Cathepsin S is a lysosomal cysteine protease of the papain family (1). It plays a major role in the processing of the MHC class II-associated invariant chain (2). It has been implicated in the pathogenesis of several diseases such as Alzheimer’s disease and degenerative disorders associated with the cells of the mononuclear phagocytic system (1). Human Cathepsin S is synthesized as a preproenzyme of 331 amino acid residues consisting a signal peptide (residues 1-16), a pro region (residues 17-114), and the mature enzyme (residues 115-331) (3-5). Cathepsin S is less abundant in tissues than Cathepsins B, L and H. The highest levels have been found in lymph nodes, spleen, macrophages, and other phagocytic cells.

Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Avian - Chicken, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization, Immunoprecipitation
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Flow Cytometry, Immunoprecipitation, Luminex Development
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Cathepsin S
Gln17-Ile331
Accession # P25774
Gln17-Ile331
Accession # P25774
Specificity
Detects human Cathepsin S in ELISAs and Western blots. In sandwich ELISAs, less than 0.05% cross-reactivity with recombinant human Cathepsin A, B, C, D, E, L, V, and X/Z/P is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Cathepsin S Antibody
Cathepsin S in Human Lymph Node.
Cathepsin S was detected in immersion fixed paraffin-embedded sections of human lymph node using Goat Anti-Human Cathepsin S Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1183) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of immersion fixed paraffin-embedded Tissue Sections.Detection of Human Cathepsin S by Western Blot
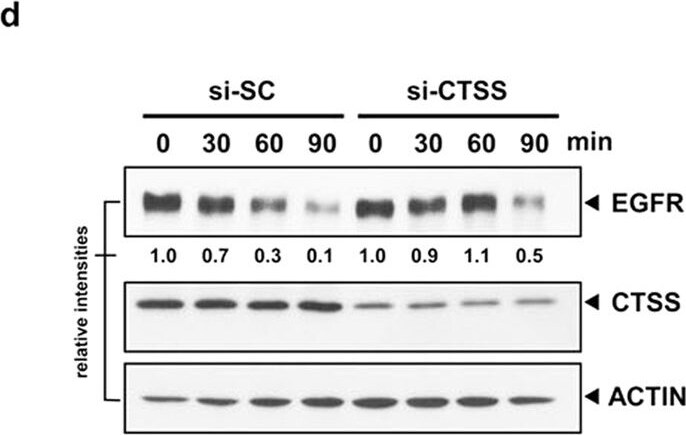
CTSS attenuates EGF-mediated EGFR degradation.(a) OEC-M1 and MDA-MB-231 cells were pretreated with 20 μM 6r or ZFL for 1 h and subsequently incubated with 100 ng/mL EGF for an additional 2 h. The total cell lysates were analysed using EGFR-specific antibodies. ACTIN was used as the internal control for semiquantitative loading in each lane. (b) The cells were stimulated with EGF (100 ng/mL) with or without the pretreatment of 20 μM 6r for the indicated durations. EGFR degradation was examined through immunostaining by using an anti-EGFR antibody. Notably, a substantial amount of EGFR was detectable even after 6 h of EGF stimulation in 6r-treated cells. (c) The OEC-M1 cells were transiently transfected with plasmids (pCMV) that encoded wild-type CTSS. After 24 h of transfection, the cells were treated with 100 ng/mL EGF for the indicated durations and the cellular EGFR, CTSS, and ACTIN signals were determined through Western blotting. The lifespan of EGF-mediated EGFR degradation was calculated by normalising the signal intensity of EGFR with that of ACTIN. (d) The MDA-MB-231 cells were transfected with specific 50 nM CTSS siRNA (si-CTSS) for 24 h and subsequently incubated with 100 ng/mL EGF for the indicated durations. The nontargeting scramble siRNA (si-SC) was used as the scramble control. (e) The MDA-MB-231 cells were transiently transfected with plasmids encoding the CTSS-C25A mutant. After 24 h of transfection, the cells were incubated with 100 ng/mL of EGF for the indicated durations. Furthermore, the cells were harvested and subjected to SDS-PAGE and Western blotting. EGFR degradation was determined using an antibody against EGFR. ACTIN signalling was included as the loading control. Image collected and cropped by CiteAb from the following open publication (https://www.nature.com/articles/srep29256), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Cathepsin S by Western Blot
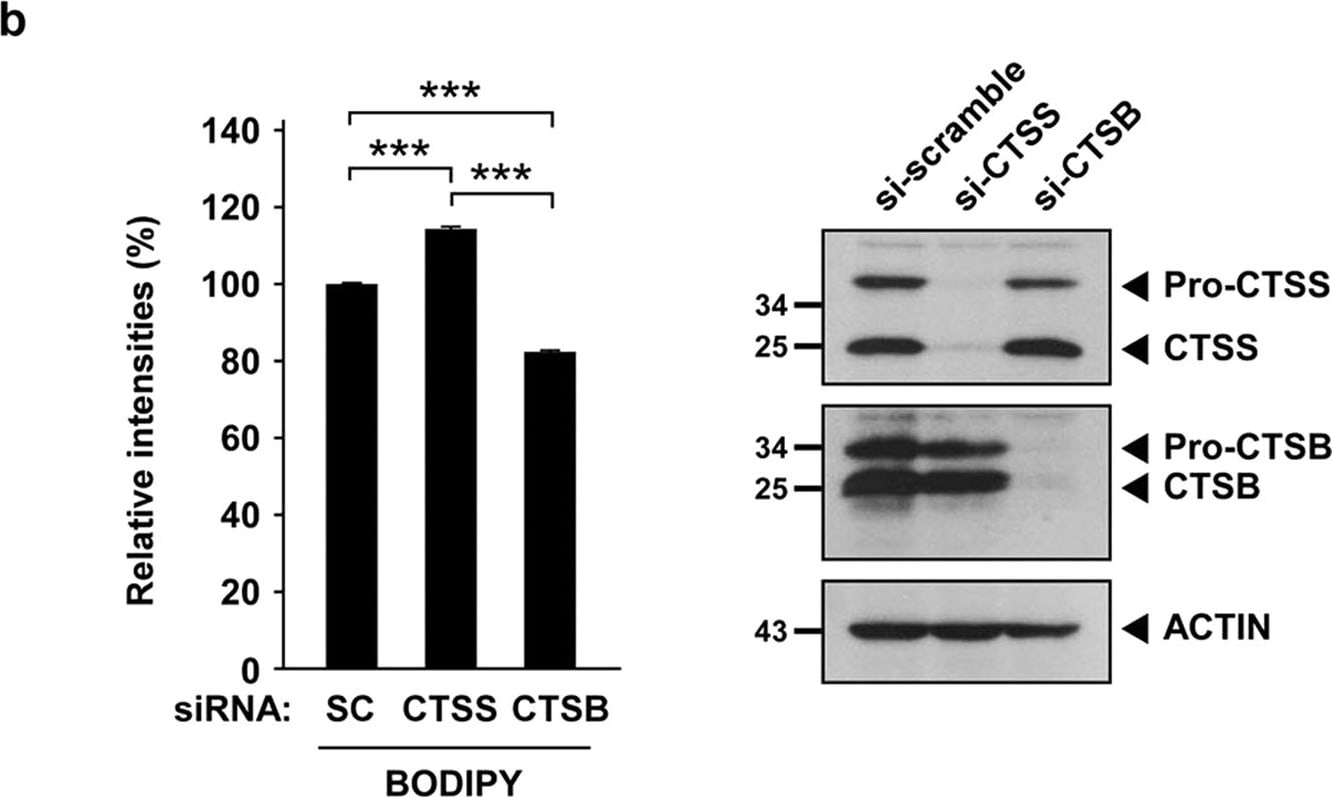
CTSS inhibition does not impair lysosomal activities.(a) OEC-M1 cells were pretreated with vesicle or 100 nM BAF for 1 h and then incubated with 20 μM 6r for 1 h further. Lysosomal proteolytic activities were determined using BODIPY–BSA and quantified through flow cytometry. (b) After 48 h of siRNA knockdown of CTSS and CTSB, the relative expression of CTSS and CTSB were determined through Western blotting (right panel). Lysosomal proteolytic activities were determined using BODIPY–BSA and quantified through flow cytometry (right panel). Data represent the mean ± SD of three independent experiments. Differences were found to be statistically significant at ***P < 0.001. Image collected and cropped by CiteAb from the following open publication (https://www.nature.com/articles/srep29256), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin S by Immunocytochemistry/ Immunofluorescence
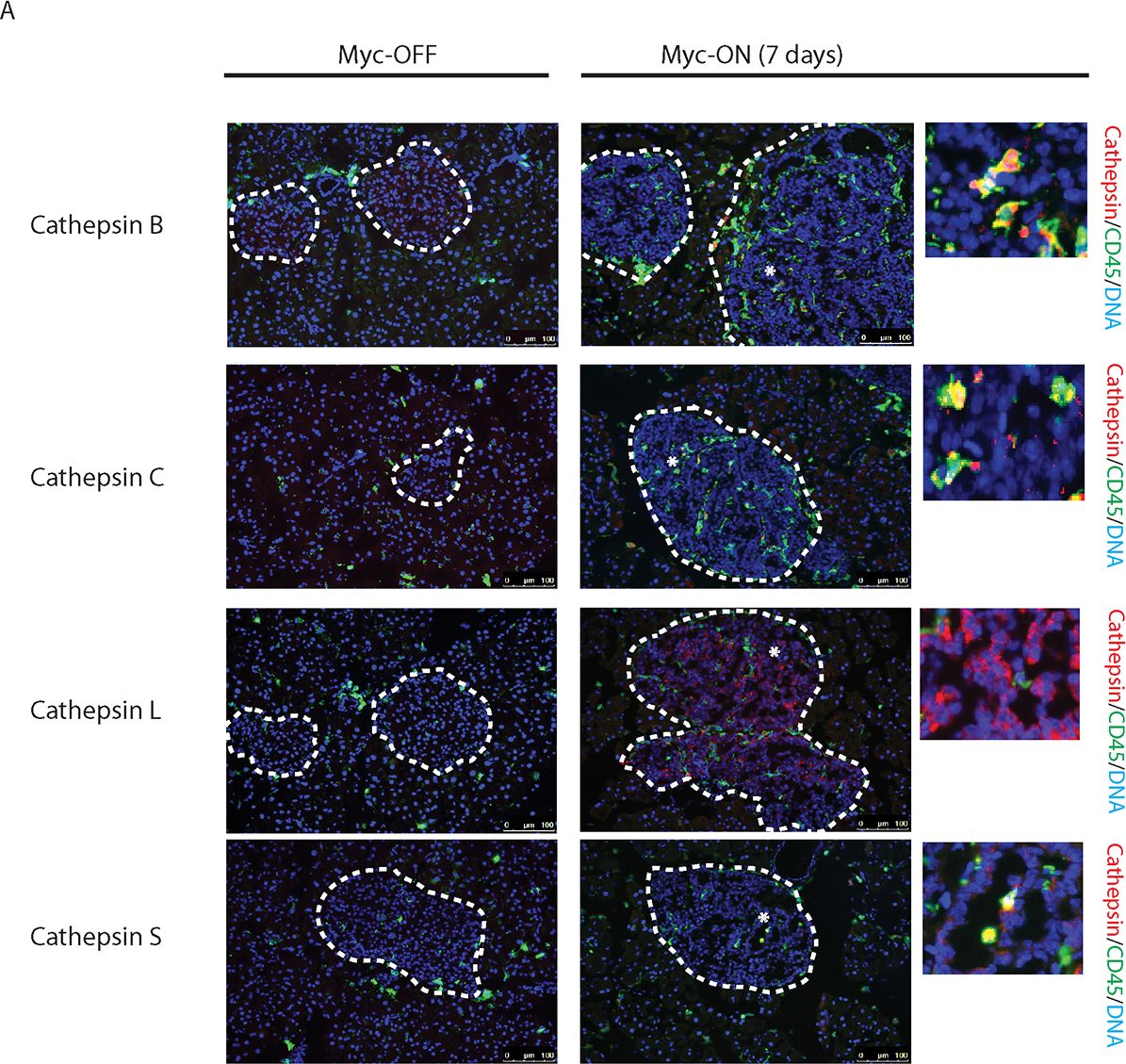
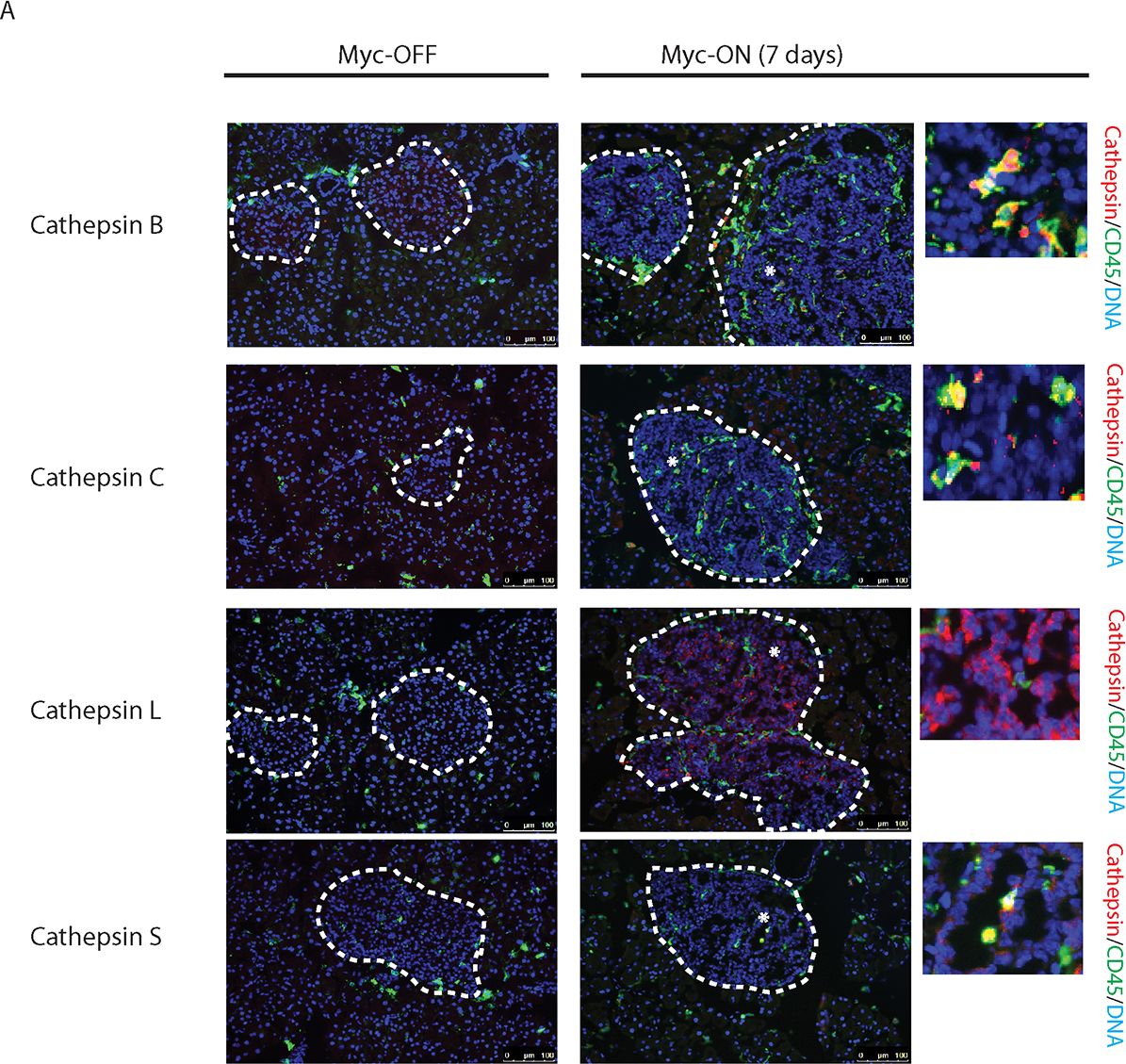
Myc induces cathepsin L expression in beta-cells of pancreatic Islets.(A) Immunohistochemical analyses for CTS B, C, L or S expression (all in red) in combination with staining for the pan-leukocyte marker CD45 (green) in pancreatic islet tumors from the MycERTAM;Bcl-xL animals. Pancreata were harvested from the MycERTAM;Bcl-xL mice treated for 7 d with TAM (Myc-On, 7 days) or control vehicle in place of TAM (Myc-OFF). The islet area is indicated by dotted line. The asterisks indicate the area of tumor represented in the insets. The panels are representatives of at least three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; eight randomized fields per analysis were examined. Scale bars, 100μm. (B) Immunohistochemical analysis for cathepsin L expression in beta-cells of pancreatic islets from MycERTAM;Bcl-xL animals identified by insulin expression. Pancreata were collected from the animals described above. Scale bars represent 25μm. The panels are representatives of three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; ten randomized fields per analysis were examined. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0120348), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin S by Immunocytochemistry/ Immunofluorescence
Myc induces cathepsin L expression in beta-cells of pancreatic Islets.(A) Immunohistochemical analyses for CTS B, C, L or S expression (all in red) in combination with staining for the pan-leukocyte marker CD45 (green) in pancreatic islet tumors from the MycERTAM;Bcl-xL animals. Pancreata were harvested from the MycERTAM;Bcl-xL mice treated for 7 d with TAM (Myc-On, 7 days) or control vehicle in place of TAM (Myc-OFF). The islet area is indicated by dotted line. The asterisks indicate the area of tumor represented in the insets. The panels are representatives of at least three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; eight randomized fields per analysis were examined. Scale bars, 100μm. (B) Immunohistochemical analysis for cathepsin L expression in beta-cells of pancreatic islets from MycERTAM;Bcl-xL animals identified by insulin expression. Pancreata were collected from the animals described above. Scale bars represent 25μm. The panels are representatives of three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; ten randomized fields per analysis were examined. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0120348), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cathepsin S by Western Blot
Proteolytic cleavage of EGFR by CTSS.(a) Recombinant EGFR was incubated with CTSS (left panel) and CTSB (right panel) at 37 °C for the indicated durations. The reaction was stopped by adding a sample buffer, and the reaction mixtures were subjected to SDS-PAGE, followed by Western blotting with an EGFR antibody. (b) Recombinant EGFR was incubated with CTSS in the presence of a CTSS inhibitor 6r or ZFL. The reaction mixtures were incubated at 37 °C for 20 min and then subjected to SDS-PAGE. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27387133), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cathepsin S by Western Blot
CTSS attenuates EGF-mediated EGFR degradation.(a) OEC-M1 and MDA-MB-231 cells were pretreated with 20 μM 6r or ZFL for 1 h and subsequently incubated with 100 ng/mL EGF for an additional 2 h. The total cell lysates were analysed using EGFR-specific antibodies. ACTIN was used as the internal control for semiquantitative loading in each lane. (b) The cells were stimulated with EGF (100 ng/mL) with or without the pretreatment of 20 μM 6r for the indicated durations. EGFR degradation was examined through immunostaining by using an anti-EGFR antibody. Notably, a substantial amount of EGFR was detectable even after 6 h of EGF stimulation in 6r-treated cells. (c) The OEC-M1 cells were transiently transfected with plasmids (pCMV) that encoded wild-type CTSS. After 24 h of transfection, the cells were treated with 100 ng/mL EGF for the indicated durations and the cellular EGFR, CTSS, and ACTIN signals were determined through Western blotting. The lifespan of EGF-mediated EGFR degradation was calculated by normalising the signal intensity of EGFR with that of ACTIN. (d) The MDA-MB-231 cells were transfected with specific 50 nM CTSS siRNA (si-CTSS) for 24 h and subsequently incubated with 100 ng/mL EGF for the indicated durations. The nontargeting scramble siRNA (si-SC) was used as the scramble control. (e) The MDA-MB-231 cells were transiently transfected with plasmids encoding the CTSS-C25A mutant. After 24 h of transfection, the cells were incubated with 100 ng/mL of EGF for the indicated durations. Furthermore, the cells were harvested and subjected to SDS-PAGE and Western blotting. EGFR degradation was determined using an antibody against EGFR. ACTIN signalling was included as the loading control. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27387133), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Cathepsin S Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lymph node
Sample: Immersion fixed paraffin-embedded sections of human lymph node
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Recombinant Human Cathepsin S (Catalog # 1183-CY), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Recombinant Human Cathepsin S (Catalog # 1183-CY), see our available Western blot detection antibodies
Western Blot
0.1 µg/mL
Sample: Recombinant Human Cathepsin S (Catalog # 1183-CY)
Sample: Recombinant Human Cathepsin S (Catalog # 1183-CY)
Neutralization
Measured by its ability to neutralize activation and the resulting activity of Recombinant Human Cathepsin S (0.25 µg/mL, Catalog # 1183-CY) in cleaving the fluorogenic peptide substrate Mca-RPKPVE-Nval-WRK(Dnp)-NH2 (10 µM, Catalog # ES002). The Neutralization Dose (ND50) is typically 0.2-1.8 µg/mL.
Human Cathepsin S Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Reviewed Applications
Read 2 reviews rated 5 using AF1183 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cathepsin S
References
- Kirschke, H. (2004) in Handbook of Proteolytic Enzymes (ed. Barrett, A.J. et al.) pp. 1104 - 1107, Academic Press, San Diego.
- Turk, V. et al. (2001) EMBO J. 20:4629.
- Shi, G.P. et al. (1992) J. Biol. Chem. 267:7258.
- Shi, G.P. et al. (1994) J. Biol. Chem. 269:11530.
- Wiederanders, B. et al. (1992) J. Biol. Chem. 267:13708.
Alternate Names
CTSS
Entrez Gene IDs
1520 (Human)
Gene Symbol
CTSS
UniProt
Additional Cathepsin S Products
Product Documents for Human Cathepsin S Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Cathepsin S Antibody
For research use only
Related Research Areas
Citations for Human Cathepsin S Antibody
Customer Reviews for Human Cathepsin S Antibody (2)
5 out of 5
2 Customer Ratings
Have you used Human Cathepsin S Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: Simple WesternSample Tested: ARP-1 human myeloma cell line and JJN3 human myeloma cell lineSpecies: HumanVerified Customer | Posted 11/12/2020
-
Application: ELISASample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 12/01/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...