Interleukin-4 (IL-4), also known as B cell-stimulatory factor-1, is a monomeric, approximately 13-18 kDa Th2 cytokine that shows pleiotropic effects during immune responses. It is a glycosylated polypeptide that contains three intrachain disulfide bridges and adopts a bundled four alpha -helix structure. Human IL-4 is synthesized with a 24 amino acid (aa) signal sequence. Alternate splicing generates an isoform with a 16 aa internal deletion. Mature human IL-4 shares 55%, 39%, and 43% aa sequence identity with bovine, mouse, and rat IL-4, respectively. Human, mouse, and rat IL-4 are species-specific in their activities. IL-4 exerts its effects through two receptor complexes. The type I receptor, which is expressed on hematopoietic cells, is a heterodimer of the ligand binding IL-4 R alpha and the common gamma chain (a shared subunit of the receptors for IL-2, -7, -9, -15, and -21). The type II receptor on nonhematopoietic cells consists of IL-4 R alpha and IL-13 R alpha 1. The type II receptor also transduces IL-13 mediated signals. IL-4 is primarily expressed by Th2-biased CD4+ T cells, mast cells, basophils, and eosinophils. It promotes cell proliferation, survival, and immunoglobulin class switch to IgG4 and IgE in human B cells, acquisition of the Th2 phenotype by naïve CD4+ T cells, priming and chemotaxis of mast cells, eosinophils, and basophils, and the proliferation and activation of epithelial cells. IL-4 plays a dominant role in the development of allergic inflammation and asthma.
Human IL‑4 Antibody
R&D Systems | Catalog # AF-204-NA


Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Neutralization
Cited:
Immunohistochemistry, Western Blot, Neutralization, Immunocytochemistry, Bioassay
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human IL‑4
His25-Ser153
Accession # NP_000580
His25-Ser153
Accession # NP_000580
Specificity
Detects human IL-4 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human IL‑4 Antibody
Cell Proliferation Induced by IL‑4 and Neutralization by Human IL‑4 Antibody.
Recombinant Human IL-4 (204-IL) stimulates proliferation in the TF-1 human erythroleukemic cell line in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Human IL-4 (0.5 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Human IL-4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-204-NA). The ND50 is typically 0.02-0.1 µg/mL.Applications for Human IL‑4 Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Human IL‑4 (Catalog # 204-IL)
Sample: Recombinant Human IL‑4 (Catalog # 204-IL)
Neutralization
Measured by its ability to neutralize IL‑4-induced proliferation in the TF‑1 human erythroleukemic cell line. Kitamura, T. et al. (1989) J. Cell Physiol. 140:323. The Neutralization Dose (ND50) is typically 0.02-0.1µg/mL in the presence of 0.5 ng/mL Recombinant Human IL‑4.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-4
Long Name
Interleukin 4
Alternate Names
BCDF, BCGF-1, BCGF1, BSF-1, BSF1, IL4, IL4E12
Entrez Gene IDs
Gene Symbol
IL4
UniProt
Additional IL-4 Products
Product Documents for Human IL‑4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑4 Antibody
For research use only
Related Research Areas
Citations for Human IL‑4 Antibody
Customer Reviews for Human IL‑4 Antibody
There are currently no reviews for this product. Be the first to review Human IL‑4 Antibody and earn rewards!
Have you used Human IL‑4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 IL-4 Signaling Pathways
IL-4 Signaling Pathways
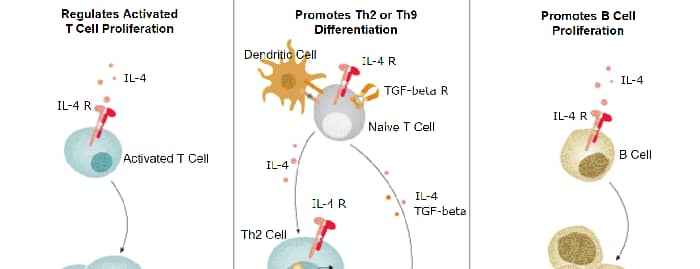
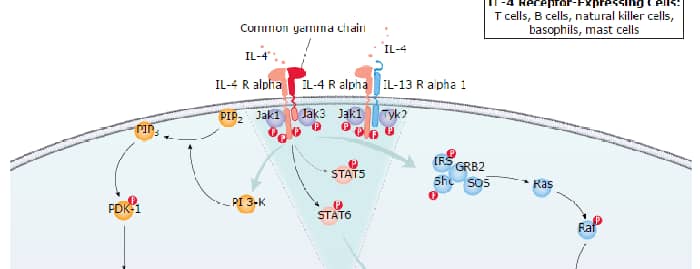 IL-4 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-4 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-9 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-9 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types



