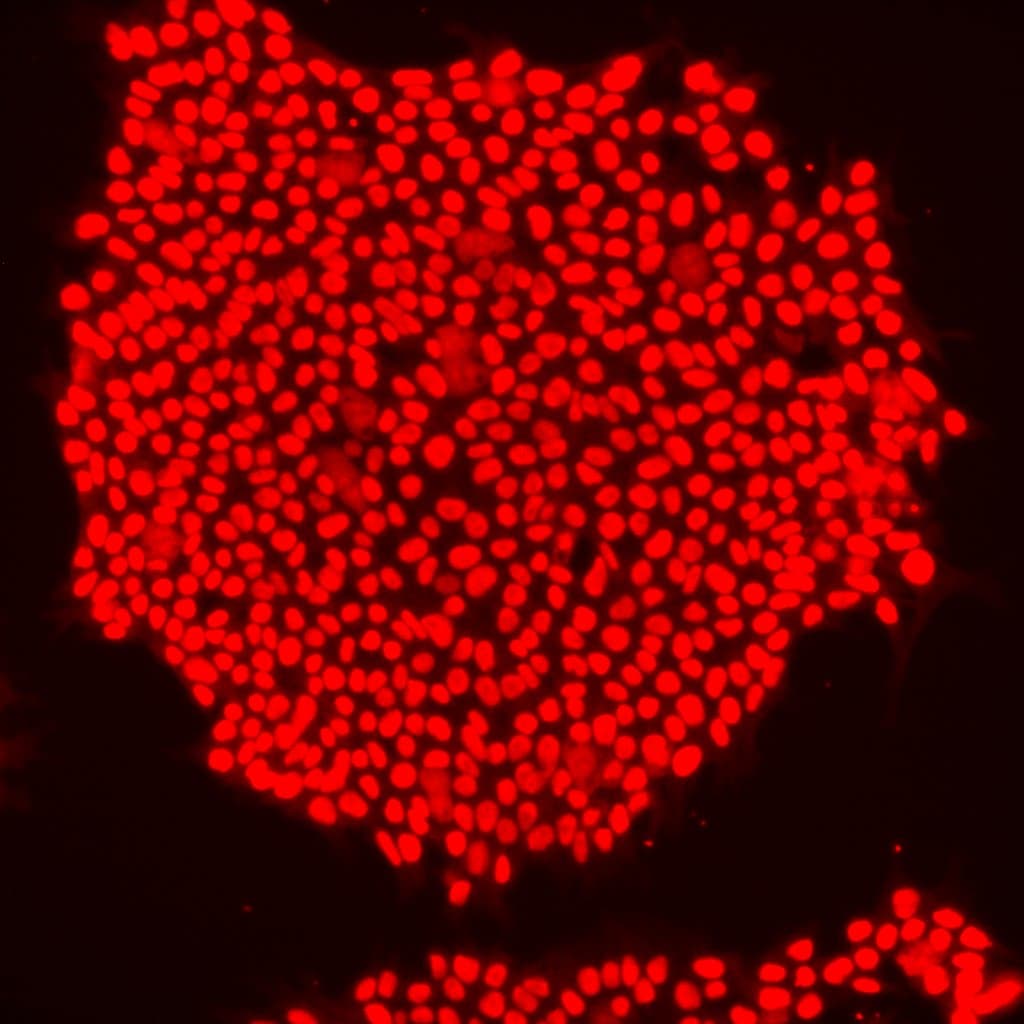
Alkaline phosphatase (ALPL) and Oct-3/4 were detected in human BG01V embryonic stem cells using 10 µg/mL Human/Mouse/Rat ALPL Monoclonal Antibody (Catalog # MAB1448) and 10 µg/mL Goat Anti-Human/Mouse Oct-3/4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1759). Cells were incubated with primary antibodies for 3 hours at room temperature. Cells were stained for ALPL using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (pseudo-stained green; Catalog # NL007), and stained for Oct-3/4 using the NorthernLights 637-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL002). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Oct‑3/4-regulated Genes by Chromatin Immunoprecipitation.
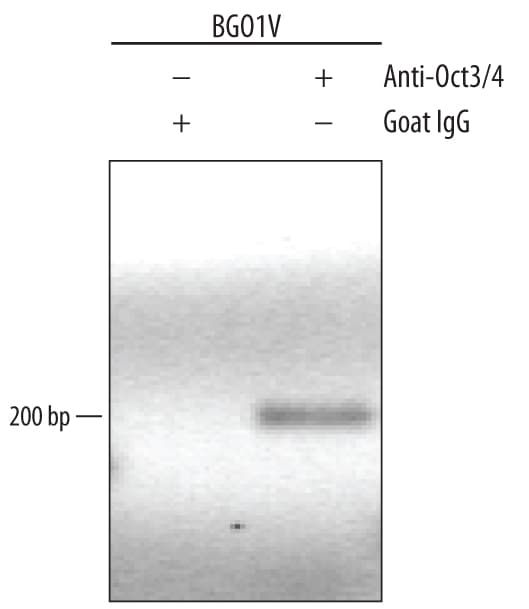
BG01V human embryonic stem cells were fixed using formaldehyde, resuspended in lysis buffer, and sonicated to shear chromatin. Oct-3/4/DNA complexes were immunoprecipitated using 5 µg Goat Anti-Human/Mouse Oct-3/4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1759) or control antibody (Catalog # AB-108-C) for 15 minutes in an ultrasonic bath, followed by Biotinylated Anti-Goat IgG Secondary Antibody (Catalog # BAF109). Immunocomplexes were captured using 50 µL of MagCellect Streptavidin Ferrofluid (Catalog # MAG999) and DNA was purified using chelating resin solution. Thenanogpromoter was detected by standard PCR.Detection of Human Oct‑3/4 by Simple WesternTM.
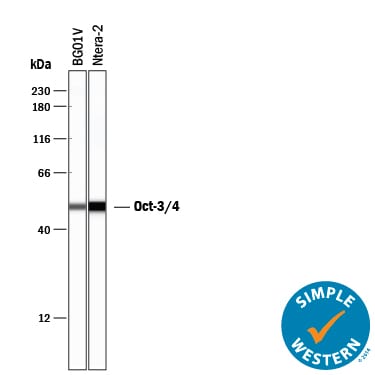
Simple Western lane view shows lysates of BG01V human embryonic stem cells and NTera-2 human testicular embryonic carcinoma cell line, loaded at 0.2 mg/mL. A specific band was detected for Oct-3/4 at approximately 50 kDa (as indicated) using 20 µg/mL of Goat Anti-Human/Mouse Oct-3/4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1759) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Human Oct-3/4 by Western Blot
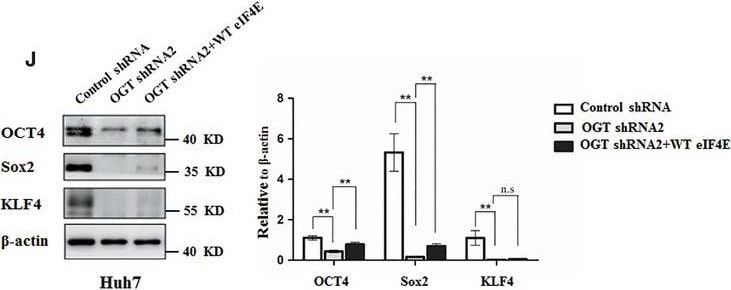
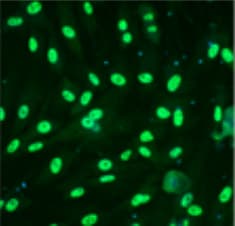
Knockdown OGT inhibits proliferation and tumorsphere formation of hepatoma cell through reducing eIF4E expression. A, Huh7 and PLC/PRF/5 cells were infected with control shRNA, OGT shRNA2 alone, or with wild‐type eIF4E lentivirus. The cell lysates were harvested for western blotting analysis using indicated antibodies. beta ‐actin expression was served as a loading control. B, Cell proliferation of Huh7 and PLC/PRF/5 cells infected with lentiviruses as in panel (A) were measured with CCK8 assay. (C‐H) Huh7 and PLC/PRF/5 cells infected with lentiviruses as in panel (A) were seeded into 96‐well plates. After 12 d, tumorsphere were counted and quantified. Representative images of sphere (scale bars, 100 μm) were shown (C, F). The diameter of sphere (D, G) and number of sphere (E, H) were count. Data represent mean ± SD of at least three independent experiments. The two‐tailed Student's t tests were used. **P < 0.01. I, Huh7 cells expressing either OGT shRNA2 alone or with wild‐type eIF4E lentivirus were incubated with PE‐labelled anti‐AC133 antibody. The percentages of CD133+ cells in graphs were analysed by flow cytometry. Black line, control IgG staining; red line, CD133 staining. J, Cell lysates were examined by western blotting with indicated antibodies. The right panel showcases relative protein amounts of different groups. Error bars represent ±SD of triplicate experiments. **P < 0.01; n.s, no significance. K, Huh7 cells were collected and subjected to immunoprecipitation with antibody against eIF4E or normal mouse IgG. Total RNAs were purified from immunocomplexes and subjected to RT‐PCR to measure Sox2, OCT4, and KLF4 mRNAs associated with eIF4E Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30677218), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Oct-3/4 by Immunocytochemistry/Immunofluorescence
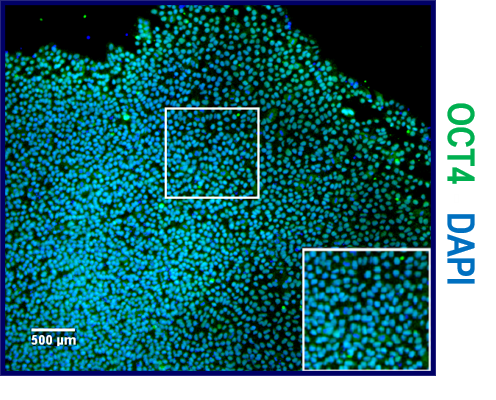
Neural induction of hPSCs. (a) Both hPSC culture and neural differentiation were performed on LN521 substrate. Cortical neurons were induced with dual SMAD inhibition, expanded in the presence of FGF2, and matured with support by a selection of neurotrophic factors. Neural progenitor cells (NPCs) could be cryopreserved at day 21 and plated at day 32 for final experiments, including microelectrode array (MEA) measurements. (b) Three different hPSC lines (one hESC line, 08/023 and two hiPSC lines, 10212.EURCCs and IMR90-4) were characterized for their efficiency in producing neuroectodermal cells in response to 12-day neural induction by dual SMAD inhibition. Cells were stained for pluripotency marker Oct4 at the pluripotent stage and after 12 days of neural induction. The presence of early neuroectodermal markers was evaluated with Sox2, FoxG1 and Pax6 staining. (c) After 21 days of differentiation, the culture contained vimentin- and Pax6-positive NPCs that could be cryopreserved. Additionally, the first Tbr2- and MAP2-positive neurons were detected at this time point. (d) The percentages of Pax6-positive cells were quantified at four time points of differentiation (mean ± s.e.m., n = 5–14, data derived from 1–3 independent differentiations). Statistical analysis was performed with the Mann-Whitney U test to compare differences between day 12 and day 46 within each hPSC line, and significant p-values are presented in the image. The scale bar is 50 µm in all images. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31748598), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse Oct‑3/4 Antibody
Application
Recommended Usage
Chromatin Immunoprecipitation (ChIP)
5 µg/106 cells
Sample: BG01V human embryonic stem cell chromatin, for detection see our available Human Nanog Primer Pair (Catalog # RDP-320-025)
Sample: BG01V human embryonic stem cell chromatin, for detection see our available Human Nanog Primer Pair (Catalog # RDP-320-025)
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed BG01V human embryonic stem cells
Sample: Immersion fixed BG01V human embryonic stem cells
Immunoprecipitation
3 µg/106 cells
Sample: NTera‑2 human testicular embryonic carcinoma cell line, see our available Western blot detection antibodies
Sample: NTera‑2 human testicular embryonic carcinoma cell line, see our available Western blot detection antibodies
Simple Western
20 µg/mL
Sample: BG01V human embryonic stem cells and NTera‑2 human testicular embryonic carcinoma cell line
Sample: BG01V human embryonic stem cells and NTera‑2 human testicular embryonic carcinoma cell line
Western Blot
0.5 µg/mL
Sample: BG01V human embryonic stem cells and D3 mouse embryonic stem cell line
Sample: BG01V human embryonic stem cells and D3 mouse embryonic stem cell line
Reviewed Applications
Read 4 reviews rated 4.5 using AF1759 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Oct-3/4
References
- Scholer, H.R. et al. (1990) Nature 344:435.
- Scholer, H.R. et al. (1989) EMBO J. 8:2543.
- Rosner, M.H. et al. (1990) Nature 345:686.
- Niwa, H. et al. (2000) Nat. Genet. 24:372.
- Pesce, M. et al. (2001) Stem Cells 19:271.
Long Name
Octamer-binding Protein 3/4
Alternate Names
3-Oct, Oct4, Otf3g, Otf4, Pou5f1
Gene Symbol
POU5F1
UniProt
Additional Oct-3/4 Products
Product Documents for Human/Mouse Oct‑3/4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Oct‑3/4 Antibody
For research use only
Citations for Human/Mouse Oct‑3/4 Antibody
Customer Reviews for Human/Mouse Oct‑3/4 Antibody (4)
4.5 out of 5
4 Customer Ratings
Have you used Human/Mouse Oct‑3/4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
4 of
4 reviews
Showing All
Filter By:
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: BG01V human embryonic stem cellsSpecies: HumanVerified Customer | Posted 09/12/2025
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: human induced pluripotent stem cellsSpecies: HumanVerified Customer | Posted 10/24/2023Oct4 staining performed on iPSCs.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: iPS2 human induced pluripotent stem cellsSpecies: HumanVerified Customer | Posted 10/18/2018Cells: human induced pluripotent stem cells Primary antibody: Goat anti-human OCT3/4 (1:200), O/N incubation at 4 degrees Secondary antibody: anti-goat AF568
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Umbilical vein endothelial cellsSpecies: HumanVerified Customer | Posted 11/23/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways