The cadherin superfamily comprises a large number of membrane glycoproteins with one or more cadherin repeats, which are involved in Ca2+ dependent cell-cell adhesion. The family can be subdivided into several major subgroups, including the type I and type II classical cadherins, desmosomal cadherins, protocadherins, seven transmembrane (Flamingo) cadherins, FAT-family cadherins, T-cadherin and other unclassified cadherins (1). Cadherin-11, also known as OB-cadherin, is a type II classical cadherin. Classical cadherins are type I transmembrane proteins with an N-terminal extracellular domain containing five tandem cadherin repeats and a C-terminal cytoplasmic domain with a characteristic sequence for binding to catenins. Type I cadherins (E-, N-, P-, R-, M-, and EP-cadherin) differ from type II cadherins (cadherin-5 to -12, -18 to -20 and -22) by the presence of the HAV tripeptide motif in the most N-terminal cadherin repeat (2). Classic cadherins mediate cell-cell adhesion preferentially via homotypic interactions and form adherens juctions that have beta -catenin and p120 (ctn) at the cytoplasmic side of the junction (3, 4). Homotypic cadherin interactions also transduce outside-in and inside-out cell signals. Cadherin signaling induces various cellular processes including cell motility, actin cytoskeleton reorganization, proliferation, and differentiation (3, 4). Cadherin-11 is expressed in a variety or normal tissues of mesodermal origin including areas of the kidney and brain, in normal osteoblasts, and in tumors of the stomach, kidney, colon, breast, and bone (osteosarcoma) (5, 6). It is also differentially expressed in the embryonic brain and may be important in regulating neural development. Human Cadherin-11 exhibits a unique mRNA splice site allowing for two forms of the protein to be expressed, a full-length 796 amino acid (aa) protein and a COOH terminus-truncated variant of 693 aa. The truncated variant has a unique cytoplasmic region due to a frameshift event (3). The full-length human and mouse Cadherin-11 share 97% homology at the aa sequence level.

Key Product Details
Validated by
Knockout/Knockdown
Species Reactivity
Validated:
Human
Cited:
Human, Canine
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Neutralization, Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 283416
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant human Cadherin‑11
Phe23-Thr617
Accession # AAA35622
Phe23-Thr617
Accession # AAA35622
Specificity
Detects human Cadherin-11 in direct ELISAs and Western blots. Does not cross-react with recombinant human (rh) Cadherin‑4, rhCadherin‑8, rhCadherin‑12, rhCadherin‑13, rhCadherin‑17, rhE-Cadherin, rhN-Cadherin, rhP-Cadherin, or rhVE-Cadherin.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human Cadherin‑11 Antibody
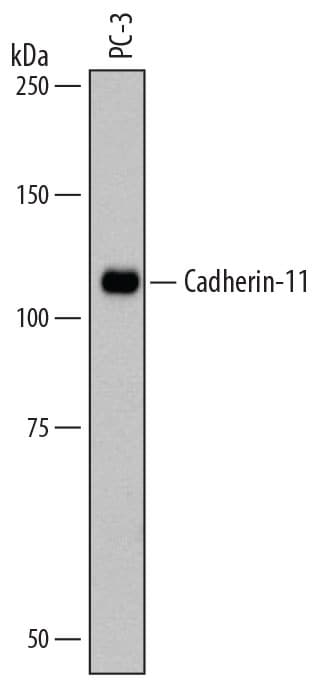
Detection of Human Cadherin‑11 by Western Blot.
Western blot shows lysates of PC-3 human prostate cancer cell line. PVDF membrane was probed with 1 µg/mL of Mouse Anti-Human Cadherin-11 Monoclonal Antibody (Catalog # MAB1790) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF007). A specific band was detected for Cadherin-11 at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Cadherin‑11 in Human Placenta.
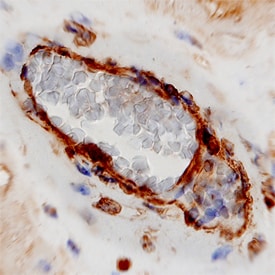
Cadherin-11 was detected in immersion fixed paraffin-embedded sections of human placenta using Mouse Anti-Human Cadherin-11 Monoclonal Antibody (Catalog # MAB1790) at 8 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Cadherin‑11 by Simple WesternTM.
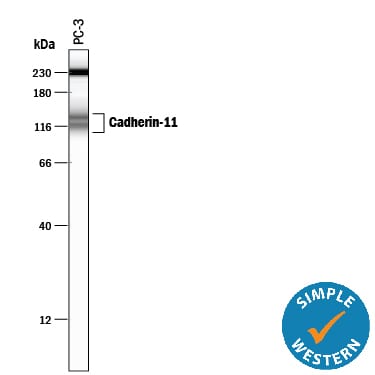
Simple Western lane view shows lysates of PC‑3 human prostate cancer cell line, loaded at 0.2 mg/mL. Specific bands were detected for Cadherin‑11 at approximately 114 & 137 kDa (as indicated) using 20 µg/mL of Mouse Anti-Human Cadherin‑11 Monoclonal Antibody (Catalog # MAB1790). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system. Non-specific interaction with the 230 kDa Simple Western standard may be seen with this antibody.Detection of Human Cadherin-11 by Western Blot
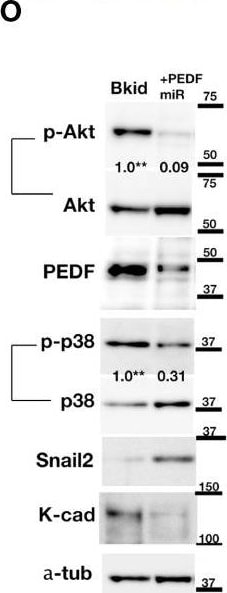
PEDF overexpression promoted the MET-like behavior in pulmonary metastasis. (A–K, M) The results of higher doses (2 x 106 cells) injections into the knee joints. (A)In vivo luciferase-imaging of 143B/143B+PEDF cells after 28 days from the injection. The signals were observed in the lung of 143B+PEDF cell-injected mice (right, six positive/10: one dead), while no signal was seen in thorasic region of 143B-injected mice (left, zero positive/nine). (B, C) The H&E stain of knee joint and femur section. The scale bar indicates 1mm. (B’, C’) SPARC-IHC of the adjacent section of (B, C). The scale bar indicates 1 mm. (B) The 143B cells (ca; arrow) were mainly observed outside of the knee bone. (B’) SPARC-IHC. The cells outside of the joint were osteosarcomas. (C) 143B+PEDF cells were observed inside of the bone (ca; arrow). (C’) SPARC-IHC of (C). The SPARC-positive cancer cells were seen both in the bone marrow (bm) and the joint. The opened arrowheads mean the position of the bone. (D) The H&E stain of the pulmonary metastatic lesion of the 143B+PEDF-injected mouse. The square of the broken line is the region of interest in (E–G). (E–G) The lung vascular border in 143B+PEDF cell-injected mouse at higher magnification. The cancer cells inside and outside the vasculature are connected with each other. The scale bar indicates 50 µm. (E) shows H&E staining of these sections. (F) shows IHC for CD31 on a similar section. The endothelial cell layer is very thin. (G) shows IHC for PEDF. PEDF-positive cells are present both inside and outside the vein. (H–K, M) The 143B+PEDF tumor in the lung at higher magnification. The scale bar indicates 50 µm. (H) shows H&E staining of the inside and outside of vasculature. (I) shows IHC for CD31 indicating the endothelial layer. (J) shows IHC for beta -catenin on both sides of the boundary. (K) shows IHC for PEDF also on both the sides. (L) IHC for beta -catenin-in the normal bronchial tube near the tumor lesion. (M) IHC for beta -catenin in the tumor far away from the vascular boundary. Arrowheads indicate strong staining on cell–cell junctions. (N) Immunoblot of 143B and Bkid cell lysates. OB-cadherin and K-cadherin was upregulated, whereas Snail2 was downregulated. The ratios of phosphorylated to whole Akt for each sample are shown between the blots. The ratio of phosphorylated to whole p38MAPK, and the ratio of phosphorylated to whole ERK1/2 are also shown between each blot. Expression of MMP9 was diminished in Bkid cells, whereas that of MMP14, MMP15, and LPP was decreased. (O) Comparisons between Bkid and Bkid+PEDFmiR cell lysates. The amount of PEDF were reduced. The ratios of phosphorylated to whole Akt and phospho-p38MAPK to whole p38 are also shown between the blots. The phosphorylation level decreased upon PEDF knockdown. Snail2 was upregulated in Bkid+PEDFmiR, whereas K-cadherin was downregulated. (P) Zymography indicating that metalloprotease activity was increased upon PEDF knockdown. The upregulated MMP was mainly MMP9. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35174090), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Cadherin‑11 Antibody
Application
Recommended Usage
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human placenta
Sample: Immersion fixed paraffin-embedded sections of human placenta
Simple Western
20 µg/mL
Sample: PC‑3 human prostate cancer cell line
Sample: PC‑3 human prostate cancer cell line
Western Blot
1 µg/mL
Sample: PC‑3 human prostate cancer cell line
Sample: PC‑3 human prostate cancer cell line
Reviewed Applications
Read 1 review rated 5 using MAB1790 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cadherin-11
References
- Angst, B.D. et al. (2001) J. Cell Sci. 113:629.
- Gessner, R. and R. Tauber (2000) Ann. N.Y. Acad. Sci. 915:136.
- Feltes, C.M. et al. (2002) Cancer Research. 62:6688.
- Wheelock, J.J. and K.R. Johnson (2003) Annu. Rev. Cell Dev. Biol. 19:207.
- Hoffmann, I. and R. Balling (1995) Dev. Biol. 169:337.
- Pishvaian, M.J. et. al. (1999) Cancer Research 59:947.
Alternate Names
Cadherin11, CDH11, CDHOB, OB-Cadherin
Gene Symbol
CDH11
UniProt
Additional Cadherin-11 Products
Product Documents for Human Cadherin‑11 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Cadherin‑11 Antibody
For research use only
Related Research Areas
Citations for Human Cadherin‑11 Antibody
Customer Reviews for Human Cadherin‑11 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human Cadherin‑11 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
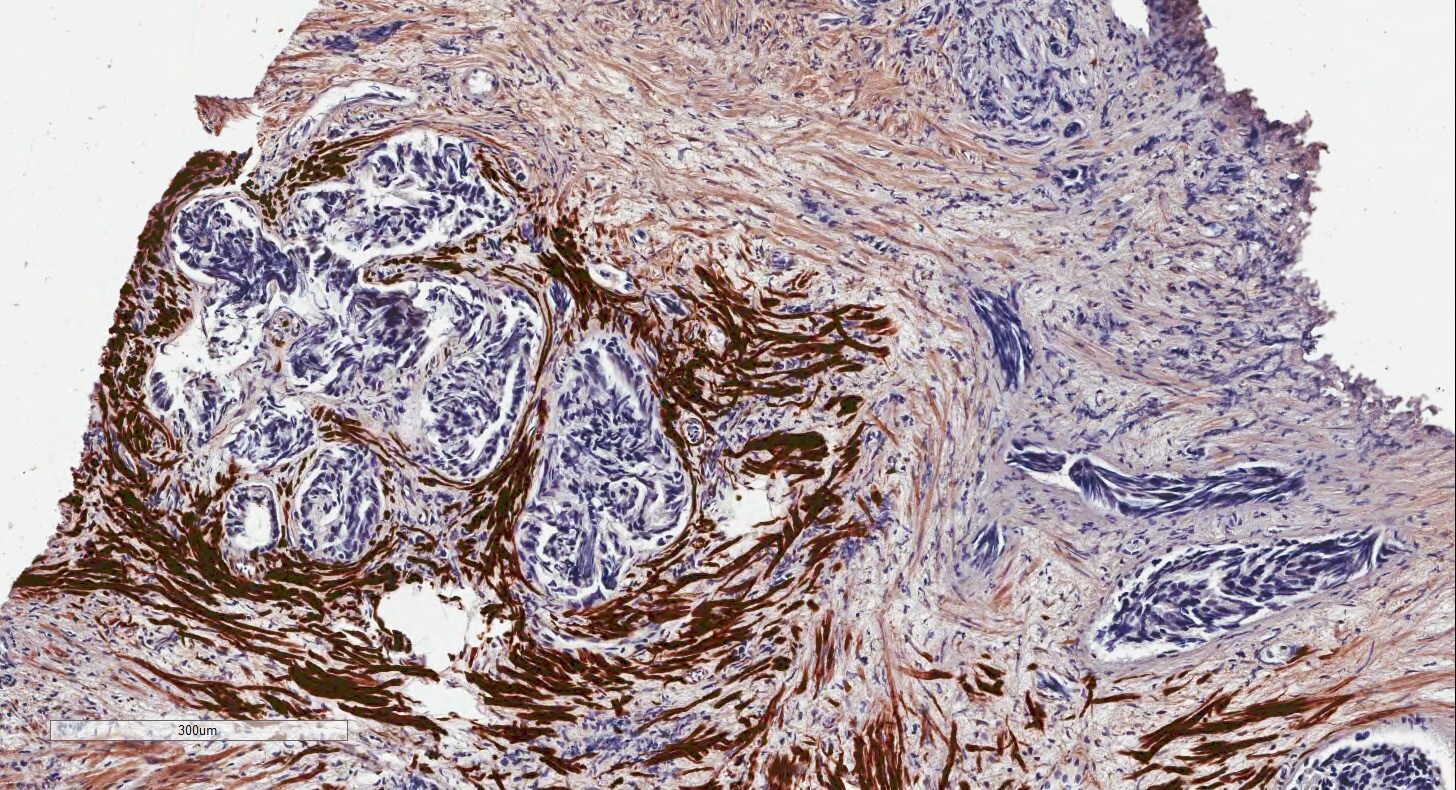
Application: Immunohistochemistry-ParaffinSample Tested: prostateSpecies: HumanVerified Customer | Posted 09/28/2015prostate
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...