Placental Cadherin (P-Cadherin or PCAD) is a member of the cadherin family of cell adhesion molecules. Cadherins are calcium-dependent transmembrane proteins, which bind to one another in a homophilic manner. On their cytoplasmic side, they associate with the three catenins, alpha, beta, and gamma (plakoglobin). This association links the cadherin protein to the cytoskeleton. Without association with the catenins, the cadherins are non-adhesive. Cadherins play a role in development, specifically in tissue formation. They may also help to maintain tissue architecture in the adult. P-Cadherin is a classical cadherin molecule. Classical cadherins consist of a large extracellular domain which contains DXD and DXNDN repeats responsible for mediating calcium-dependent adhesion, a single-pass transmembrane domain, and a short carboxy-terminal cytoplasmic domain responsible for interacting with the catenins. Constitutive P-Cadherin expression is found in the epidermis, mesothelium, corneal epithelium, and uterine decidua. Mouse P-Cadherin is an 822 amino acid (aa) protein with a 27 aa signal sequence and a 795 aa propeptide. The mature protein begins at aa 100 and has a 542 aa extracellular region, a 27 aa transmembrane region, and a 153 aa cytoplasmic region.

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Mouse
Cited:
Mouse, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Adhesion Blockade, Flow Cytometry, Immunocytochemistry, Simple Western, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Flow Cytometry, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse P-Cadherin
Glu100-Gly647
Accession # Q8BSL6
Glu100-Gly647
Accession # Q8BSL6
Specificity
Detects P‑Cadherin in ELISAs and Western blots. In sandwich immunoassays, less than 2% cross-reactivity with recombinant human (rh) P‑Cadherin is observed and less than 0.3% cross-reactivity with recombinant mouse E-Cadherin, rhN-Cadherin, and rhCadherin-8 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse P‑Cadherin Antibody
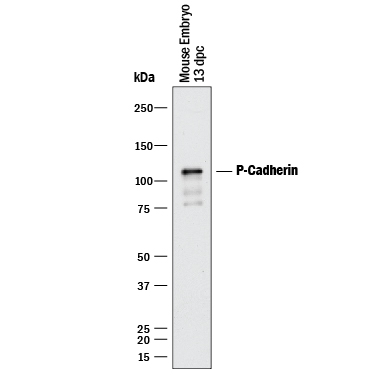
Detection of P‑Cadherin by Western Blot.
Western blot shows lysates of mouse embryo tissue. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Mouse P-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF761) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for P-Cadherin at approximately 115 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.P‑Cadherin in A431 Human Cell Line.
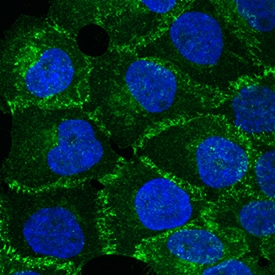
P-Cadherin was detected in immersion fixed A431 human epithelial carcinoma cell line using Goat Anti-Mouse P-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF761) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 493-conjugated Anti-Goat IgG Secondary Antibody (green; NL003) and counterstained with DAPI (blue). Specific staining was localized to intercellular junctions. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.P‑Cadherin in Mouse Embryo.
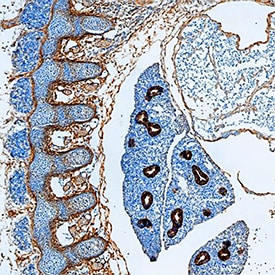
P-Cadherin was detected in immersion fixed frozen sections of mouse embryo (15 dpc) using Goat Anti-Mouse P-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF761) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to connective tissue and lungs. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of P‑Cadherin in Human Liver.
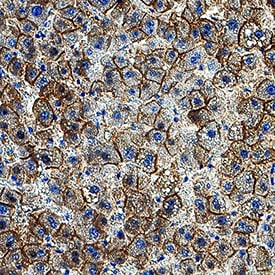
P‑Cadherin was detected in immersion fixed paraffin-embedded sections of human liver using Goat Anti-Mouse P‑Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF761) at 0.5 µg/ml for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using VisUCyte Antigen Retrieval Reagent-Basic (Catalog # VCTS021). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to the cell membrane of hepatocytes. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Mouse P‑Cadherin by Simple WesternTM.
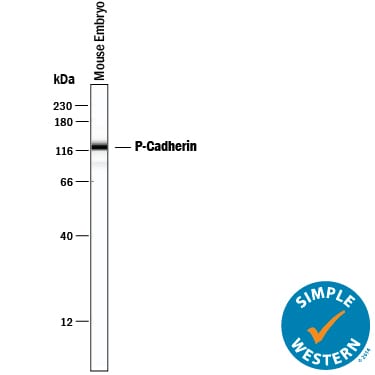
Simple Western lane view shows lysates of mouse embryo tissue, loaded at 0.2 mg/mL. A specific band was detected for P-Cadherin at approximately 115 kDa (as indicated) using 25 µg/mL of Goat Anti-Mouse P-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF761) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Mouse P-Cadherin by Western Blot
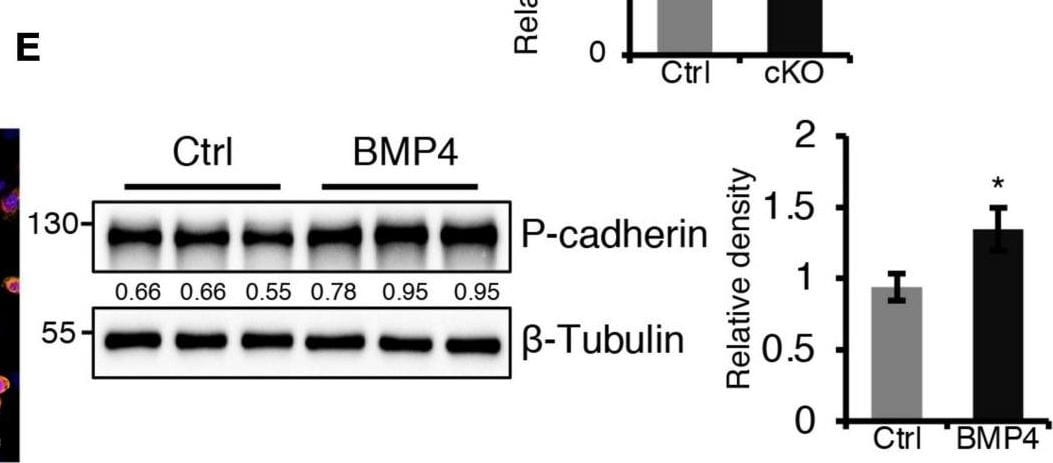
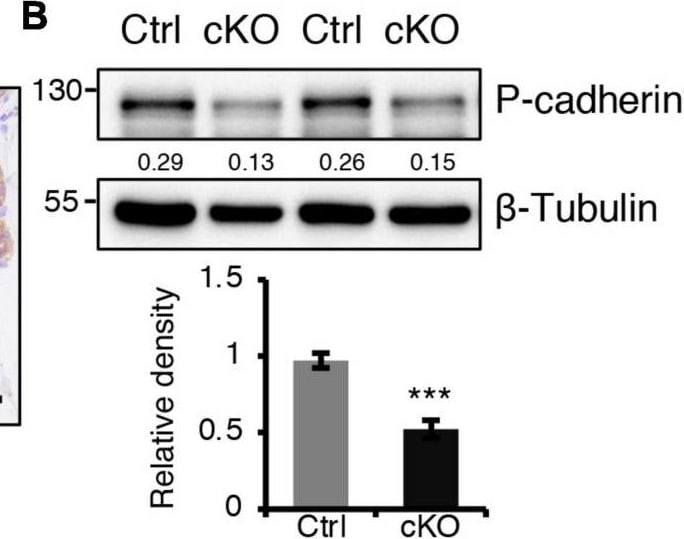
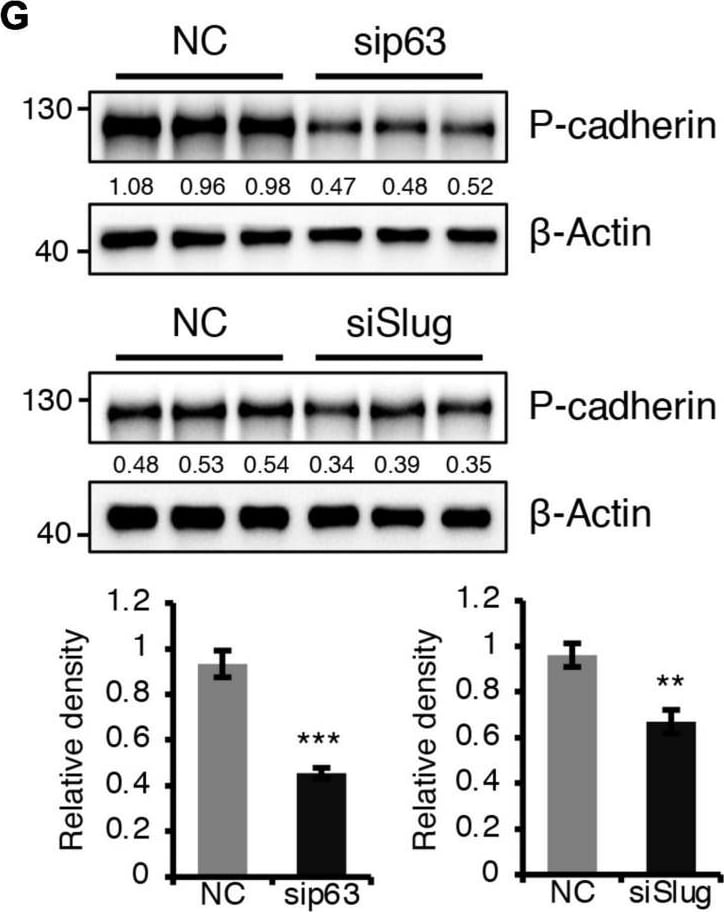
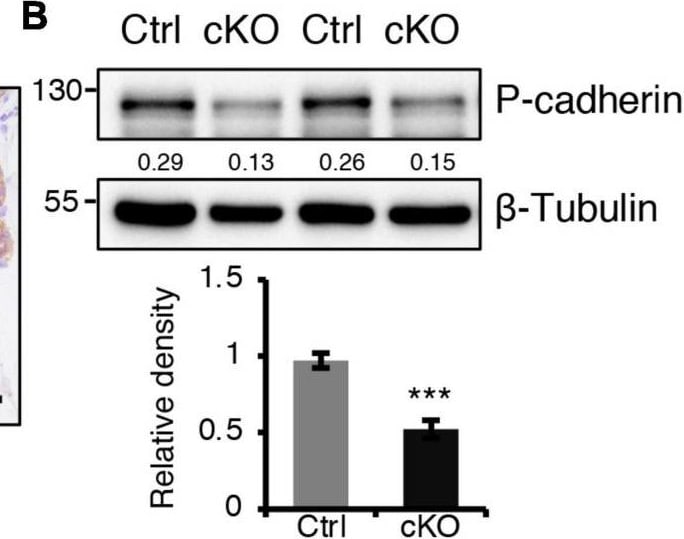
BMPR1a regulated P-cadherin expression via p63 and Slug. (A) Immunohistochemistry staining for P-cadherin in control (n = 4 mice) and cKO (n = 3 mice) mammary glands at pregnancy day 14.5. Scale bar, 50 μm. (B) Western blotting for P-cadherin in mammary epithelial cells isolated from control and cKO mice at pregnancy day 14.5. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 mice. (C) qRT-PCR analysis of Cdh3 in FACS-sorted control and cKO myoepithelial cells at pregnancy day 14.5. n = 4 biological replicates. (D) P-cadherin (red) and K14 (green) double immunofluorescence staining in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. n = 3 biological replicates. Scale bar, 25 μm. (E) Western blotting for P-cadherin in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 biological replicates. (F) Scatter plot showing the correlation between BMPR1A and CDH3 expression in mammary glands from TCGA and GTEx data. Pearson’s coefficient test was performed to assess statistical significance. (G) Western blotting for P-cadherin in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Actin. n = 3 biological replicates. (H) Immunofluorescence for P-cadherin (green) in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. n = 3 biological replicates. Scale bar, 25 μm. (I) Western blotting for K14, P-cadherin, p63 and Slug in HC11 cells treated with scramble RNA, p63 siRNA, Slug siRNA, or p63 siRNA and Slug siRNA at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of K14/ beta -Actin, P-cadherin/ beta -Actin, p63/ beta -Actin and Slug/ beta -Actin. n = 3 biological replicates. Data were presented as means ± SD. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34336839), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Western Blot
BMPR1a regulated P-cadherin expression via p63 and Slug. (A) Immunohistochemistry staining for P-cadherin in control (n = 4 mice) and cKO (n = 3 mice) mammary glands at pregnancy day 14.5. Scale bar, 50 μm. (B) Western blotting for P-cadherin in mammary epithelial cells isolated from control and cKO mice at pregnancy day 14.5. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 mice. (C) qRT-PCR analysis of Cdh3 in FACS-sorted control and cKO myoepithelial cells at pregnancy day 14.5. n = 4 biological replicates. (D) P-cadherin (red) and K14 (green) double immunofluorescence staining in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. n = 3 biological replicates. Scale bar, 25 μm. (E) Western blotting for P-cadherin in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 biological replicates. (F) Scatter plot showing the correlation between BMPR1A and CDH3 expression in mammary glands from TCGA and GTEx data. Pearson’s coefficient test was performed to assess statistical significance. (G) Western blotting for P-cadherin in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Actin. n = 3 biological replicates. (H) Immunofluorescence for P-cadherin (green) in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. n = 3 biological replicates. Scale bar, 25 μm. (I) Western blotting for K14, P-cadherin, p63 and Slug in HC11 cells treated with scramble RNA, p63 siRNA, Slug siRNA, or p63 siRNA and Slug siRNA at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of K14/ beta -Actin, P-cadherin/ beta -Actin, p63/ beta -Actin and Slug/ beta -Actin. n = 3 biological replicates. Data were presented as means ± SD. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34336839), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Western Blot
BMPR1a regulated P-cadherin expression via p63 and Slug. (A) Immunohistochemistry staining for P-cadherin in control (n = 4 mice) and cKO (n = 3 mice) mammary glands at pregnancy day 14.5. Scale bar, 50 μm. (B) Western blotting for P-cadherin in mammary epithelial cells isolated from control and cKO mice at pregnancy day 14.5. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 mice. (C) qRT-PCR analysis of Cdh3 in FACS-sorted control and cKO myoepithelial cells at pregnancy day 14.5. n = 4 biological replicates. (D) P-cadherin (red) and K14 (green) double immunofluorescence staining in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. n = 3 biological replicates. Scale bar, 25 μm. (E) Western blotting for P-cadherin in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 biological replicates. (F) Scatter plot showing the correlation between BMPR1A and CDH3 expression in mammary glands from TCGA and GTEx data. Pearson’s coefficient test was performed to assess statistical significance. (G) Western blotting for P-cadherin in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Actin. n = 3 biological replicates. (H) Immunofluorescence for P-cadherin (green) in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. n = 3 biological replicates. Scale bar, 25 μm. (I) Western blotting for K14, P-cadherin, p63 and Slug in HC11 cells treated with scramble RNA, p63 siRNA, Slug siRNA, or p63 siRNA and Slug siRNA at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of K14/ beta -Actin, P-cadherin/ beta -Actin, p63/ beta -Actin and Slug/ beta -Actin. n = 3 biological replicates. Data were presented as means ± SD. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34336839), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Western Blot
BMPR1a regulated P-cadherin expression via p63 and Slug. (A) Immunohistochemistry staining for P-cadherin in control (n = 4 mice) and cKO (n = 3 mice) mammary glands at pregnancy day 14.5. Scale bar, 50 μm. (B) Western blotting for P-cadherin in mammary epithelial cells isolated from control and cKO mice at pregnancy day 14.5. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 mice. (C) qRT-PCR analysis of Cdh3 in FACS-sorted control and cKO myoepithelial cells at pregnancy day 14.5. n = 4 biological replicates. (D) P-cadherin (red) and K14 (green) double immunofluorescence staining in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. n = 3 biological replicates. Scale bar, 25 μm. (E) Western blotting for P-cadherin in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 biological replicates. (F) Scatter plot showing the correlation between BMPR1A and CDH3 expression in mammary glands from TCGA and GTEx data. Pearson’s coefficient test was performed to assess statistical significance. (G) Western blotting for P-cadherin in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Actin. n = 3 biological replicates. (H) Immunofluorescence for P-cadherin (green) in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. n = 3 biological replicates. Scale bar, 25 μm. (I) Western blotting for K14, P-cadherin, p63 and Slug in HC11 cells treated with scramble RNA, p63 siRNA, Slug siRNA, or p63 siRNA and Slug siRNA at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of K14/ beta -Actin, P-cadherin/ beta -Actin, p63/ beta -Actin and Slug/ beta -Actin. n = 3 biological replicates. Data were presented as means ± SD. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34336839), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Western Blot
BMPR1a regulated P-cadherin expression via p63 and Slug. (A) Immunohistochemistry staining for P-cadherin in control (n = 4 mice) and cKO (n = 3 mice) mammary glands at pregnancy day 14.5. Scale bar, 50 μm. (B) Western blotting for P-cadherin in mammary epithelial cells isolated from control and cKO mice at pregnancy day 14.5. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 mice. (C) qRT-PCR analysis of Cdh3 in FACS-sorted control and cKO myoepithelial cells at pregnancy day 14.5. n = 4 biological replicates. (D) P-cadherin (red) and K14 (green) double immunofluorescence staining in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. n = 3 biological replicates. Scale bar, 25 μm. (E) Western blotting for P-cadherin in HC11 mammary epithelial cells treated with BMP4 (50 ng/mL) for 24 h. beta -Tubulin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Tubulin. n = 3 biological replicates. (F) Scatter plot showing the correlation between BMPR1A and CDH3 expression in mammary glands from TCGA and GTEx data. Pearson’s coefficient test was performed to assess statistical significance. (G) Western blotting for P-cadherin in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of P-cadherin/ beta -Actin. n = 3 biological replicates. (H) Immunofluorescence for P-cadherin (green) in HC11 cells treated with p63 siRNA (sip63)/Slug siRNA (siSlug) and scramble RNA (NC) at 48 h. n = 3 biological replicates. Scale bar, 25 μm. (I) Western blotting for K14, P-cadherin, p63 and Slug in HC11 cells treated with scramble RNA, p63 siRNA, Slug siRNA, or p63 siRNA and Slug siRNA at 48 h. beta -Actin was used as a loading control. Statistical analysis the expression of K14/ beta -Actin, P-cadherin/ beta -Actin, p63/ beta -Actin and Slug/ beta -Actin. n = 3 biological replicates. Data were presented as means ± SD. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34336839), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
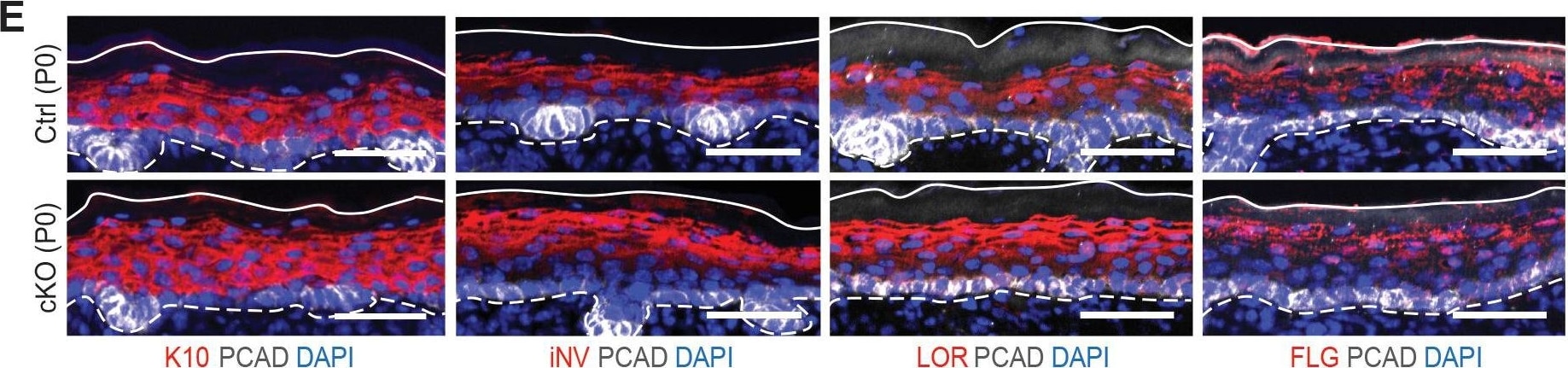
Additional analysis of epidermal perturbations upon Mettl3 cKO. (E) Images of P0 back skin sagittal sections immulabeled for K10, involucrin (iNV), loricrin (LOR)&filaggrin (FLG) demonstrating the expression pattern of standard differentiation markers is not severely interrupted by Mettl3 cKO (scale bars: 50 µm). Solid&dashed lines as in (B). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
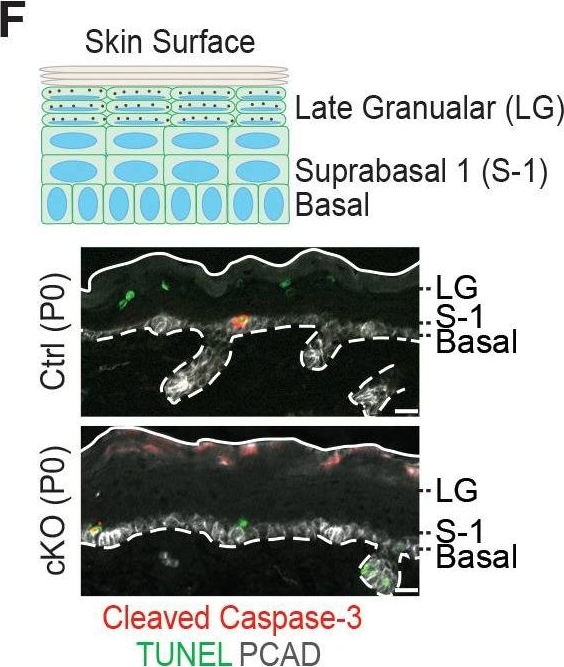
Additional analysis of epidermal perturbations upon Mettl3 cKO. (F) Representative images of P0 back skin sagittal sections with cleaved Caspase-3 immunostaining&TUNEL staining (scale bars: 20 µm). White solid lines indicate skin surface&dashed lines indicate dermal-epithelial border. Quantification of TUNEL+&cleaved Caspase-3+ cells in the designated layers at P0. Only double-positive cells denote apoptosis; TUNEL+ only cells are typically seen as granular cells lose their nuclei&transition as dead flattened cells to the stratum corneum. This is defective in Mettl3 cKO skin. The TUNEL+ cells in the suprabasal 1 layer likely correspond to the occasional cytolytic suprabasal cells that we saw at the ultrastructural level (not shown). Quantification of the labeled cells is shown (error bars: standard deviation, for each condition n = 3 biological replicates × 10 images per replicate, *p<0.05 and **p<0.01 by unpaired two-tailed Student’s t-test).Figure 6—figure supplement 2—source data 1.Quantification of PCAD, ECAD immunofluorescence signals in (A).Figure 6—figure supplement 2—source data 2.Quantification of EdU+ cells&the suprabasal/basal cell number ratio in (B).Figure 6—figure supplement 2—source data 3.Quantification of cell sizes by cytospin in (C).Figure 6—figure supplement 2—source data 4.Quantification of cell death events in epidermis in (F).Quantification of PCAD, ECAD immunofluorescence signals in (A).Quantification of EdU+ cells&the suprabasal/basal cell number ratio in (B).Quantification of cell sizes by cytospin in (C).Quantification of cell death events in epidermis in (F). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
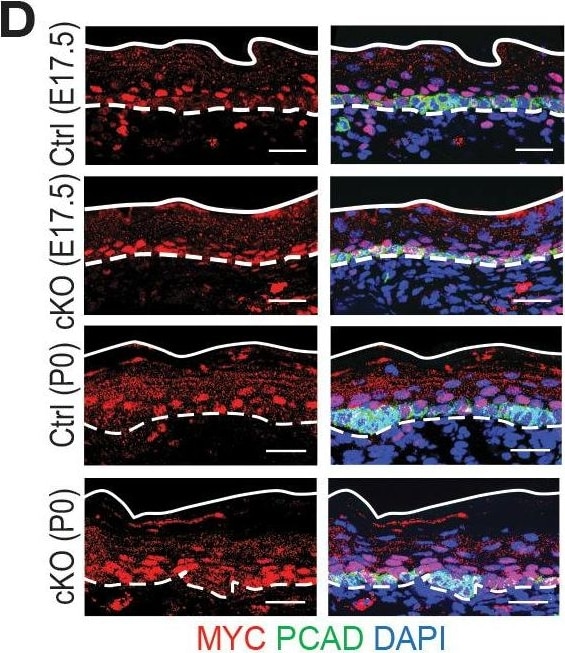
Additional analysis of features affected by the upregulated genes upon Mettl3 cKO.(A) ECDF plots of the Z score (cKO/Ctrl) from scRNA-seq demonstrate transcripts with higher levels of m6A modification (assessed by the coding sequence SN-uTPM per nt value from miCLIP) in wild-type tend to be more upregulated upon METTL3 loss. The p values were calculated through Wilcoxon rank sum test. (B, C) Violin plots illustrating the relative expression levels of Bmyc and Myc mRNA in corresponding groups of Mettl3 cKO versus control cells. Z score assessment of expressional difference between cKO and control [Z (cKO/Ctrl)] and false discovery rate (FDR) is calculated by MAST. The upregulation is verified with qPCR on total RNA samples extracted from YFP+ skin epithelial cells FACS isolated from E16.5 embryos with Tbp mRNA as internal control (error bars: standard deviation, for each condition n = 3 biological replicates, **p<0.01 by unpaired two-tailed Student’s t-test). (D) Images from E17.5 and P0 sagittal sections immunolabeled for MYC and PCAD (scale bars: 25 µm). White solid lines denote skin surface and dashed lines denote dermal:epidermal border. Quantifications of MYC immunofluorescence specifically in the Epi basal cells (identified based on PCAD staining) reveals the upregulation of MYC in cKO samples (for each condition n = 3 biological replicates ×7 images per replicate, *p<0.05 and ***p<0.001 by unpaired two-tailed Student’s t-test).Figure 7—figure supplement 1—source data 1.Bmyc qPCR in (B).Figure 7—figure supplement 1—source data 2.Myc qPCR in (C).Figure 7—figure supplement 1—source data 3.Quantification of MYC immunofluorescence signals in (D).Bmyc qPCR in (B).Myc qPCR in (C).Quantification of MYC immunofluorescence signals in (D). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
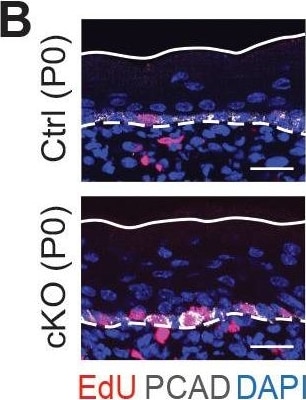
Additional analysis of epidermal perturbations upon Mettl3 cKO. (B) Left panel: representative images from P0 sagittal sections with a 45-min EdU pulse prior to tissue collection (scale bars: 25 µm). Solid lines, skin surface boundary; dashed lines, dermal-epidermal border. Middle panel: quantification of the ratio of EdU+ cells among all basal cells (for each condition n = 3 biological replicates ×10 images per replicate, ***p<0.001 by unpaired two-tailed Student’s t-test). Right panel: quantification of the suprabasal cell number/basal cell number ratio according to DAPI staining of the nuclei (for each condition n = 5 biological replicates ×10 images per replicate, ***p<0.001 by unpaired two-tailed Student’s t-test). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
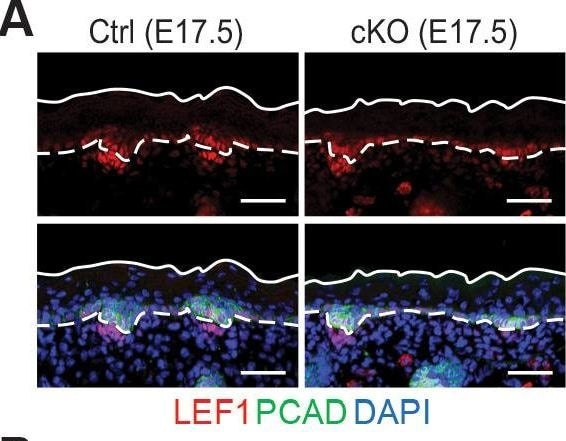
Loss of m6A results in diminished WNT signaling&signs of perturbed HF fate.(A) Left panel: representative images from E17.5 sagittal sections immunolabeled for LEF1&PCAD (scale bars: 50 µm). White solid lines denote skin surface&dashed lines denote dermal-epidermal border. Right panel: quantification of LEF1 immunofluorescence at indicated compartments (for each condition n = 3 biological replicates ×7 images per replicate, *p<0.05&***p<0.001 by unpaired two-tailed Student’s t-test). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
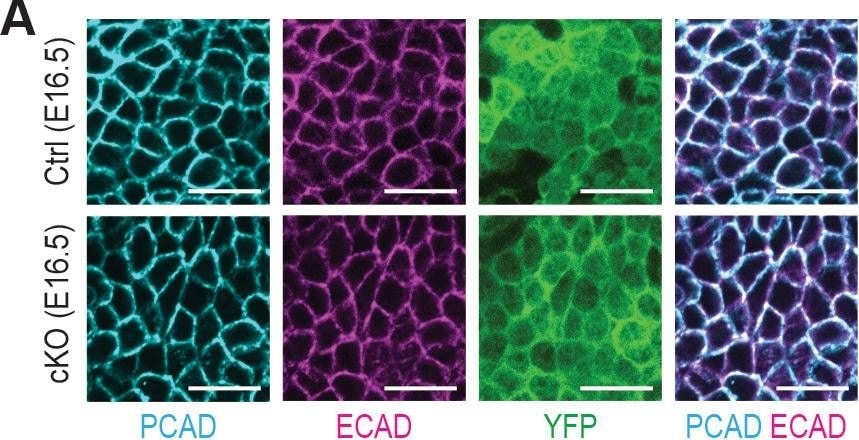
Additional analysis of epidermal perturbations upon Mettl3 cKO.(A) Confocal images of E16.5 whole-mount back skin immunolabeled for PCAD, ECAD&YFP (scale bars: 20 µm). PCAD&ECAD expression was quantified in the basal progenitors of skin epithelium (middle line corresponds to the median; for each condition the data are from two biological replicates; ****p<0.0001 by Mann Whitney test). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
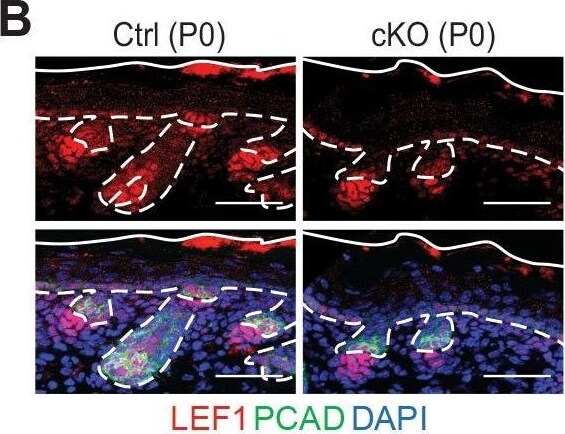
Loss of m6A results in diminished WNT signaling&signs of perturbed HF fate. (B) Left panel: representative images from P0 sagittal sections immunolabeled for LEF1&PCAD (scale bars: 50 µm). Solid&dashed lines as in (A). Right panel: quantifications of LEF1 immunofluorescence in indicated compartments (for each condition n = 3 biological replicates ×7 images per replicate, *p<0.05&***p<0.001 by unpaired two-tailed Student’s t-test). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
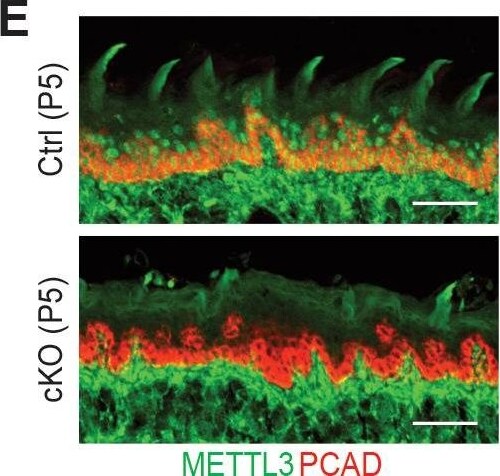
Additional information on the Mettl3 cKO phenotypes.(A) Representative pictures of P6 control and cKO littermates (scale bars: 1 cm). (B) Quantification of body weights of control and cKO littermates (error bars: standard deviation, for each condition n = 5 biological replicates from three litters). (C) Measurements of TEWL (error bars: standard deviation, for each condition n = 4 biological replicates, for the comparison between control and cKO at each time point, p>0.05 by unpaired two-tailed Student’s t-test). (D) Representative pictures of P5 pups’ mouths demonstrating the growth of the teeth (black arrows). (E) Images of P5 tongue sagittal sections immunolabeled for METTL3 and PCAD demonstrating the tongue surface of the Mettl3 cKO animals tends to be smoother than that of the control animals, which suggests filiform papillae are not well-formed in the cKO animals (scale bars: 50 µm). (F) Hematoxylin and eosin (H and E) stained P6 back skin sagittal sections (scale bars: 100 µm).Figure 2—figure supplement 2—source data 1.Quantification of neonates' body weights in (B).Figure 2—figure supplement 2—source data 2.Quantification of TEWL in (C).Quantification of neonates' body weights in (B).Quantification of TEWL in (C). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse P-Cadherin by Immunohistochemistry
Krt14-Cre driven conditional Mettl3 knockout mice display severe defects in HF morphogenesis. (B) Confocal images of E16.5 whole-mount back skin immunolabeled for P-cadherin (PCAD), METTL3&YFP (scale bars: 20 µm). Note that nuclear METTL3 immunofluorescence is selectively depleted from the YFP+ cells in cKO skin. White dashed lines denote the dermal-epidermal border. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32845239), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse P‑Cadherin Antibody
Application
Recommended Usage
Adhesion Blockade
The adhesion of A431 human epithelial carcinoma cells (1 x 105 cells/well) to immobilized Recombinant Mouse P-Cadherin Fc Chimera (Catalog # 761-MP, 10 µg/mL, 100 µL/well) was maximally inhibited (80-100%) by 50 µg/mL of the antibody.
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: XB2 mouse teratoma keratinocyte cell line
Sample: XB2 mouse teratoma keratinocyte cell line
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed XB2 mouse teratoma keratinocyte cell line and A431 human epithelial carcinoma cell line
Sample: Immersion fixed XB2 mouse teratoma keratinocyte cell line and A431 human epithelial carcinoma cell line
Immunohistochemistry
0.5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (15 dpc) and immersion fixed paraffin-embedded sections of human liver.
Sample: Immersion fixed frozen sections of mouse embryo (15 dpc) and immersion fixed paraffin-embedded sections of human liver.
Simple Western
25 µg/mL
Sample: Mouse embryo tissue
Sample: Mouse embryo tissue
Western Blot
0.5 µg/mL
Sample: Mouse embryo tissue
Sample: Mouse embryo tissue
Mouse P-Cadherin Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Reviewed Applications
Read 1 review rated 3 using AF761 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: P-Cadherin
References
- Bussemakers, M.J.G. et al. (1993) Mol. Biol. Reports 17:123.
- Overduin, M. et al. (1995) Science 267:386.
- Takeichi, M. (1991) Science 251:1451.
- Nose, A. et al. (1987) EMBO J. 6:3655.
Long Name
Placental Cadherin
Alternate Names
CAD3, Cadherin-3, CDH3, CDHP, HJMD, PCAD, PCadherin
Gene Symbol
CDH3
UniProt
Additional P-Cadherin Products
Product Documents for Mouse P‑Cadherin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse P‑Cadherin Antibody
For research use only
Related Research Areas
Citations for Mouse P‑Cadherin Antibody
Customer Reviews for Mouse P‑Cadherin Antibody (1)
3 out of 5
1 Customer Rating
Have you used Mouse P‑Cadherin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunofluorescenceSample Tested: See PMID 20826529Species: MouseVerified Customer | Posted 01/09/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...