Human ST2/IL-33R Antibody
R&D Systems | Catalog # MAB523

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization
Cited:
Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, AlphaLISA, Mass Cytometry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 97203
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant human ST2/IL-33 R
Lys19-Phe328
Accession # BAA02233
Lys19-Phe328
Accession # BAA02233
Specificity
Detects human ST2/IL-33 R in ELISAs and Western blots. In Western blots, no cross-reactivity with recombinant mouse ST2L, recombinant human (rh) IL-1 RI, rhIL-1 RII, rhIL-1 RA, rhIL-1 alpha, or rhIL-1 beta is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human ST2/IL-33R Antibody
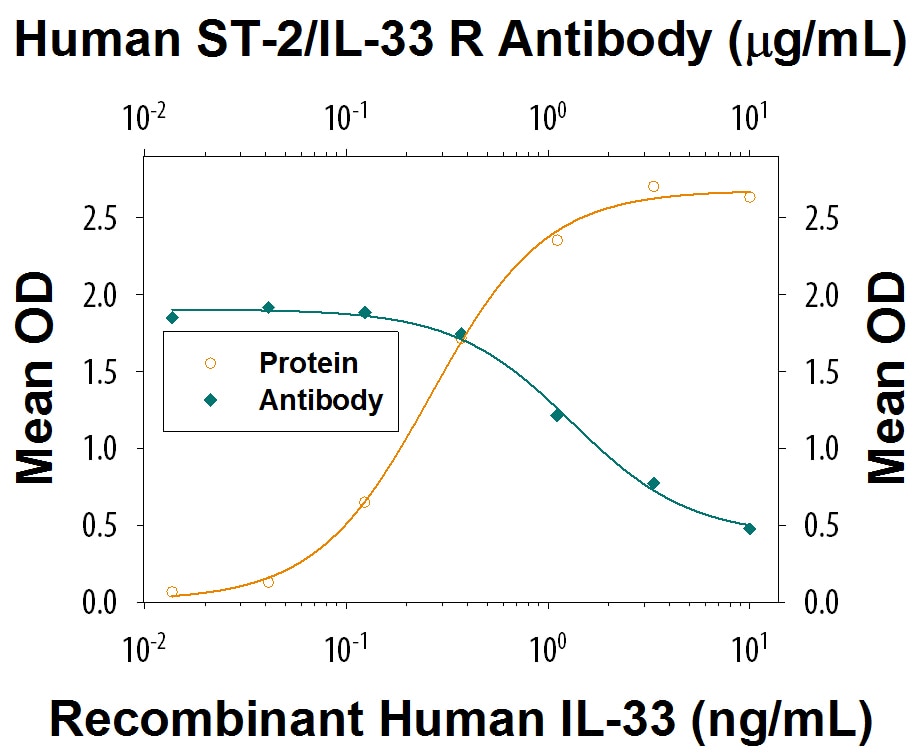
IFN-gamma Secretion Induced by IL‑33 and Neutralization by Human ST2/IL-33 R Antibody.
Recombinant Human IL-33 induces IFN-gamma secretion in Human Peripheral Blood Mononuclear cells (PBMC) in the presence of Recombinant Human IL-12 (Catalog # 219-IL) in a dose-dependent manner (orange line), as measured by the Human IFN-gamma Quantikine ELISA Kit (Catalog # DIF50C). Under these conditions, IFN-gamma secretion elicited by IL-33 is neutralized (green line) by increasing concentrations of Mouse Anti-Human/Primate ST2/IL-33 R Monoclonal Antibody (Catalog # MAB523). The ND50 is typically 0.3-1.5 µg/mL.Detection of Human ST2/IL-33R by Immunocytochemistry/Immunofluorescence
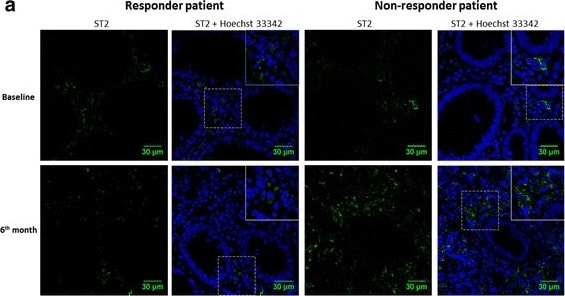
Immunolocalization of ST2 in colonic tissue from responder and non-responder patients during baseline and 6-month follow-up. a Total ST2 immunoreactivity was restricted to the cellular infiltrate in the lamina propria at 6 months in responder patients (right), while in patients showing reactivation, total ST2 was increased in inflamed mucosa, also confined to the cellular infiltrate (left); baseline examination revealed extensive immune cell infiltration of the intestinal mucosa and damaged tissue with loss of architecture. b Total ST2 immunoreactivity at 6 months expressed as arbitrary units (A.U.) and normalized to baseline levels in responders and non-responders (n = 4 in each group) was higher in the non-responders. Hoechst 33342/ ST2 (blue/green) (60X) Image collected and cropped by CiteAb from the following publication (https://bmcgastroenterol.biomedcentral.com/articles/10.1186/s12876-016-…), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human ST2/IL-33R Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Recombinant Human ST2/IL‑1 R4 Fc Chimera (Catalog # 523-ST)
Sample: Recombinant Human ST2/IL‑1 R4 Fc Chimera (Catalog # 523-ST)
Neutralization
Measured by its ability to neutralize IL‑33-induced IFN-gamma secretion in Human Peripheral Blood Mononuclear cells (PBMC).The Neutralization Dose (ND50) is typically 0.3-1.5 μg/mL in the presence of 1 ng/mL Recombinant Human IL‑33.
Human/Primate ST2/IL-33 R Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: ST2/IL-33R
References
-
Barksby, H.E. et al. (2007) Clin. Exp. Immunol. 149:217.
-
Gadina, M. and C.A. Jefferies (2007) Science STKE 2007:pe31.
-
Tominaga, S. et al. (1992) Biochim. Biophys. Acta 1171:215.
-
Li, H. et al. (2000) Genomics 67:284.
-
Schmitz, J. et al. (2005) Immunity 23:479.
-
Lecart, S. et al. (2002) Eur. J. Immunol. 32:2979.
-
Brint, E.K. et al. (2004) Nat. Immunol. 5:373.
-
Weinberg, E.O. et al. (2002) Circulation 106:2961.
-
Sanada S. et al. (2007) J. Clin. Invest. 117:1538.
-
Palmer, G. et al. (2008) Cytokine 42:358.
-
Chackerian, A.A. et al. (2007) J. Immunol. 179:2551.
-
Allakhverdi, Z. et al. (2007) J. Immunol. 179:2051.
-
Verri Jr., W.A. et al. (2008) Proc. Natl. Acad. Sci. 105:2723.
-
Miller, A.M. et al. (2008) J. Exp. Med. 205:339.
-
Hayakawa, H. et al. (2007) J. Biol. Chem. 282:26369.
Long Name
Interleukin 33 Receptor
Alternate Names
DER4, Fit-1, IL-1 R4, IL-1R4, IL-1RL1, IL-33 R, IL-33R, IL1R4, IL33R, Ly84, ST2L, ST2V, T1
Gene Symbol
IL1RL1
UniProt
Additional ST2/IL-33R Products
Product Documents for Human ST2/IL-33R Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human ST2/IL-33R Antibody
For research use only
Related Research Areas
Citations for Human ST2/IL-33R Antibody
Customer Reviews for Human ST2/IL-33R Antibody
There are currently no reviews for this product. Be the first to review Human ST2/IL-33R Antibody and earn rewards!
Have you used Human ST2/IL-33R Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 Th2 Differentiation Pathway
Th2 Differentiation Pathway