Clusterin, also known as Apolipoprotein J, Sulfated Glycoprotein 2 (SGP-2), TRPM-2, and SP-40, is a secreted multi-functional protein that was named for its ability to induce cellular clustering. It binds a wide range of molecules and may function as a chaperone of misfolded extracellular proteins. It also participates in the control of cell proliferation, apoptosis, and carcinogenesis (1, 2). Clusterin is predominantly expressed in adult testis, ovary, adrenal gland, liver, heart, and brain and in many epithelial tissues during embryonic development (3). Human Clusterin is synthesized as a precursor that contains two coiled coil domains, three nuclear localization signals (NLS), and one heparin binding domain (4-6). Intracellular cleavages of the precursor remove the signal peptide and generate comparably sized alpha and beta chains which are secreted as an 80 kDa N-glycosylated disulfide-linked heterodimer (7, 8). Mature human Clusterin shares 77% amino acid sequence identity with mouse and rat Clusterin. High μg/mL concentrations of Clusterin circulate predominantly as a component of high density lipoprotein particles, and these are internalized and degraded through interactions with LRP-2/Megalin (9, 10). In human, an alternately spliced 50 kDa isoform of Clusterin (nCLU) lacks the signal peptide and remains intracellular (5, 11). This molecule is neither glycosylated nor cleaved into alpha and beta chains (11). In the cytoplasm, nCLU destabilizes the actin cytoskeleton and inhibits NF kappa B activation (12, 13). Cellular exposure to ionizing radiation promotes the translocation of nCLU to the nucleus where it interacts with Ku70 and promotes apoptosis (5, 11). This function contrasts with the cytoprotective effect of secreted Clusterin (14). During colon cancer tumor progression there is a downregulation of the intracellular form and an upregulation of the glycosylated secreted form (11).
Human Clusterin Antibody
R&D Systems | Catalog # MAB2937

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Rat
Applications
Validated:
Immunohistochemistry, Western Blot, Immunoprecipitation
Cited:
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 350227
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Clusterin isoform 1
Asp23-Arg227 (beta) & Ser228-Glu449 (alpha)
Accession # NP_001822
Asp23-Arg227 (beta) & Ser228-Glu449 (alpha)
Accession # NP_001822
Specificity
Detects human Clusterin in direct ELISAs and Western blots. Does not cross-react with recombinant human Clusterin-like 1, recombinant mouse Clusterin, or recombinant rat Clusterin.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Scientific Data Images for Human Clusterin Antibody
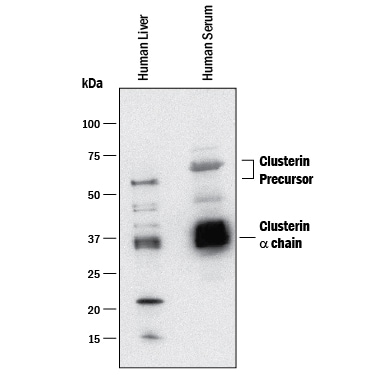
Detection of Human Clusterin by Western Blot.
Western blot shows lysates of human liver tissue and human serum. PVDF membrane was probed with 0.5 µg/mL of Mouse Anti-Human Clusterin Monoclonal Antibody (Catalog # MAB2937) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). Specific bands were detected for Clusterin Precursor at approximately 60-65 kDa and Clusterin a chain at approximately 36 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Clusterin in Mouse Spleen.
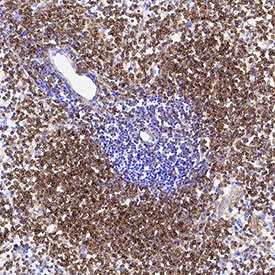
Clusterin was detected in immersion fixed frozen sections of mouse spleen using Mouse Anti-Human Clusterin Monoclonal Antibody (Catalog # MAB2937) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC001). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Clusterin in Human Prostate Cancer Tissue.
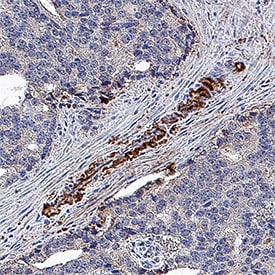
Clusterin was detected in immersion fixed paraffin-embedded sections of human prostate cancer tissue using Mouse Anti-Human Clusterin Monoclonal Antibody (Catalog # MAB2937) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC001). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Human Clusterin by Western Blot
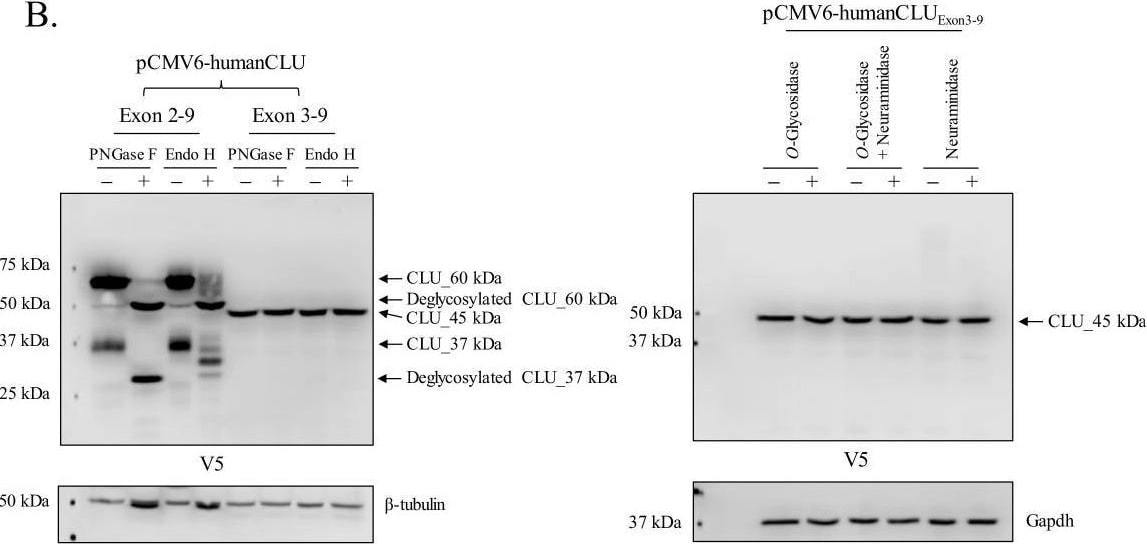
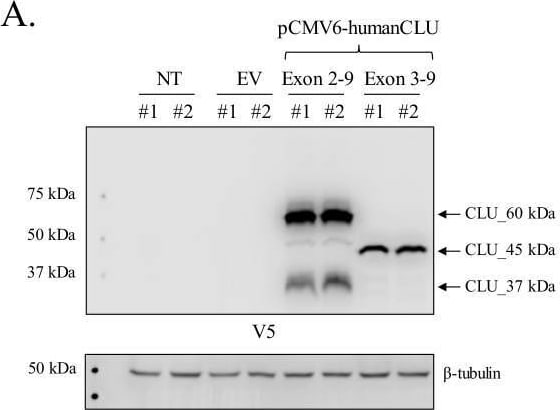
Human CLU_45 kDa is translated from an AUG start site in Exon 3.(A) Empty vector (pCMV6), V5-tagged pCMV6-human CLU Exon 2–9, or Exon 3–9 constructs were transfected into human SH-SY5Y cells. Cell lysates were harvested and probed for CLU immunoreactivity using anti-V5 (left panel) or anti-Clusterin (right panel). (B: left panel) pCMV6-human CLU Exon 2–9 or Exon 3–9 constructs were transfected into human SH-SY5Y cells. The resulting cell lysates were treated with PNGase F or Endo H and probed for CLU immunoreactivity using anti-V5. (B: right panel) Cell lysates harvested from pCMV6-human CLU Exon 3–9-transfected SH-SY5Y cells were treated with O-glycosidase, neuraminidase, or a combination of the two and probed for expression of CLU_45 kDa protein using anti-V5. (C) pCMV6-human CLU Exon 3–9-transfected SH-SY5Y cells were subjected to cellular fractionation using the Qiagen Qproteome Mitochondrial Isolation Kit and analyzed for CLU immunoreactivity using anti-V5. Fraction isolation was confirmed using calnexin (ER), Gapdh (cytosol), lamin A/C and lamin B1 (nucleus), and VDAC and Tom20 (mitochondria). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31738162), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Rat Clusterin by Western Blot
Human CLU_45 kDa is translated from an AUG start site in Exon 3.(A) Empty vector (pCMV6), V5-tagged pCMV6-human CLU Exon 2–9, or Exon 3–9 constructs were transfected into human SH-SY5Y cells. Cell lysates were harvested and probed for CLU immunoreactivity using anti-V5 (left panel) or anti-Clusterin (right panel). (B: left panel) pCMV6-human CLU Exon 2–9 or Exon 3–9 constructs were transfected into human SH-SY5Y cells. The resulting cell lysates were treated with PNGase F or Endo H and probed for CLU immunoreactivity using anti-V5. (B: right panel) Cell lysates harvested from pCMV6-human CLU Exon 3–9-transfected SH-SY5Y cells were treated with O-glycosidase, neuraminidase, or a combination of the two and probed for expression of CLU_45 kDa protein using anti-V5. (C) pCMV6-human CLU Exon 3–9-transfected SH-SY5Y cells were subjected to cellular fractionation using the Qiagen Qproteome Mitochondrial Isolation Kit and analyzed for CLU immunoreactivity using anti-V5. Fraction isolation was confirmed using calnexin (ER), Gapdh (cytosol), lamin A/C and lamin B1 (nucleus), and VDAC and Tom20 (mitochondria). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31738162), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Clusterin Antibody
Application
Recommended Usage
Immunohistochemistry
5-25 µg/mL
Sample: Immersion fixed frozen sections of mouse spleen and immersion fixed paraffin-embedded sections of human prostate cancer tissue
Sample: Immersion fixed frozen sections of mouse spleen and immersion fixed paraffin-embedded sections of human prostate cancer tissue
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Recombinant Human Clusterin (Catalog # 2937-HS), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Recombinant Human Clusterin (Catalog # 2937-HS), see our available Western blot detection antibodies
Western Blot
2 µg/mL
Sample: Human liver tissue and human serum
Sample: Human liver tissue and human serum
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Clusterin
References
- Carver, J.A. et al. (2003) IUBMB Life 55:661.
- Shannan, B. et al. (2006) Cell Death Differ. 13:12.
- French, L.E. et al. (1993) J. Cell Biol. 122:1119.
- Kirszbaum, L. et al. (1989) EMBO J. 8:711.
- Leskov, K.S. et al. (2003) J. Biol. Chem. 278:11590.
- Pankhurst, G.J. et al. (1998) Biochemistry 37:4823.
- Burkey, B.F. et al. (1991) J. Lipid. Res. 32:1039.
- de Silva, H.V. et al. (1990) J. Biol. Chem. 265:14292.
-
Jenne, D.E. et al. (1991) J. Biol. Chem. 266:11030.
-
Kounnas, M.Z. et al. (1995) J. Biol. Chem. 270:13070.
-
Pucci, S. et al. (2004) Oncogene 23:2298.
-
Moretti, R. M. et al. (2007) Cancer Res. 67:10325.
-
Santilli, G. et al. (2003) J. Biol. Chem. 278:38214.
-
Trougakos, I.P. et al. (2004) Cancer Res. 64:1834.
Alternate Names
APOJ, CLI, CLU, SGP-2, SP-40, TRPM-2
Gene Symbol
CLU
UniProt
Additional Clusterin Products
Product Documents for Human Clusterin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Clusterin Antibody
For research use only
Related Research Areas
Citations for Human Clusterin Antibody
Customer Reviews for Human Clusterin Antibody
There are currently no reviews for this product. Be the first to review Human Clusterin Antibody and earn rewards!
Have you used Human Clusterin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...