CD3 epsilon is one of at least four invariant proteins that associate with the variable antigen recognition chains of the T cell receptor and function in signal transduction.
Best Seller
Human CD3 epsilon Antibody
R&D Systems | Catalog # MAB100
Clone UCHT1 was used by HLDA to establish CD designation

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Feline, Primate
Applications
Validated:
Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, T Cell Stimulation, CyTOF-reported
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Neutralization, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, Binding Assay, Bioassay, Cell Culture, ELISA Development, In vitro assay, Functional Assay, Stimulation, T Cell Activation, Tissue Culture
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # UCHT1
Loading...
Product Specifications
Immunogen
Human thymocytes (1)
Specificity
Recognizes the epsilon -chain of the CD3/T cell antigen receptor complex (McMichael, A.J. et al. (1987) Leucocyte Typing III: White Cell Differentiation Antigens, Oxford University Press, New York; Knapp, W. et al. (1989) Leucocyte Typing IV: White Cell Differentiation Antigens, Oxford University Press, New York; Schlossman, S. et al. (1995) Leucocyte Typing V: White Cell Differentiation Antigens, Oxford University Press, New York).
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human CD3 epsilon Antibody
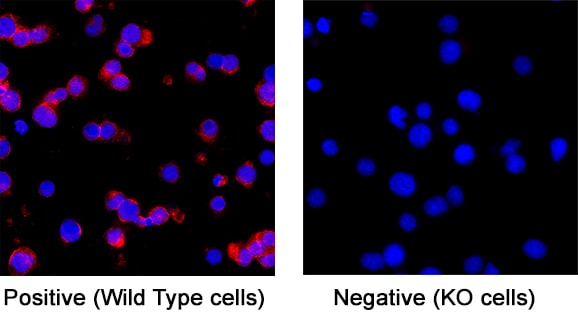
CD3 epsilon Specificity is Shown by Immunocytochemistry in Knockout Cell Line
CD3 epsilon was detected in immersion fixed Jurkat human acute T cell leukemia cell line (Positive) and absent in Jurkat CD3 epsilon Knockout (Negative) control using Mouse Anti-Human CD3 epsilon Monoclonal Antibody (Catalog # MAB100) at 3 µg/ml for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to the cell membrane of wildtype Jurkat cells. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.CD3 epsilon in Human PBMCs.
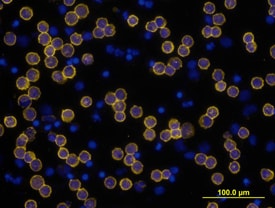
CD3e was detected in immersion fixed human peripheral blood mononuclear cells (PBMCs) using Mouse Anti-Human CD3e Monoclonal Antibody (Catalog # MAB100) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (yellow; Catalog # NL007) and counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of Human CD3 epsilon by Western Blot.
Western blot shows lysates of MOLT‑4 human acute lymphoblastic leukemia cell line, Jurkat human acute T cell leukemia cell line, Raji human Burkitt's lymphoma cell line (negative control), and THP‑1 human acute monocytic leukemia cell line (negative control). PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human CD3 epsilon Monoclonal Antibody (Catalog # MAB100) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (HAF018). A specific band was detected for CD3 epsilon at approximately 21 kDa (as indicated). GAPDH (MAB5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Detection of CD3 epsilon in Human Lymphocytes by Flow Cytometry.
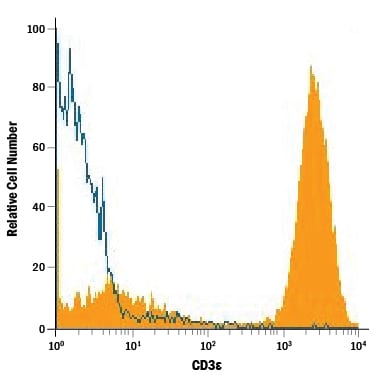
Human peripheral blood lymphocytes were stained with Mouse Anti-Human CD3e Monoclonal Antibody (Catalog # MAB100, filled histogram) or isotype control antibody (Catalog # MAB002, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0102B).Detection of Mouse CD3 epsilon by Flow Cytometry
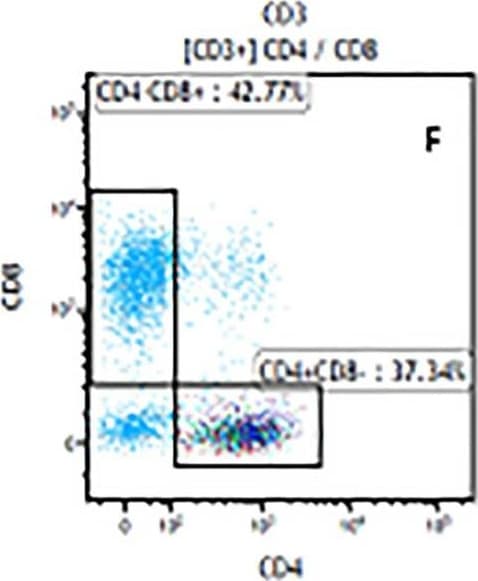
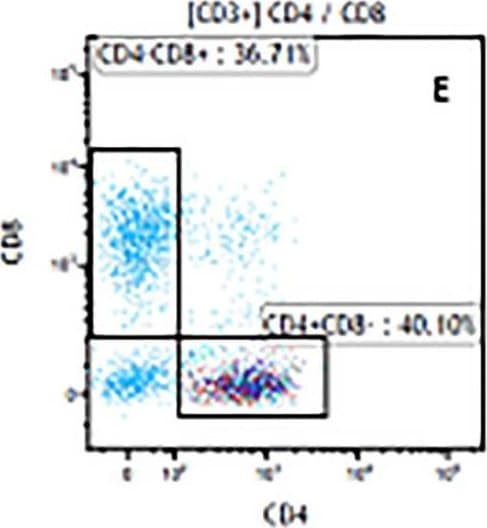
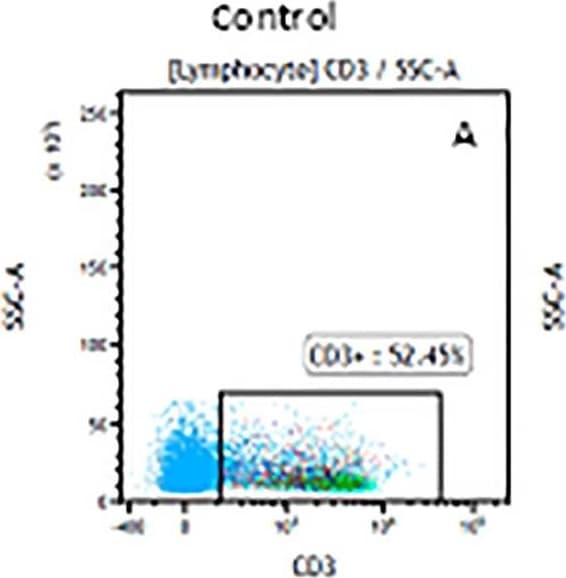
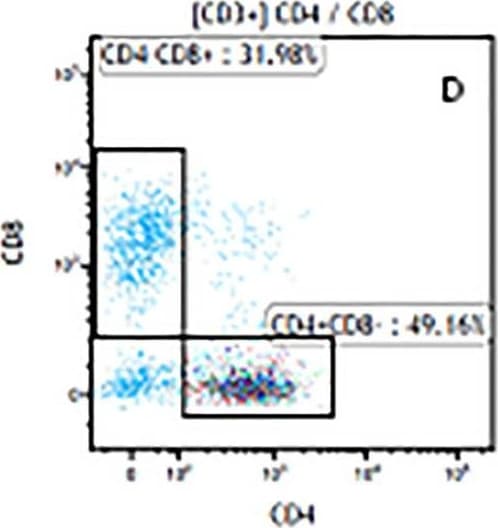
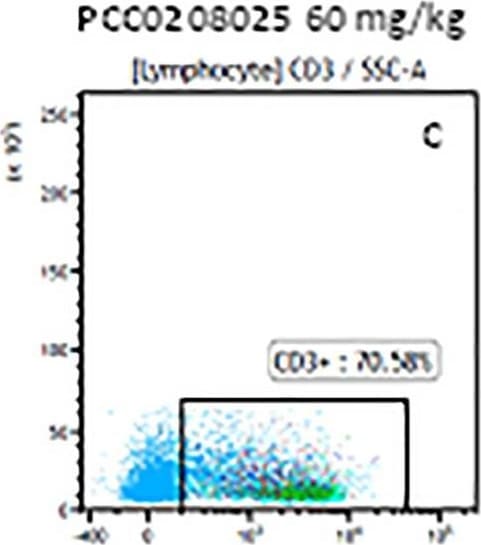
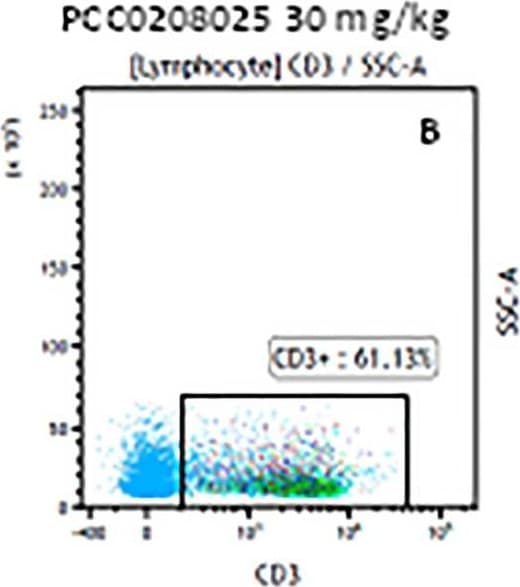
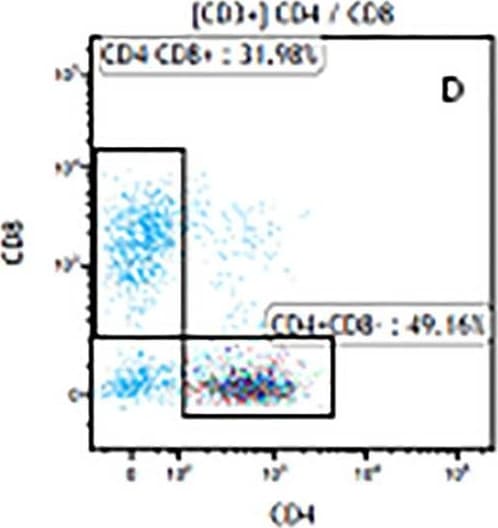
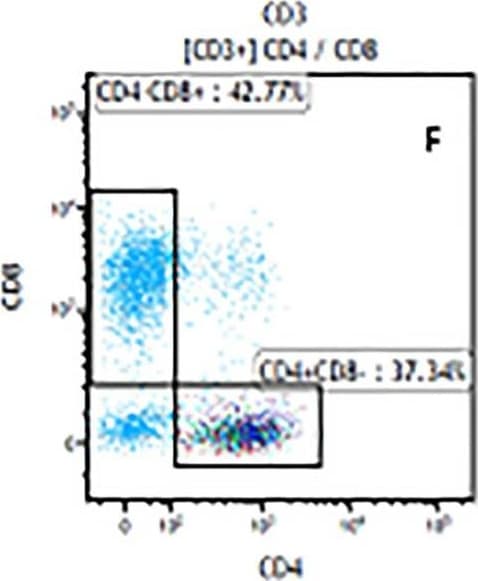
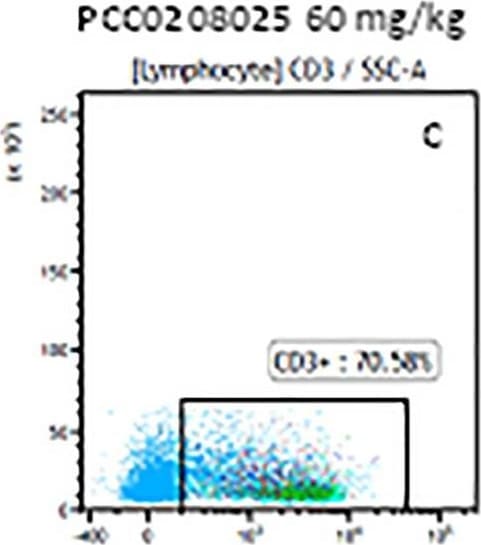
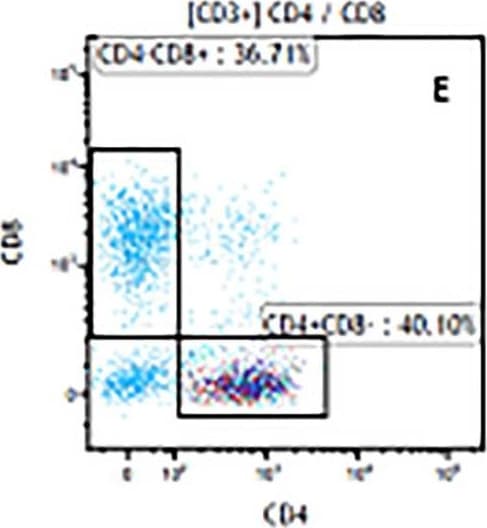
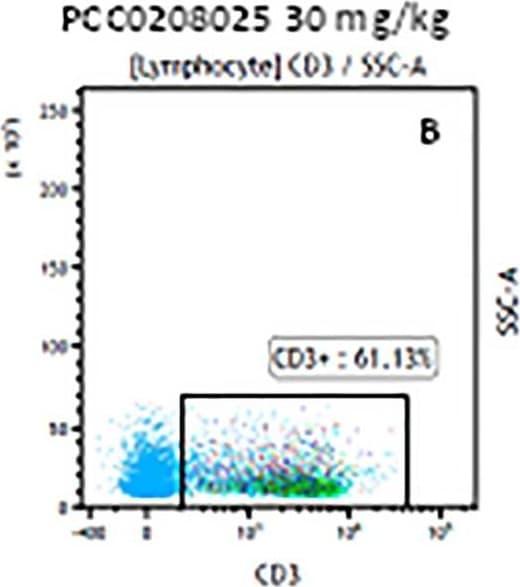
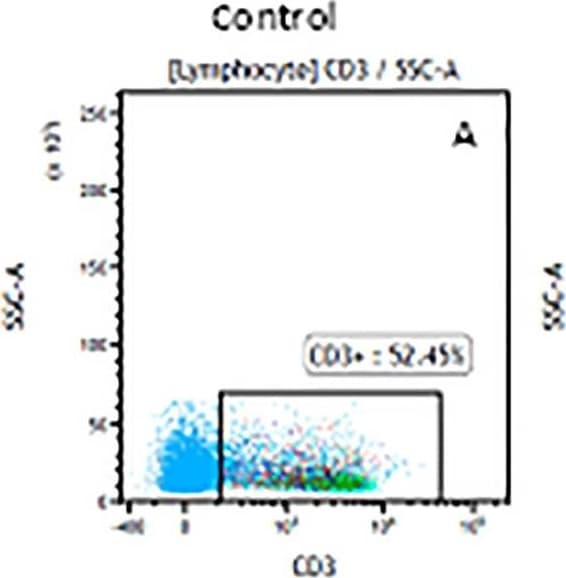
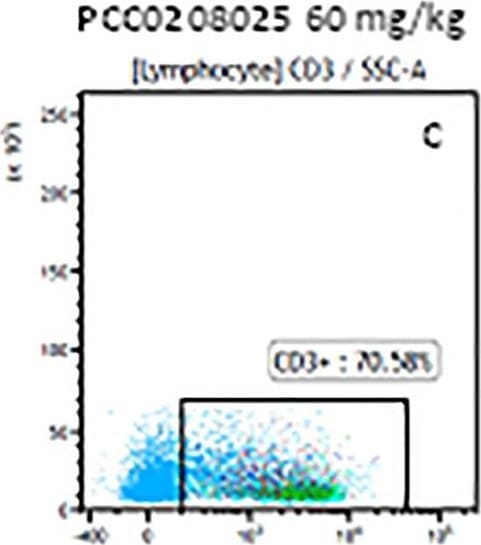
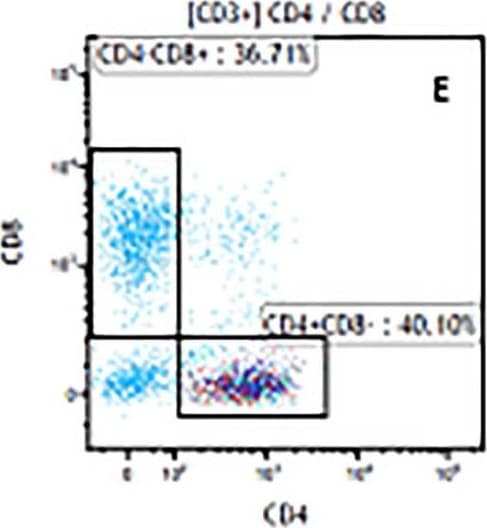
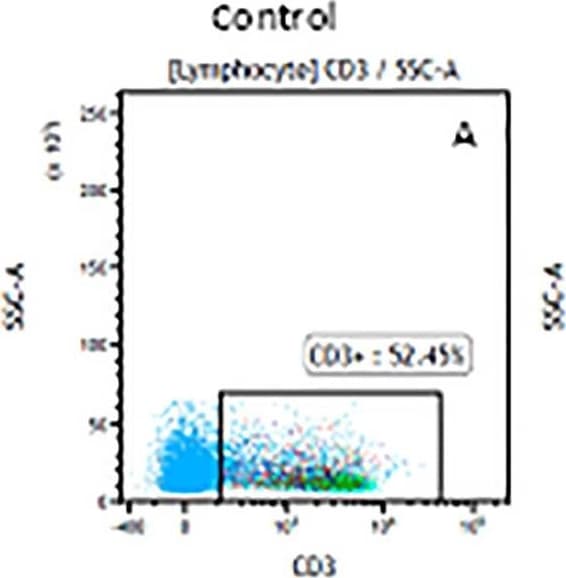
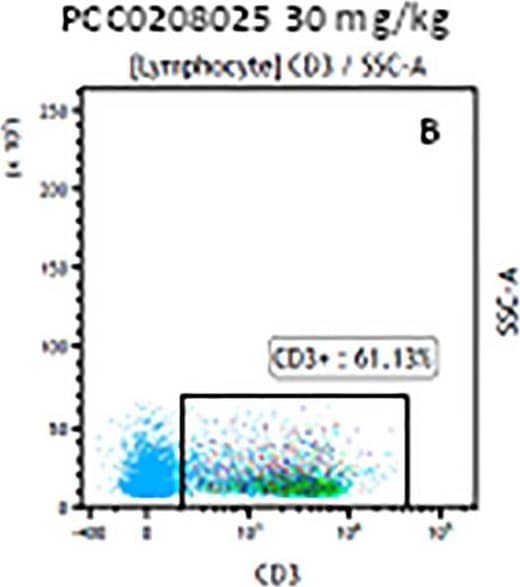
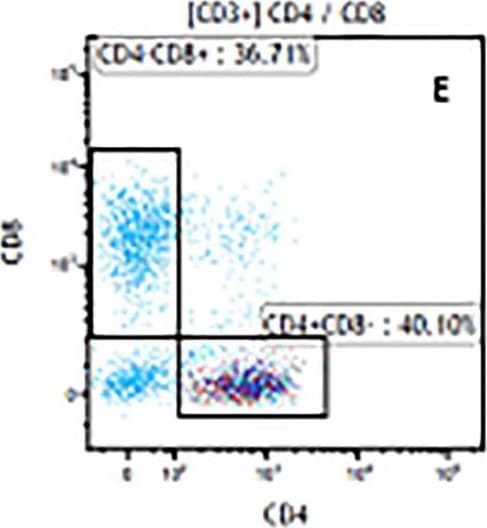
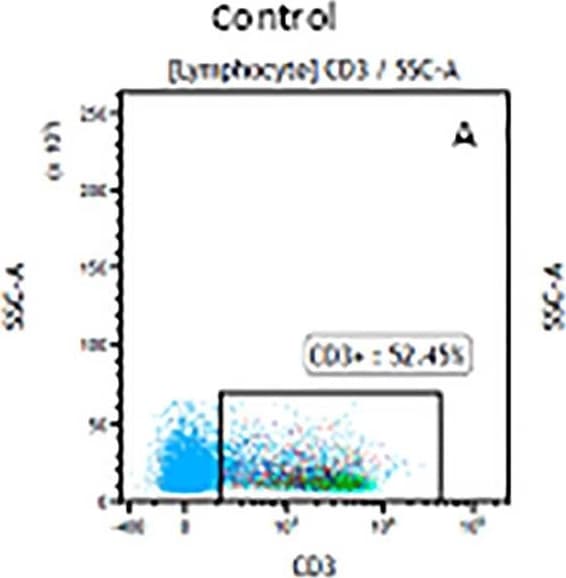
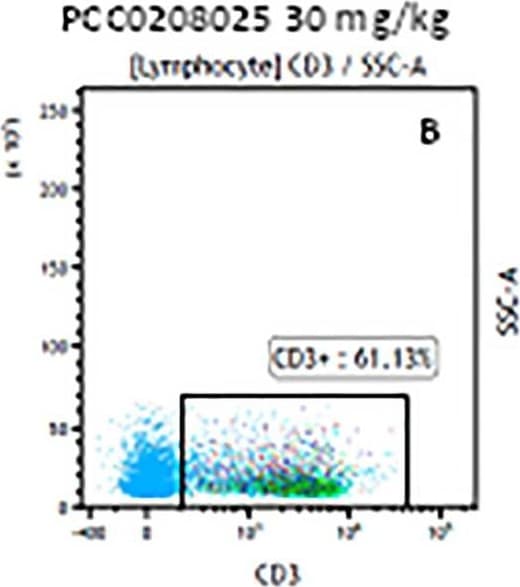
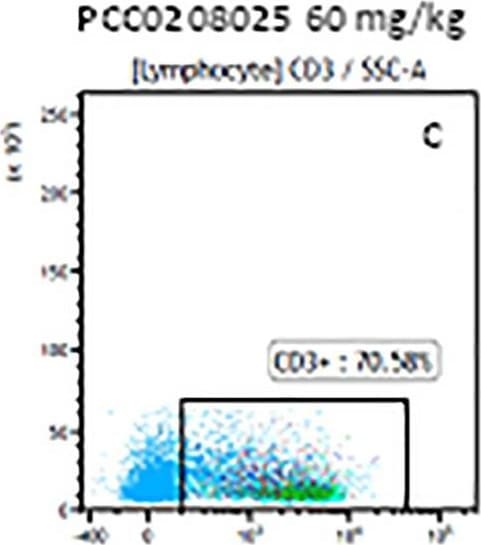
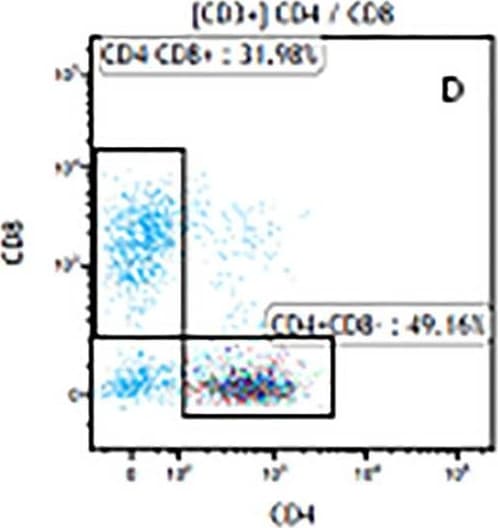
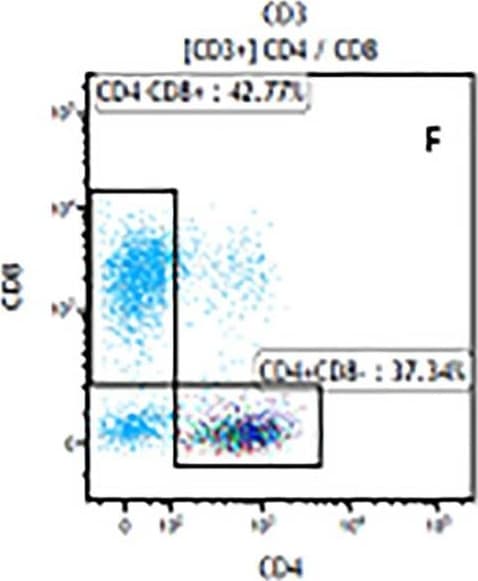
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human CD3 epsilon Antibody by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human CD3 epsilon Antibody by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human CD3 epsilon Antibody by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human CD3 epsilon Antibody by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human CD3 epsilon Antibody by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human CD3 epsilon Antibody by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice.The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CD3 epsilon by Flow Cytometry
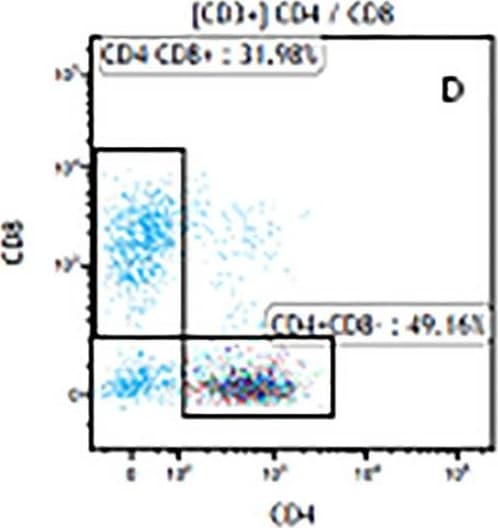
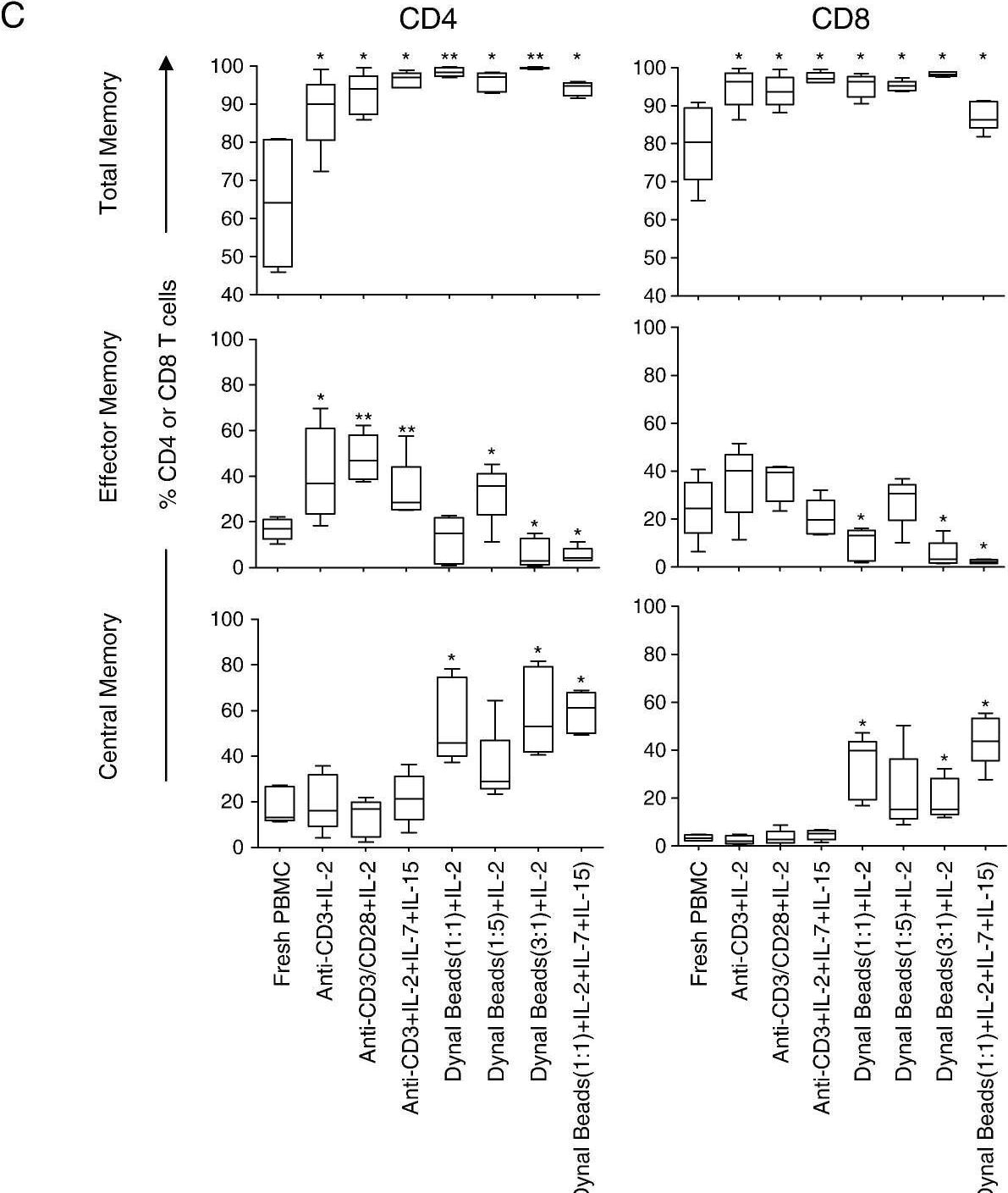
Impact of polyclonal expansion on T cell memory phenotype in blood. Representative plots showing (A) naive and antigen-experienced cells (total memory) based on differential staining with CD27 and CD45RO; and (B) differentiation markers expression (CD45RO, CD27, and CCR7) on CD8+ T cells from blood. Eight distinct memory subsets were defined from these markers: Central memory cells, (CD45RO+CD27+CCR7+), transitional memory cells (CD45RO+CD27+CCR7−), effector memory cells (CD45RO+CD27+CCR7+), CD45RO+CD27−CCR7+, naive T cells (CD45RO−CD27+CCR7+), intermediate memory cells (CD45RO−CD27+CCR7−), effector (CD45RO−CD27−CCR7−) and CD45RO−CD27−CCR7−cells. (C) Comparison of the frequency of total, effector memory and central memory subsets expressed as percentages of total CD4+ and CD8+ T cells in chronically HIV-infected individuals (n = 5) before and after expansion using seven different protocols. Each box and whisker plot shows the median (central line), IQR (outer lines of box) and 5–95% range (error bars) of 5 HIV-infected individuals. * indicates p < 0.05 while ** indicates p < 0.01 using Wilcoxon rank test. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/20149794), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice. The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice. The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice. The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice. The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice. The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CD3 epsilon by Flow Cytometry
The representative figures for T cell subsets counted by flow cytometry from tumors in B16-F10-bearing mice. The cell counts for CD3+ (A, B and C), CD3+CD4+ (D, E and F), CD3+CD8+ (D, E and F), CD4+CD25+CD127low/- (G, H and I) and CD8+IFN-gamma + (J, K and L) T lymphocytes from mouse tumor were determined by flow cytometry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32214351), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human CD3 epsilon Antibody
Application
Recommended Usage
CyTOF-reported
Behbehani, G.K. et al. (2015) Cancer Discov. 5: 988. Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: Human peripheral blood lymphocytes
Sample: Human peripheral blood lymphocytes
Immunocytochemistry
3-25 µg/mL
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs) and Jurkat human acute T cell leukemia cell line
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs) and Jurkat human acute T cell leukemia cell line
Immunoprecipitation
McMichael, A.J. et al. (1987) Leucocyte Typing III: White Cell Differentiation Antigens, Oxford University Press, New York
T Cell Stimulation
This antibody can be used to activate T cells when immobilized at 1-10 μg/mL (100 μL/well).
Western Blot
2 µg/mL
Sample: MOLT‑4 human acute lymphoblastic leukemia cell line and Jurkat human acute T cell leukemia cell line
Sample: MOLT‑4 human acute lymphoblastic leukemia cell line and Jurkat human acute T cell leukemia cell line
Reviewed Applications
Read 4 reviews rated 5 using MAB100 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CD3 epsilon
References
- Beverly, P.C.L. and R.E. Callard (1981) Eur. J. Immunol. 11:329.
Alternate Names
CD3e
Gene Symbol
CD3E
Additional CD3 epsilon Products
Product Documents for Human CD3 epsilon Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CD3 epsilon Antibody
For research use only
Related Research Areas
Citations for Human CD3 epsilon Antibody
Customer Reviews for Human CD3 epsilon Antibody (4)
5 out of 5
4 Customer Ratings
Have you used Human CD3 epsilon Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
4 of
4 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Spleen tissueSpecies: MouseVerified Customer | Posted 04/18/2018
-
Application: ImmunohistochemistrySample Tested: Tumor cell lyastesSpecies: HumanVerified Customer | Posted 04/13/2018
-
Application: Flow CytometrySample Tested: Blood mononuclear cells (PBMCs)Species: HumanVerified Customer | Posted 03/21/2018
-
Application: ELISASample Tested: Human recombinant antibodySpecies: HumanVerified Customer | Posted 10/25/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...