Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ala21-Thr153
Accession # NP_000577
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Human IL‑2 Antibody
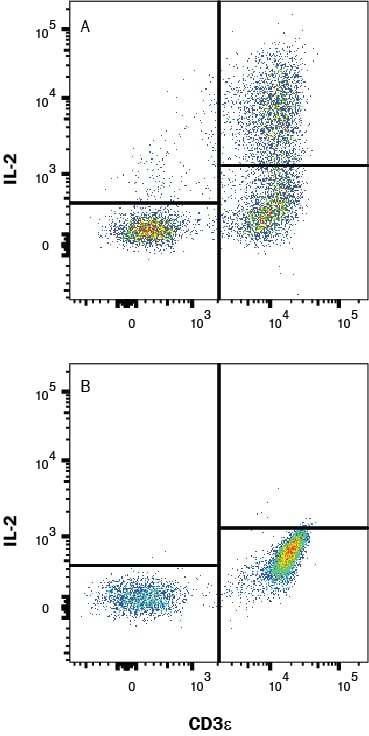
Detection of IL‑2 in PBMC's treated with PMA and Ca2+ ionomycin vs untreated PBMC's by Flow Cytometry
PBMC's treated with PMA (50ng/mL) and Ca2+ ionomycin (200ng/mL) overnight (A) vs untreated PBMC's (B) were stained with Mouse Anti-Human IL‑2 Monoclonal Antibody (Catalog # mab202) and Mouse Anti-Human CD3 epsilon APC‑conjugated Monoclonal Antibody (Catalog # FAB100A) followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0102B). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005). View our protocol for Staining Intracellular Molecules.Cell Proliferation Induced by IL‑2 and Neutralization by Human IL‑2 Antibody.
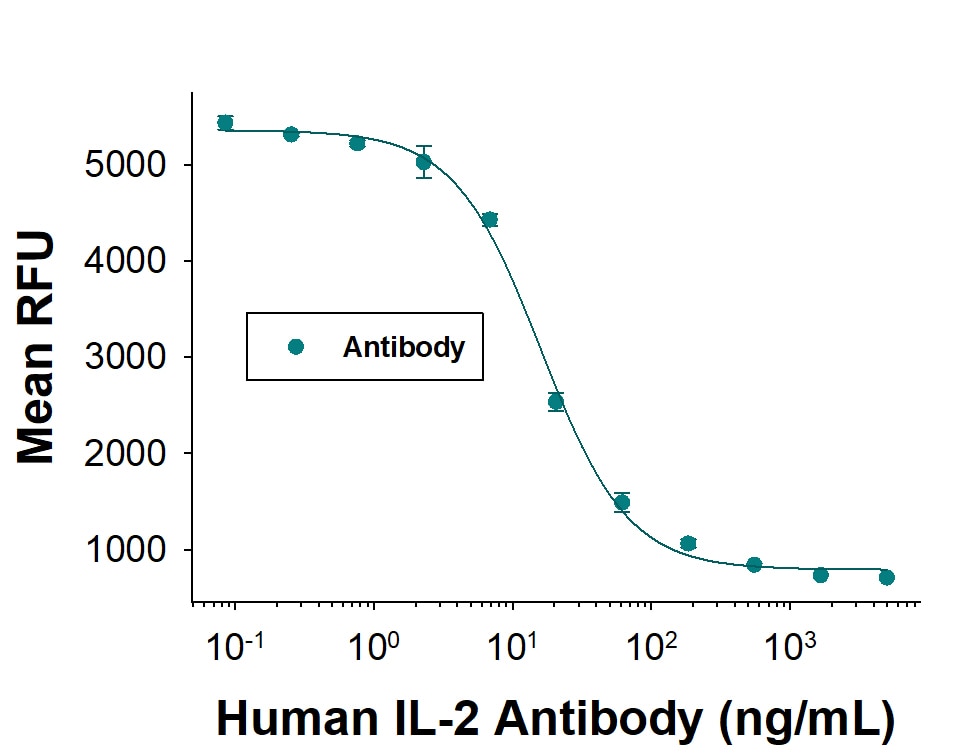
Human IL 2 Antibody (Catalog # MAB202) neutralizes IL-2-induced proliferation in the CTLL-2 mouse cytotoxic T cell line. The Neuralization Dose (ND50) is typically 5.00-60.0 ng/mL.IL‑2 in Human PBMCs.
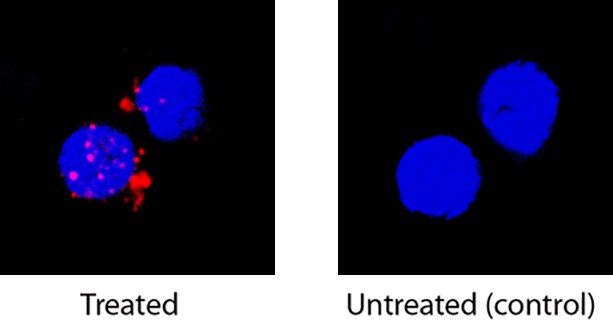
IL-2 was detected in immersion fixed human peripheral blood mononuclear cells (PBMCs) treated with calcium ionomycin and PMA using Mouse Anti-Human IL-2 Monoclonal Antibody (Catalog # MAB202) at 8 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of Human IL-2 by Flow Cytometry
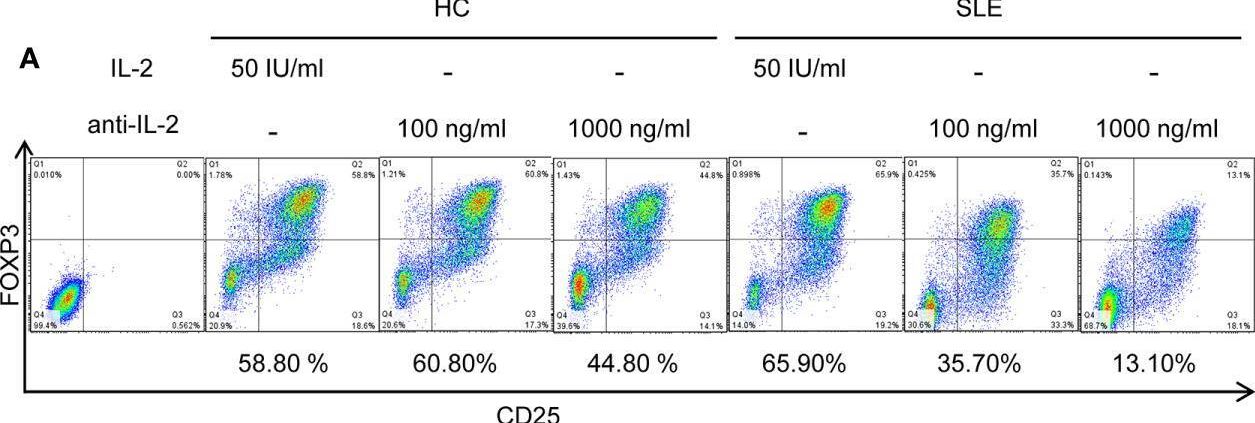
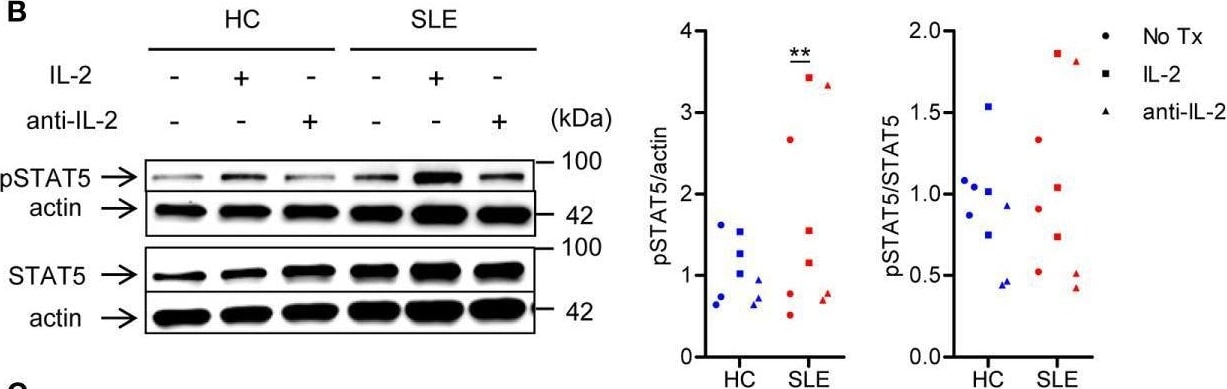
SLE CD4+ T cells are poised to activate IL-2 signaling during Treg differentiation. (A) Naïve CD4+ T cells were isolated from a systemic lupus erythematosus (SLE) patient & matched healthy control (HC) subject, & cultured for 3 days in the presence of anti-CD3/CD28 & TGF-beta (5 ng/ml) with IL-2 (50 IU/ml) or anti-IL-2 (100 or 1,000 ng/ml). The frequency of CD4+CD25+FOXP3+ cells was determined by flow cytometry. Numbers below the plots represent the frequency of CD4+CD25+FOXP3+ Tregs. The dot plots on the left end represent isotype control staining. (B) CD4+ T cells isolated from matched SLE & HC subjects were cultured for 3 days in the presence of anti-CD3/CD28 & TGF-beta (20 ng/ml) with or without IL-2 (100 IU/ml) or anti-IL-2 (100 ng/ml). Total STAT5 & its phosphorylation at tyrosine 694 were detected by immunoblotting. Representative immunoblot staining (left panel). The signal intensity of phospho-STAT5 & total STAT5 was normalized to that of actin. The normalized pSTAT5 signal intensity (middle panel) & the ratio of normalized pSTAT5 signal intensity over normalized STAT5 signal intensity (right panel) from 3 pairs of matched HC & SLE subjects. (C) Untouched T cells from matched SLE & HC subjects were cultured for 3 days without anti-CD3/CD28 stimulation. Expression of CD25 & FOXP3 in CD4+ cells were determined by flow cytometry. Representative flow cytometry dot plots are shown (left panel). Cumulative data of frequency of CD4+CD25+FOXP3+ & CD4+CD25+ cells, mean fluorescence intensity (MFI) of CD25 expression in CD4+ T cells, & the proportion of CD4+CD25+FOXP3+ cells among CD4+CD25+ cells from 17 pairs of matched SLE & HC subjects (right panel). Data were analyzed by a paired two-tailed t-test (*p<0.05, **p<0.01, ****p<0.0001). Image collected & cropped by CiteAb from the following open publication (https://www.frontiersin.org/articles/10.3389/fimmu.2021.635531/full), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human IL-2 by Western Blot
SLE CD4+ T cells are poised to activate IL-2 signaling during Treg differentiation. (A) Naïve CD4+ T cells were isolated from a systemic lupus erythematosus (SLE) patient & matched healthy control (HC) subject, & cultured for 3 days in the presence of anti-CD3/CD28 & TGF-beta (5 ng/ml) with IL-2 (50 IU/ml) or anti-IL-2 (100 or 1,000 ng/ml). The frequency of CD4+CD25+FOXP3+ cells was determined by flow cytometry. Numbers below the plots represent the frequency of CD4+CD25+FOXP3+ Tregs. The dot plots on the left end represent isotype control staining. (B) CD4+ T cells isolated from matched SLE & HC subjects were cultured for 3 days in the presence of anti-CD3/CD28 & TGF-beta (20 ng/ml) with or without IL-2 (100 IU/ml) or anti-IL-2 (100 ng/ml). Total STAT5 & its phosphorylation at tyrosine 694 were detected by immunoblotting. Representative immunoblot staining (left panel). The signal intensity of phospho-STAT5 & total STAT5 was normalized to that of actin. The normalized pSTAT5 signal intensity (middle panel) & the ratio of normalized pSTAT5 signal intensity over normalized STAT5 signal intensity (right panel) from 3 pairs of matched HC & SLE subjects. (C) Untouched T cells from matched SLE & HC subjects were cultured for 3 days without anti-CD3/CD28 stimulation. Expression of CD25 & FOXP3 in CD4+ cells were determined by flow cytometry. Representative flow cytometry dot plots are shown (left panel). Cumulative data of frequency of CD4+CD25+FOXP3+ & CD4+CD25+ cells, mean fluorescence intensity (MFI) of CD25 expression in CD4+ T cells, & the proportion of CD4+CD25+FOXP3+ cells among CD4+CD25+ cells from 17 pairs of matched SLE & HC subjects (right panel). Data were analyzed by a paired two-tailed t-test (*p<0.05, **p<0.01, ****p<0.0001). Image collected & cropped by CiteAb from the following open publication (https://www.frontiersin.org/articles/10.3389/fimmu.2021.635531/full), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human IL-2 by Flow Cytometry
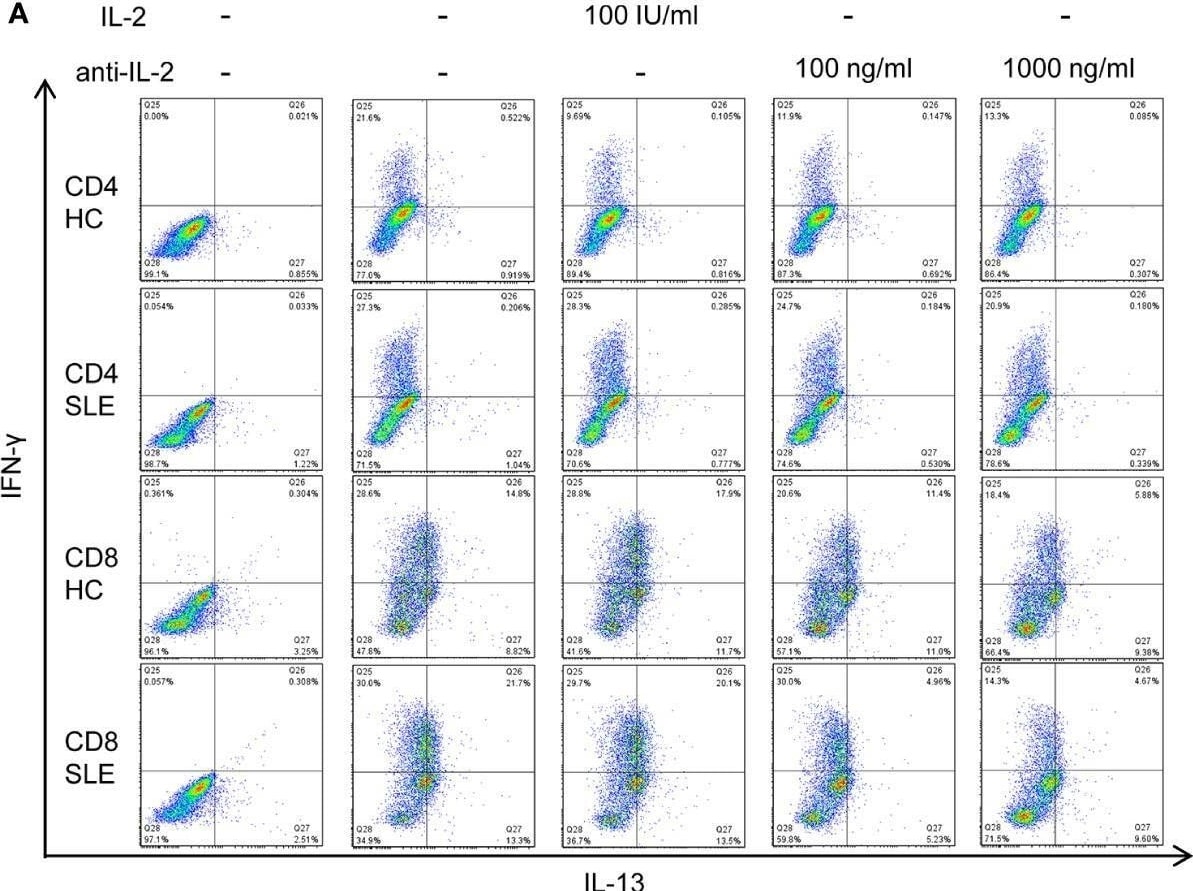
IL-2 expands IL-13+IFN-gamma + CD8+ T cells in systemic lupus erythematosus (SLE). (A) CD4+ and CD8+ T cells from matched SLE and health control (HC) subjects were cultured as described in Figure 2, and IL-13 and IFN-gamma expression was determined by flow cytometry. (B) Cumulative data of mean fluorescence intensity (MFI) of IFN-gamma and the frequency of IFN-gamma + and IL-13+IFN-gamma + cells. Statistical analysis was made by two-way ANOVA followed by Bonferroni’s correction for multiple comparisons (*p<0.05, **p<0.01, ****p<0.0001). Image collected and cropped by CiteAb from the following open publication (https://www.frontiersin.org/articles/10.3389/fimmu.2021.635531/full), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human IL-2 by Western Blot
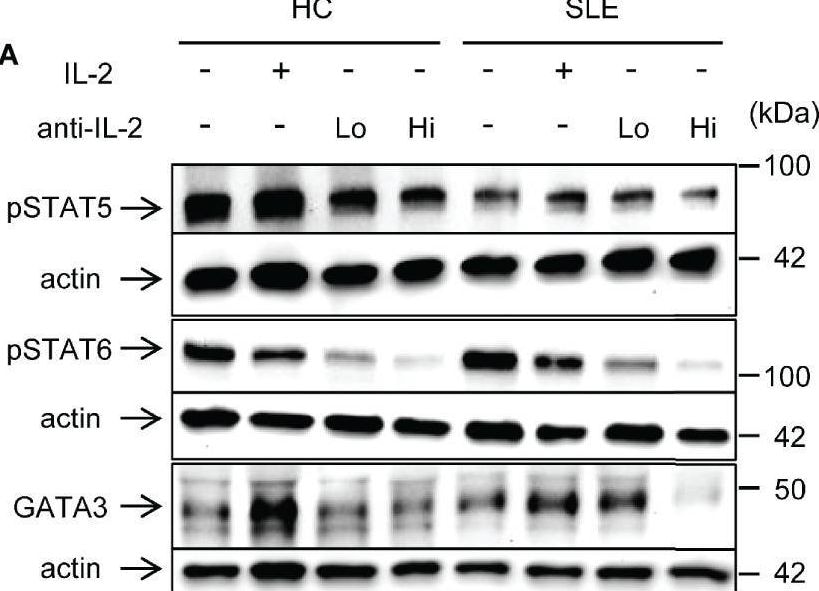
IL-2 induces STAT6 phosphorylation and GATA3 expression in systemic lupus erythematosus (SLE) CD8+ T cells. (A) CD8+ T cells from matched SLE and health control (HC) subjects were cultured as described in Figure 2. Expression of GATA-3 and phosphorylation of STAT5 at tyrosine 694 and STAT6 at tyrosine 641 were detected by immunoblotting. Representative immunoblot staining was presented. Lo and Hi concentrations of anti-IL-2 denote 100 and 1,000 ng/ml, respectively. (B) The signal intensity of phospho-STAT5, phospho-STAT6, and GATA-3 were normalized to that of actin. Cumulative data from 9 pairs of matched HC and SLE subjects. Data were analyzed by a two-tailed t-test (*p<0.05, **p<0.01, ***p<0.001). (C) Pearson’s and Spearman’s correlation analyses were performed to determine the association between the expression of cytokines (IL-13, IL-5, and IFN-gamma ) and transcription factors (phospho-STAT5, phospho-STAT6, and GATA-3). The blue and red plots represent data from HC and SLE patients, respectively. Spearman correlation coefficient was presented for the association between IL-13 and phospho-STAT5, IL-5 and phospho-STAT-5, and IFN-gamma and GATA-3. Pearson correlation coefficient was presented for the remainder of associations. Image collected and cropped by CiteAb from the following open publication (https://www.frontiersin.org/articles/10.3389/fimmu.2021.635531/full), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human IL‑2 Antibody
CyTOF-ready
Immunocytochemistry
Sample: Immersion fixed human peripheral blood mononuclear cells treated with calcium ionomycin and PMA
Intracellular Staining by Flow Cytometry
Sample: Human peripheral blood mononuclear cells treated with PMA and Ca2+ ionomycin, fixed with paraformaldehyde, and permeabilized with saponin
Neutralization
Reviewed Applications
Read 2 reviews rated 4.5 using MAB202 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-2
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional IL-2 Products
Product Documents for Human IL‑2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑2 Antibody
For research use only
Related Research Areas
Citations for Human IL‑2 Antibody
Customer Reviews for Human IL‑2 Antibody (2)
Have you used Human IL‑2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Flow CytometrySample Tested: BUD-8 human fibroblast cell lineSpecies: HumanVerified Customer | Posted 11/16/2020
-
Application: Western BlotSample Tested: SerumSpecies: HumanVerified Customer | Posted 12/12/2019
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Human IL‑2 Antibody
-
Q: Can Catalog # MAB202 (anti-Human IL-2 antibody) be used for neutralization of mouse IL-2?
A: No. MAB202 is not cross-reactive to mouse IL-2 and so cannot be used for neutralization of mouse IL-2.
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-2 Signaling Pathways
IL-2 Signaling Pathways
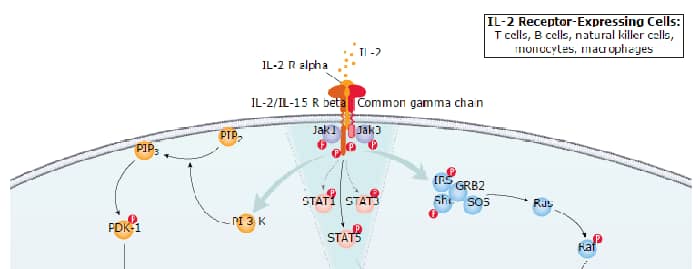 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types

 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th2 Differentiation Pathway
Th2 Differentiation Pathway